Abstract
Background
The six-transmembrane protein of the prostate 2 (STAMP2) is crucial for damage-induced inflammation. Excessive inflammatory responses in microglia have been discovered as the critical factor in triggering neurological diseases. However, inconclusive evidence suggests that STAMP2 alleviates neuroinflammatory diseases by regulating the inflammatory state of microglia.
Objectives
To ascertain whether the favorable effects of STAMP2 on neuroinflammation act through microglia.
Results
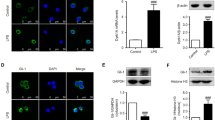
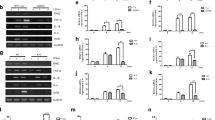
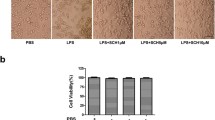
STAMP2 served as an anti-inflammatory modulator to inhibit the production of inflammatory and reactive oxygen species (ROS) factors and attenuated neurotoxicity. Downregulation of STAMP2 increased the levels of IL-1β, IL-6, ROS, and glutamate, and exogenous STAMP2 knock-in rescued both the exacerbated inflammation and oxidative stress induced by STAMP2 knockdown in BV-2 cells. Mechanistic study showed that STAMP2 siRNA accelerated the expression of p-P65 and p-IkBα, whereas STAMP2OE interrupted the NF-kB translocation. In addition, STAMP2 could reduce neurotoxicity by promoting cell survival and suppressing apoptosis.
Conclusion
These findings revealed that STAMP2 is involved in regulating the LPS-NF-kB signal pathway to modulate inflammatory processes in microglia cells, which could represent a possible therapeutic therapy for neurodegenerative disorders.
Graphical abstract





Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and any raw data can be obtained from the corresponding author upon request.
References
Batool M et al (2020) The six-transmembrane protein Stamp2 ameliorates pulmonary vascular remodeling and pulmonary hypertension in mice. Basic Res Cardiol 115:68
Butovsky O et al (2006) Microglia activated by IL-4 or IFN-gamma differentially induce neurogenesis and oligodendrogenesis from adult stem/progenitor cells. Mol Cell Neurosci 31:149–160
Calvo-Rodriguez M et al (2017) Aging and amyloid beta oligomers enhance TLR4 expression, LPS-induced Ca(2+) responses, and neuron cell death in cultured rat hippocampal neurons. J Neuroinflamm 14:24
Casula M et al (2011) Toll-like receptor signaling in amyotrophic lateral sclerosis spinal cord tissue. Neuroscience 179:233–243
Cerbai F et al (2012) The neuron-astrocyte-microglia triad in normal brain ageing and in a model of neuroinflammation in the rat hippocampus. PLoS ONE 7:e45250
De Caris MG et al (2020) Blueberry counteracts BV-2 microglia morphological and functional switch after LPS challenge. Nutrients 12:1830
Ding L et al (2021) Glutaminase in microglia: a novel regulator of neuroinflammation. Brain Behav Immun 92:139–156
Dohi K et al (2010) Gp91phox (NOX2) in classically activated microglia exacerbates traumatic brain injury. J Neuroinflamm 7:41
Floyd RA (1999) Neuroinflammatory processes are important in neurodegenerative diseases: an hypothesis to explain the increased formation of reactive oxygen and nitrogen species as major factors involved in neurodegenerative disease development. Free Radic Biol Med 26:1346–1355
Gao X et al (2020) Beta-naphthoflavone inhibits LPS-induced inflammation in BV-2 cells via AKT/Nrf-2/HO-1-NF-kappaB signaling axis. Immunobiology 225:151965
Gui Y, Sun L, Liu R, Luo J (2021) Pachymic acid inhibits inflammation and cell apoptosis in lipopolysaccharide (LPS)-induced rat model with pneumonia by regulating NF-kappaB and MAPK pathways. Allergol Immunopathol (madr) 49:87–93
He Y et al (2021) Coniferyl aldehyde alleviates LPS-induced WI-38 cell apoptosis and inflammation injury via JAK2-STAT1 pathway in acute pneumonia. Allergol Immunopathol (madr) 49:72–77
Hu X et al (2015) Microglial and macrophage polarization-new prospects for brain repair. Nat Rev Neurol 11:56–64
Kim YS, Joh TH (2006) Microglia, major player in the brain inflammation: their roles in the pathogenesis of Parkinson’s disease. Exp Mol Med 38:333–347
Koning N, Bo L, Hoek RM, Huitinga I (2007) Downregulation of macrophage inhibitory molecules in multiple sclerosis lesions. Ann Neurol 62:504–514
Krause DL, Muller N (2010) Neuroinflammation, microglia and implications for anti-inflammatory treatment in Alzheimer's disease. Int J Alzheimers Dis 2010:732806. https://doi.org/10.4061/2010/732806
Lai Y, Feldman KL, Clark RS (2005) Enzyme-linked immunosorbent assays (ELISAs). Crit Care Med 33:S433-434
Lana D et al (2021) The emerging role of the interplay among astrocytes, microglia, and neurons in the hippocampus in health and disease. Front Aging Neurosci 13:651973
Lee YJ et al (2012) Inhibitory effect of 4-O-methylhonokiol on lipopolysaccharide-induced neuroinflammation, amyloidogenesis and memory impairment via inhibition of nuclear factor-kappaB in vitro and in vivo models. J Neuroinflamm 9:35
Lee SW et al (2018) Palmitate induces lipoapoptosis in Schwann cells through ROS generation-mediated STAMP2 downregulation. Biochem Biophys Res Commun 503:1260–1266
Lelakova V et al (2020) Therapeutic potential of prenylated stilbenoid macasiamenene F through its anti-inflammatory and cytoprotective effects on LPS-challenged monocytes and microglia. J Ethnopharmacol 263:113147
Liu S, Li J, Hu L (2021) MiR-216a-5p alleviates LPS-induced inflammation in the human bronchial epithelial cell by inhibition of TGF-beta1 signaling via down-regulating TGFBR2. Allergol Immunopathol (madr) 49:64–71
Miron VE et al (2013) M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci 16:1211–1218
Oh YJ et al (2018) Cilostazol improves HFD-induced hepatic steatosis by upregulating hepatic STAMP2 expression through AMPK. Mol Pharmacol 94:1401–1411
Ohgami RS, Campagna DR, McDonald A, Fleming MD (2006) The Steap proteins are metalloreductases. Blood 108:1388–1394
Puigdellivol M, Allendorf DH, Brown GC (2020) Sialylation and galectin-3 in microglia-mediated neuroinflammation and neurodegeneration. Front Cell Neurosci 14:162
Sato K, Tatsunami R, Wakame K (2021) Epalrestat suppresses inflammatory response in lipopolysaccharide-stimulated RAW264.7 cells. Allergol Immunopathol (madr) 49:1–8
Shayo SC et al (2022) Dietary obesity and glycemic excursions cause a parallel increase in STEAP4 and pro-inflammatory gene expression in murine PBMCs. Diabetol Int 13:358–371
ten Freyhaus H et al (2012) Stamp2 controls macrophage inflammation through nicotinamide adenine dinucleotide phosphate homeostasis and protects against atherosclerosis. Cell Metab 16:81–89
Wang X et al (2014) Pseudoginsenoside-F11 (PF11) exerts anti-neuroinflammatory effects on LPS-activated microglial cells by inhibiting TLR4-mediated TAK1/IKK/NF-kappaB, MAPKs and Akt signaling pathways. Neuropharmacology 79:642–656
Wellen KE et al (2007) Coordinated regulation of nutrient and inflammatory responses by STAMP2 is essential for metabolic homeostasis. Cell 129:537–548
Yuan L et al (2016) Oxytocin inhibits lipopolysaccharide-induced inflammation in microglial cells and attenuates microglial activation in lipopolysaccharide-treated mice. J Neuroinflamm 13:77
Zhou J et al (2013) Steap4 plays a critical role in osteoclastogenesis in vitro by regulating cellular iron/reactive oxygen species (ROS) levels and cAMP response element-binding protein (CREB) activation. J Biol Chem 288:30064–30074
Zhuang L et al (2021) Dynamic changes of inflammation and apoptosis in cerebral ischemia-reperfusion injury in mice investigated by ferumoxytol-enhanced magnetic resonance imaging. Mol Med Rep 23:1–13
Acknowledgements
Not applicable.
Funding
No funding was used in this study.
Author information
Authors and Affiliations
Contributions
ZC and JJ designed the study and carried them out, ZC and JJ supervised the data collection, analyzed the data, and interpreted the data, ZC, JJ and QL prepared the manuscript for publication and reviewed the draft of the manuscript. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author Zengbian Chen declares that he/she has no conflict of interest; the author Jie Jin declares that he/she has no conflict of interest; the author Qi Lu declares that he/she has no conflict of interest.
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Z., Jin, J. & Lu, Q. STAMP2 alleviates microglial neurotoxicity by inhibiting LPS-induced NF-κB activation. Mol. Cell. Toxicol. 20, 335–342 (2024). https://doi.org/10.1007/s13273-023-00346-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-023-00346-1