Abstract
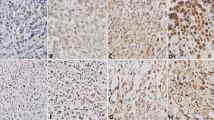
Osteosarcoma is a highly aggressive bone disease with a tendency to metastasize to the lung. The 5-year survival of patients with metastatic osteosarcoma is only 20 %. Many studies have demonstrated SDF-1/CXCR4 and MMP9 play important roles in the metastasis of malignant tumors, including osteosarcoma. The aim of this study was to investigate the association of CXCR4 and MMP9 expression with clinicopathological features and pulmonary metastasis in osteosarcoma. Using tumor tissue microarrays, we analyzed the expression of CXCR4 and MMP9 among 34 primary osteosarcomas with pulmonary metastasis and 62 primary osteosarcomas without metastasis. A median time of 57.5 months (range: 6 to 171 months) follow-up was performed to evaluate tumor metastasis and the patient survival. The prognostic values were determined by univariate Kaplan-Meier survival analysis and multivariate Cox proportional hazard model analysis. The accuracy of oncologic outcome prediction was evaluated by receiver-operating characteristics (ROC) curves (AUC). The expression of CXCR4 and MMP9 was significantly correlated in tumor tissues (P = 0.026). Both CXCR4 and MMP9 were independent predictors for overall survival and metastasis-free survival by Cox multivariate analysis, and high expression for both CXCR4 and MMP9 were even more significant and better biomarkers for osteosarcoma metastasis and survival. The combination of CXCR4 and MMP9 high expression is very likely to be a valuable independent predictor of lung metastasis and survival in osteosarcoma patients.


Similar content being viewed by others
References
Ritter J, Bielack SS. Osteosarcoma. Ann Oncol Off J Eur Soc Med Oncol ESMO. 2010;21 Suppl 7:vii320–5.
Bacci G, Rocca M, Salone M, Balladelli A, Ferrari S, Palmerini E, et al. High grade osteosarcoma of the extremities with lung metastases at presentation: Treatment with neoadjuvant chemotherapy and simultaneous resection of primary and metastatic lesions. J Surg Oncol. 2008;98:415–20.
Kager L, Zoubek A, Potschger U, Kastner U, Flege S, Kempf-Bielack B, et al. Primary metastatic osteosarcoma: presentation and outcome of patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol Off J Am Soc Clin Oncol. 2003;21:2011–8.
Balkwill F. Cancer and the chemokine network. Nat Rev Cancer. 2004;4:540–50.
Teicher BA, Fricker SP. Cxcl12 (sdf-1)/cxcr4 pathway in cancer. Clin Cancer Res Off J Am Assoc Cancer Res. 2010;16:2927–31.
Muller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME, et al. Involvement of chemokine receptors in breast cancer metastasis. Nature. 2001;410:50–6.
Taichman RS, Cooper C, Keller ET, Pienta KJ, Taichman NS, McCauley LK. Use of the stromal cell-derived factor-1/cxcr4 pathway in prostate cancer metastasis to bone. Cancer Res. 2002;62:1832–7.
Yang YC, Lee ZY, Wu CC, Chen TC, Chang CL, Chen CP. Cxcr4 expression is associated with pelvic lymph node metastasis in cervical adenocarcinoma. Int J Gynecol Cancer Off J Int Gynecol Cancer Soc. 2007;17:676–86.
Wagner PL, Hyjek E, Vazquez MF, Meherally D, Liu YF, Chadwick PA, et al. Cxcl12 and cxcr4 in adenocarcinoma of the lung: association with metastasis and survival. J Thorac Cardiovasc Surg. 2009;137:615–21.
Perissinotto E, Cavalloni G, Leone F, Fonsato V, Mitola S, Grignani G, et al. Involvement of chemokine receptor 4/stromal cell-derived factor 1 system during osteosarcoma tumor progression. Clin Cancer Res Off J Am Assoc Cancer Res. 2005;11:490–7.
Egeblad M, Werb Z. New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer. 2002;2:161–74.
Shuman Moss LA, Jensen-Taubman S, Stetler-Stevenson WG. Matrix metalloproteinases: changing roles in tumor progression and metastasis. Am J Pathol. 2012;181:1895–9.
Baldwin RM, Haghandish N, Daneshmand M, Amin S, Paris G, Falls TJ, et al. Protein arginine methyltransferase 7 promotes breast cancer cell invasion through the induction of mmp9 expression. Oncotarget. 2015;6:3013–32.
Chen Y, Jiang T, Mao A, Xu J. Esophageal cancer stem cells express plgf to increase cancer invasion through mmp9 activation. Tumour Biol J Int Soc Oncodev Biol Med. 2014;35:12749–55.
Huang Q, Lan F, Wang X, Yu Y, Ouyang X, Zheng F, et al. Il-1beta-induced activation of p38 promotes metastasis in gastric adenocarcinoma via upregulation of ap-1/c-fos, mmp2 and mmp9. Mol Cancer. 2014;13:18.
Wang X, Lu H, Urvalek AM, Li T, Yu L, Lamar J, et al. Klf8 promotes human breast cancer cell invasion and metastasis by transcriptional activation of mmp9. Oncogene. 2011;30:1901–11.
Chinni SR, Sivalogan S, Dong Z, Filho JC, Deng X, Bonfil RD, et al. Cxcl12/cxcr4 signaling activates akt-1 and mmp-9 expression in prostate cancer cells: The role of bone microenvironment-associated cxcl12. Prostate. 2006;66:32–48.
Nian WQ, Chen FL, Ao XJ, Chen ZT. Cxcr4 positive cells from lewis lung carcinoma cell line have cancer metastatic stem cell characteristics. Mol Cell Biochem. 2011;355:241–8.
Lee CC, Lai JH, Hueng DY, Ma HI, Chung Y, Sun YY, et al. Disrupting the cxcl12/cxcr4 axis disturbs the characteristics of glioblastoma stem-like cells of rat rg2 glioblastoma. Cancer Cell Int. 2013;13:85.
Marchesi F, Monti P, Leone BE, Zerbi A, Vecchi A, Piemonti L, et al. Increased survival, proliferation, and migration in metastatic human pancreatic tumor cells expressing functional cxcr4. Cancer Res. 2004;64:8420–7.
Brand S, Dambacher J, Beigel F, Olszak T, Diebold J, Otte JM, et al. Cxcr4 and cxcl12 are inversely expressed in colorectal cancer cells and modulate cancer cell migration, invasion and mmp-9 activation. Exp Cell Res. 2005;310:117–30.
Samara GJ, Lawrence DM, Chiarelli CJ, Valentino MD, Lyubsky S, Zucker S, et al. Cxcr4-mediated adhesion and mmp-9 secretion in head and neck squamous cell carcinoma. Cancer Lett. 2004;214:231–41.
Murphy PM. Chemokines and the molecular basis of cancer metastasis. N Engl J Med. 2001;345:833–5.
Oda Y, Yamamoto H, Tamiya S, Matsuda S, Tanaka K, Yokoyama R, et al. Cxcr4 and vegf expression in the primary site and the metastatic site of human osteosarcoma: Analysis within a group of patients, all of whom developed lung metastasis. Modern Pathol Off J US Can Acad Pathol Inc. 2006;19:738–45.
Laverdiere C, Hoang BH, Yang R, Sowers R, Qin J, Meyers PA, et al. Messenger rna expression levels of cxcr4 correlate with metastatic behavior and outcome in patients with osteosarcoma. Clin Cancer Res Off J Am Assoc Cancer Res. 2005;11:2561–7.
Mira E, Lacalle RA, Buesa JM, de Buitrago GG, Jimenez-Baranda S, Gomez-Mouton C, et al. Secreted mmp9 promotes angiogenesis more efficiently than constitutive active mmp9 bound to the tumor cell surface. J Cell Sci. 2004;117:1847–57.
Choi SH, Lee HJ, Jin YB, Jang J, Kang GY, Lee M, et al. Mmp9 processing of hspb1 regulates tumor progression. PLoS One. 2014;9:e85509.
Hiratsuka S, Nakamura K, Iwai S, Murakami M, Itoh T, Kijima H, et al. Mmp9 induction by vascular endothelial growth factor receptor-1 is involved in lung-specific metastasis. Cancer Cell. 2002;2:289–300.
van Kempen LC, Coussens LM. Mmp9 potentiates pulmonary metastasis formation. Cancer Cell. 2002;2:251–2.
Liang S, Ren Z, Han X, Yang J, Shan L, Li L, et al. Pla2g16 expression in human osteosarcoma is associated with pulmonary metastasis and poor prognosis. PLoS One. 2015;10:e0127236.
Meyers PA, Heller G, Healey JH, Huvos A, Applewhite A, Sun M, et al. Osteogenic sarcoma with clinically detectable metastasis at initial presentation. J Clin Oncol Off J Am Soc Clin Oncol. 1993;11:449–53.
Jaffe N, Carrasco H, Raymond K, Ayala A, Eftekhari F. Can cure in patients with osteosarcoma be achieved exclusively with chemotherapy and abrogation of surgery? Cancer. 2002;95:2202–10.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhiwu Ren and Shoulei Liang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ren, Z., Liang, S., Yang, J. et al. Coexpression of CXCR4 and MMP9 predicts lung metastasis and poor prognosis in resected osteosarcoma. Tumor Biol. 37, 5089–5096 (2016). https://doi.org/10.1007/s13277-015-4352-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4352-8