Abstract
Background
Sevoflurane has obvious side effects during anesthesia, which caused neuronal apoptosis and neuroinflammation. How to reduce sevoflurane-induced neurotoxicity is an urgent problem to be solved.
Objective
To assess the role of USP18 in anesthetic sevoflurane-induced neurotoxicity, detect its effects on neuroinflammation, oxidative stress, and apoptosis, and explore the related potential signaling pathway.
Results
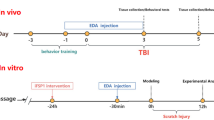
USP18 alleviated sevoflurane-induced learning and memory dysfunction and changed distribution of neurons. USP18 also reduced sevoflurane-induced neuroinflammation. We further found that USP18 reduced the sevoflurane-induced oxidative stress in the mice model. USP18 also attenuated sevoflurane-induced neuronal apoptosis. Mechanically, USP18 reduced sevoflurane-induced neurotoxicity via AKT and NF-κB pathway.
Conclusion
USP18 was involved in the pathology of sevoflurane-induced neurotoxicity.





Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
Arimoto KI et al (2017) STAT2 is an essential adaptor in USP18-mediated suppression of type I interferon signaling. Nat Struct Mol Biol 24:279–289. https://doi.org/10.1038/nsmb.3378
Diao W et al (2020) USP18 promotes cell proliferation and suppressed apoptosis in cervical cancer cells via activating AKT signaling pathway. BMC Cancer 20:741. https://doi.org/10.1186/s12885-020-07241-1
He H et al (2020) Polyphyllin VII induces apoptotic cell death via inhibition of the PI3K/Akt and NFkappaB pathways in A549 human lung cancer cells. Mol Med Rep 21:597–606. https://doi.org/10.3892/mmr.2019.10879
Hu B, Ge C, Zhu C (2021) USP18 negatively regulates and inhibits lipopolysaccharides-induced sepsis by targeting TAK1 activity. Int Immunol. https://doi.org/10.1093/intimm/dxab029
Kung YL et al (2021) Cardioprotective potential of amygdalin against angiotensin II induced cardiac hypertrophy, oxidative stress and inflammatory responses through modulation of Nrf2 and NF-kappaB activation. Environ Toxicol 36:926–934. https://doi.org/10.1002/tox.23094
Lee WT, Nakamura M, Cho JH, Jang IS (2021) Sevoflurane excites nociceptive sensory neurons by inhibiting K(+) conductances in rats. Neurosci Lett 756:135951. https://doi.org/10.1016/j.neulet.2021.135951
Liu W et al (2021) Deubiquitinase USP18 regulates reactive astrogliosis by stabilizing SOX9. Glia 69:1782–1798. https://doi.org/10.1002/glia.23992
Lu T et al (2010) Regulation of NF-kappaB by NSD1/FBXL11-dependent reversible lysine methylation of p65. Proc Natl Acad Sci USA 107:46–51. https://doi.org/10.1073/pnas.0912493107
Ma YM et al (2016) Novel CHOP activator LGH00168 induces necroptosis in A549 human lung cancer cells via ROS-mediated ER stress and NF-kappaB inhibition. Acta Pharmacol Sin 37:1381–1390. https://doi.org/10.1038/aps.2016.61
Osei Kuffour E et al (2018) USP18 (UBP43) abrogates p21-Mediated Inhibition of HIV-1. J Virol. https://doi.org/10.1128/JVI.00592-18
Pan A et al (2021) USP18-deficiency in cervical carcinoma is crucial for the malignant behavior of tumor cells in an ERK signal-dependent manner. Oncol Lett 21:421. https://doi.org/10.3892/ol.2021.12682
Shu Q, Zhao X, Geng X, Wang X (2020) CD82 Aggravates sevoflurane-induced neurotoxicity by regulating TRPM7 in developing neurons. Signa Vitae 16:142–147
Tan Y, Zhou G, Wang X, Chen W, Gao H (2018) USP18 promotes breast cancer growth by upregulating EGFR and activating the AKT/Skp2 pathway. Int J Oncol 53:371–383. https://doi.org/10.3892/ijo.2018.4387
Velasco Gallego ML, Martin Jurado O, Hatt JM (2021) Effects of isoflurane and sevoflurane alone and in combination with butorphanol or medetomidine on the bispectral index in chickens. BMC Vet Res 17:201. https://doi.org/10.1186/s12917-021-02895-w
Wang H et al (2021a) Restoring VTA DA neurons excitability accelerates emergence from sevoflurane general anesthesia of anxiety state. Biochem Biophys Res Commun 565:21–28. https://doi.org/10.1016/j.bbrc.2021.05.079
Wang F, Li C, Shao J, Ma J (2021b) Sevoflurane induces inflammation of microglia in hippocampus of neonatal rats by inhibiting Wnt/beta-Catenin/CaMKIV pathway. J Pharmacol Sci 146:105–115. https://doi.org/10.1016/j.jphs.2021.02.004
Wang HV, Forestier S, Corces VG (2021c) Exposure to sevoflurane results in changes of transcription factor occupancy in sperm and inheritance of autism. Biol Reprod. https://doi.org/10.1093/biolre/ioab097
Williamson PN et al (2021) Review of the development of haemodynamic modelling techniques to capture flow behaviour in arteries affected by aneurysm, atherosclerosis and stenting. J Biomech Eng. https://doi.org/10.1115/1.4053082
Xie L, Fang Q, Wei X, Zhou L, Wang S (2021) Exogenous insulin-like growth factor 1 attenuates sevoflurane anesthesia-induced cognitive dysfunction in aged rats. J Neurophysiol 125:2117–2124. https://doi.org/10.1152/jn.00124.2021
Xu R, Zhu Y, Jia J, Li WX, Lu Y (2021) RIPK1/RIPK3-mediated necroptosis is involved in sevoflurane-induced neonatal neurotoxicity in the rat hippocampus. Cell Mol Neurobiol. https://doi.org/10.1007/s10571-021-01098-z
Yang Y et al (2019) Amygdalin reduces lipopolysaccharide-induced chronic liver injury in rats by down-regulating PI3K/AKT, JAK2/STAT3 and NF-kappaB signalling pathways. Artif Cells Nanomed Biotechnol 47:2688–2697. https://doi.org/10.1080/21691401.2019.1634084
Yoon J et al (2021) Arrhythmia incidence and associated factors during volatile induction of general anesthesia with sevoflurane: a retrospective analysis of 950 adult patients. Anaesth Crit Care Pain Med 40:100878. https://doi.org/10.1016/j.accpm.2021.100878
Zhang A, Pan W, Lv J, Wu H (2017) Protective effect of amygdalin on LPS-induced acute lung injury by inhibiting NF-kappaB and NLRP3 signaling pathways. Inflammation 40:745–751. https://doi.org/10.1007/s10753-017-0518-4
Zhang L et al (2021) High expression of USP18 is associated with the growth of colorectal carcinoma. Histol Histopathol 36:697–704. https://doi.org/10.14670/HH-18-346
Zhou H, Li S, Wang G (2019) Euxanthone ameliorates sevoflurane-induced neurotoxicity in neonatal mice. J Mol Neurosc 68:275–286. https://doi.org/10.1007/s12031-019-01303-1
Zhu H et al (2014) Anti-inflammatory effects of the bioactive compound ferulic acid contained in Oldenlandia diffusa on collagen-induced arthritis in rats. Evid-Based Complement Altern Med 2014:573801. https://doi.org/10.1155/2014/573801
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
FW and YY designed the study and supervised the data collection; YW analyzed the data and interpreted the data; YL prepared the manuscript for publication and reviewed the draft of the manuscript. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author Fengjuan Wang declares that he/she has no conflict of interest. The author Yu Yao declares that he/she has no conflict of interest. The author Yinghui Wu declares that he/she has no conflict of interest. The author Yan Lu declares that he/she has no conflict of interest.
Ethical approval
Ethical approval was obtained from the Ethics Committee of Tongji Medical College, Huazhong University of Science and Technology.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, F., Yao, Y., Wu, Y. et al. USP18 alleviates neurotoxicity induced by sevoflurane via AKT and NF-κB pathways. Mol. Cell. Toxicol. 18, 431–438 (2022). https://doi.org/10.1007/s13273-021-00217-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-021-00217-7