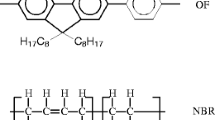
Abstract
The luminescent properties of conjugated copolymers could be harmed due to thermo-oxidative degradation, limiting their applications. To overcome these problems, incorporating flexible and stable polymers, such as elastomers, is a simple and advantageous approach to obtaining luminescent, flexible, and thermo-resistant films. Thin films based on blends of a luminescent thiophene/fluorene copolymer (PTPF) and two elastomers, nitrile rubber (NBR) and natural rubber (NR) were prepared by a straightforward method, and thermal degradation tests were carried out at different temperatures. Oxidized structures in the PTPF chains can be observed using FTIR, and up to 330 °C for NR and to 395 °C for NBR, no significant changes were observable, however, over these temperatures, the blends also lost their luminescent properties. The characterization of the films at increasing degradation stages points to possible mechanisms associated with the degradation processes suggesting a strategy to guarantee incremented thermal protection to the conjugated material, maintaining its structural and optical properties at higher temperatures than ambient temperature, making these flexible-luminescent films interesting for applications in which the thermal resistance is a key factor to consider.
Graphical abstract

Thermal degradation study and obtention of thermo-resistant luminescent films based on blends of thiophene/fluorene copolymer and nitrile or natural rubber





Similar content being viewed by others
References
J. Tian, L. Fu, Z. Liu, H. Geng, Y. Sun, G. Lin, X. Zhang, G. Zhang, D. Zhang, Adv. Funct. Mater. (2019). https://doi.org/10.1002/adfm.201807176
Z.C. Smith, D.M. Meyer, M.G. Simon, C. Staii, D. Shukla, S.W. Thomas, Macromolecules (2015). https://doi.org/10.1021/ma502289n
M. Mamada, R. Komatsu, C. Adachi, ACS Appl. Mater. Interfaces (2020). https://doi.org/10.1021/acsami.0c05449
I. Hamilton, M. Suh, K. Kim, D.Y. Jeon, D.D.C. Bradley, J.S. Kim, Org. Electron. (2020). https://doi.org/10.1016/j.orgel.2019.105496
G. Huseynova, Y.H. Kim, J.H. Lee, J. Lee, J. Inform. Display. (2020). https://doi.org/10.1080/15980316.2019.1707311
N. Pilicode, N.K.M.M. Acharya, P. Naik, S.M.N.A.V. Adhikari, J Photochem Photobiol A Chem. (2019). https://doi.org/10.1016/j.jphotochem.2019.04.012
I.F. Perepichka, D.F. Perepichka, H. Meng, F. Wudl, Adv. Mater. (2005). https://doi.org/10.1002/adma.200500461
R. Ramani, J. Srivastava, S. Alam, Thermochim. Acta (2010). https://doi.org/10.1016/j.tca.2009.10.019
J.K. Jung, Y. Il Moon, K.S. Chung, K.T. Kim, L. McKeen, P. Sarkar, A.K. Bhowmick, S.C. Ng, J.M. Xu, H.S.O. Chan, Synth Methods (2000). https://doi.org/10.1016/S0379-6779(99)00186-1
G.A. Parolin, A.S. Menandro, C.G. Barbosa, L.O. Péres, Synth. Methods (2019). https://doi.org/10.1016/j.synthmet.2019.05.005
Y. Aoyama, T. Yamanari, N. Koumura, H. Tachikawa, M. Nagai, Y. Yoshida, Polym. Degrad. Stab. (2013). https://doi.org/10.1016/j.polymdegradstab.2013.01.006
M. Manceau, A. Rivaton, J.L. Gardette, S. Guillerez, N. Lemaître, Polym. Degrad. Stab. (2009). https://doi.org/10.1016/j.polymdegradstab.2009.03.005
L. Liu, S. Qiu, B. Wang, W. Zhang, P. Lu, Z. Xie, M. Hanif, Y. Ma, J. Shen, J. Phys. Chem. B (2005). https://doi.org/10.1021/jp0547818
X. Guo, M. Chen, Z. Zhang, J. Xu, H. Li, ACS Appl. Polym. Mater. (2021). https://doi.org/10.1021/acsapm.1c00317
V. Pakhnyuk, J.W. Onorato, E.J. Steiner, T.A. Cohen, C.K. Luscombe, Polym. Int. 69, 308–316 (2020)
D. Liu, Z. Ding, Y. Wu, S.F. Liu, Y. Han, K. Zhao, Macromolecules (2022). https://doi.org/10.1021/acs.macromol.1c01537
D. Choi, H. Kim, N. Persson, P. Chu, M. Chang, J. Kang, S. Graham, E. Reichmanis, Chem. Mater. (2016). https://doi.org/10.1021/acs.chemmater.5b04804
Y.-T. Hsieh, J.-Y. Chen, S. Fukuta, P.-C. Lin, T. Higashihara, C.-C. Chueh, W.-C. Chen, ACS Appl. Mater. Interfaces (2018). https://doi.org/10.1021/acsami.8b04582
L. McKeen, Elastomers, in The Effect of Sterilization Methods on Plastics and Elastomers, (4th edition, 2018), p. 305–351 https://doi.org/10.1016/B978-1-4557-2598-4.00013-7.
V. Pakhnyuk, J.W. Onorato, E.J. Steiner, T.A. Cohen, C.K. Luscombe, Polym. Int. (2020). https://doi.org/10.1002/pi.5954
P. Sarkar, A.K. Bhowmick, J. Appl. Polym. Sci. (2018). https://doi.org/10.1002/app.45701
N.O. Braga, D.G.S.M. Cavalcante, A.S. Gomes, E. Yoshihara, R.F. Bianchi, A.E. Job, J. Lumin. (2019). https://doi.org/10.1016/j.jlumin.2019.02.037
J. Hacaloǧlu, S. Yiǧit, U. Akbulut, L. Toppare, Polymer (Guildf). (1997). https://doi.org/10.1016/S0032-3861(97)00056-6
O. Chaikumpollert, K. Sae-heng, O. Wakisaka, A. Mase, Polym. Degrad. Stab. (2011). https://doi.org/10.1016/j.polymdegradstab.2011.08.010
J.K. Jung, Y. Il Moon, K.S. Chung, K.T. Kim, Macromol. Res. (2020). https://doi.org/10.1007/s13233-020-8080-6
J.R. Dunn, Nitrile Rubbers in: Encyclopedia of Materials: Science and Technology (Second edition, 2001), p. 6163.
C.G. Barbosa, R. Faez, L.O. Péres, J. Fluoresc. (2016). https://doi.org/10.1007/s10895-016-1858-4
G.E.J. Hicks, S. Li, N.K. Obhi, C.N. Jarrett-Wilkins, D.S. Seferos, Adv. Mater. (2021). https://doi.org/10.1002/adma.202006287
S. Inagaki, Y.C. Lin, H. Tanaka, T. Koganezawa, W.C. Chen, T. Higashihara, Mater. Chem. Phys. (2022). https://doi.org/10.1016/j.matchemphys.2022.125911
J. Mei, Z. Bao, Chem. Mater. (2014). https://doi.org/10.1021/cm4020805
Y.C. Lin, M. Matsuda, C.K. Chen, W.C. Yang, C.C. Chueh, T. Higashihara, W.C. Chen, Macromolecules (2021). https://doi.org/10.1021/acs.macromol.1c00534
H.C. Tien, Y.W. Huang, Y.C. Chiu, Y.H. Cheng, C.C. Chueh, W.Y. Lee, J. Mater. Chem. C Mater. (2021). https://doi.org/10.1039/d0tc06059c
A.S. Menandro, G.A. Parolin, C.G. Barbosa, R. Faez, L.O. Péres, Macromol. Symp. (2019). https://doi.org/10.1002/masy.201800023
N. Miyaura, A. Suzuki, Chem. Rev. (1995). https://doi.org/10.1021/cr00039a007
S.L. McFarlane, D.G. Piercey, L.S. Coumont, R.T. Tucker, M.D. Fleischauer, M.J. Brett, J.G.C. Veinot, Macromolecules (2009). https://doi.org/10.1021/ma8022348
C.Y. Yu, W.L. Lin, Eur. Polym. J. (2014). https://doi.org/10.1016/j.eurpolymj.2014.02.002
C.Z. He, Z. Peng, J.P. Zhong, S.Q. Liao, X.D. She, Y.Y. Luo, H.S. Tan, Adv Mat Res. (2011). https://doi.org/10.4028/www.scientific.net/AMR.306-307.50
X. Cao, C. Xu, Y. Wang, Y. Liu, Y. Liu, Y. Chen, Polym. Test. (2013). https://doi.org/10.1016/j.polymertesting.2013.04.005
R. Matroniani, S.H. Wang, Polimeros. (2017). https://doi.org/10.1590/0104-1428.2423
H. Gaspar, L. Fernandes, L. Brandão, G. Bernardo, Polym. Test. (2014). https://doi.org/10.1016/j.polymertesting.2014.01.013
S. Gunasekaran, R.K. Natarajan, A. Kala, Spectrochimica Acta Part A. (2007). https://doi.org/10.1016/j.saa.2006.11.039
R.M. Silverstein, F.X. Webster, D.J. Kiemle, Spectrometric Identification of Organic Compounds, (7th ed., 2005).
M. Celina, J. Wise, D.K.O.K.T. Gillen, R.L. Clough, Polym. Degrad. Stabil. 60, 493–504 (1998). https://doi.org/10.1016/S0141-3910(97)00113-4
J. Zhao, R. Yang, R. Iervolino, B. Van Der Vorst, S. Barbera, Polym. Degrad. Stabil. J. 115, 32–37 (2015)
R. Grisorio, G.P. Suranna, P. Mastrorilli, C.F. Nobile, Adv. Funct. Mater. (2007). https://doi.org/10.1002/adfm.200600083
J. Jo, D. Vak, Y.Y. Noh, S.S. Kim, B. Lim, D.Y. Kim, J. Mater. Chem. (2008). https://doi.org/10.1039/b714138f
J. Morgado, A.T. Pereira, A.M. Bragança, Q. Ferreira, S.C.M. Fernandes, C.S.R. Freire, L. Alcácer, A.J.D. Silvestre, C.P. Neto, Express Polym Lett (2013). https://doi.org/10.3144/expresspolymlett.2013.94
R. Sreeja, S. Najidha, S. Jayan, P. Predeep, M. Mazur, P.D. Sharma, Polymer (Guildf) (2006). https://doi.org/10.1016/j.polymer.2005.09.024
Y. Kim, J. Bouffard, S.E. Kooi, T.M. Swager, J. Am. Chem. Soc. 127, 13726–13731 (2005)
S. Tirapattur, M. Bellet, N. Drolet, M. Leclerc, G. Durocher, Chem. Phys. Lett. (2003). https://doi.org/10.1016/S0009-2614(03)00178-7
J.R. Lakowicz, Principles of Fluorescence Spectroscopy, 3° edition (Springer Science+Business Media LLC, Baltimore, 2006). https://doi.org/10.1007/978-0-387-46312-4
L. Liu, S. Tang, M. Liu, Z. Xie, W. Zhang, P. Lu, M. Hanif, Y. Ma, J. Phys. Chem. B (2006). https://doi.org/10.1021/jp062612x
Acknowledgements
We are thankful to FAPESP (2018/04340-4, 2016/13943-9, 2015/14681-5 and 2014/50869-6), CNPq (401109/2014-3, 308686/2017-9) and CAPES (23038.000776/201754) via the projects of the National Institute for Science and Technology on Organic Electronics (INEO) for the financial support. Rebeca R. Rodrigues receives a CAPES fellowship (Finance code 001).
Author information
Authors and Affiliations
Contributions
Agnaldo G. N. de Souza: Investigation. Yasmin B. da Silva: Investigation. Rebeca R. Rodrigues: Writing—Original Draft. Alessandra S. Menandro: Methodology, Formal analysis, Writing—Original Draft. Laura O. Péres: Supervision, Funding acquisition, Writing—Review & Editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Souza, A.G.N., da Silva, Y.B., Rodrigues, R.R. et al. Heat-resistant luminescent films: a thermal study of fluorene/thiophene copolymer-elastomer blends. Macromol. Res. (2024). https://doi.org/10.1007/s13233-024-00261-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13233-024-00261-x