Abstract
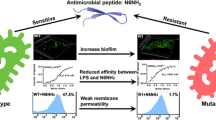
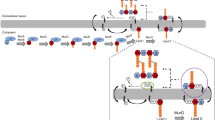
Due to innate and acquired resistance in Enterococcus faecalis against most antibiotics, identification of new alternatives has increased interest in diverse populations of potent cationic antimicrobial peptides (CAMPs) for treatment and natural food biopreservation. The CAMPs, after crossing the cell wall to the periplasmic space, kill their target strain by forming pores in the cell membrane. However, reports of resistance against these CAMPs necessitated the understanding of step(s) interfered with while acquiring this resistance, for designing effective CAMP analogs. In this direction, we selected stable and gradual dose-dependent pediocin PA-1 single exposure resistant (Pedr) mutants of E. faecalis, which conferred cross-protection to diverse CAMPs, viz., HNP-1, nisin and alamethicin but not to polymyxin B, lysozyme and vancomycin. With these Pedr mutants of E. faecalis there was: a gradual neutralization in cell wall surface charge involving D-alanylation of wall teichoic acids (WTA) and lipoteichoic acids (LTA), increase in cell-surface hydrophobicity, increased cell aggregation and biofilm formation and ultra-structural changes in the cell wall, and a reduction of periplasmic space. In addition, a gradual decrease in expression of mannose PTS two (mpt) operon was also observed with distinct changes in growth rate achieving the same biomass production during the stationary phase. These results show that resistance to these CAMPs is not due to mpt directly acting as a docking molecule but due to changes in the cell wall, which increased the permeability barrier to CAMPs diffusion to reach the periplasmic space.








Similar content being viewed by others
References
Arias CA, Murray BE (2009) Antibiotic-resistant bugs in the 21st century - a clinical super-challenge. N Engl J Med 360:439–443
Arias CA, Panesso D, McGrath DM, Qin X, Mojica MF, Miller C, Diaz L, Tran TT, Rincon S, Barbu EM, Reyes J, Roh JH, Lobos E, Sodergren E, Pasqualini R, Arap W, Quinn JP, Shamoo Y, Murray BE, Weinstock GM (2011) Genetic basis for in vivo daptomycin resistance in enterococci. New Engl J Med 365:892–900
Arous S, Dalet K, Hechard Y (2004) Involvement of the mpo operon in resistance to class IIa bacteriocins in Listeria monocytogenes. FEMS Microbiol Lett 238(1):37–41
Baldassarri L, Cecchini R, Bertuccini L, Ammendolia MG, Iosi F, Arciola CR, Montanaro L, Di Rosa R, Gherardi G, Dicuonzo G, Orefici G, Creti R (2001) Enterococcus spp. produces slime and survives in rat peritoneal macrophages. Med Microbiol Immunol (Berlin) 190:113–120
Butler MT, Qingfeng W, Harshey RM (2010) Cell density and mobility protect swarming bacteria against antibiotics. Proc Natl Acad Sci U S A 107:3776–3781
Cabo ML, Murado MA, Gonzalez MP, Pastoriza L (1999) A method for bacteriocin quantification. J Appl Microbiol 87:907–914
Calvez S, Rince A, Auffray Y, Prevost H, Drider D (2007) Identification of new genes associated with intermediate resistance of Enterococcus faecalis to divercin V41, a pediocin-like bacteriocin. Microbiology 153:1609–1618
Cotter PD, Hill C, Ross P (2005) Bacteriocins: developing innate immunity for food. Nat Rev Microbiol 3(10):777–788
Dalet K, Cenatiempo Y, Cossart P, Hechard Y (2001) A σ54-dependent PTS permease of the mannose family is responsible for sensitivity of Listeria monocytogenes to mesentericin Y105. Microbiology 147:3263–3269
Dover RS, Bitler A, Nezer R, Shmuel-Galia L, Firon A, Shimoni E, Trieu-Cuot P, Shai Y (2012) D-Alanylation of lipoteichoic acids confers resistance to cationic peptides in Group B Streptococcus by increasing the cell wall density. PLoS Pathog 8(9):e1002891
Dykes GA, Hastings JW (1998) Fitness costs associated with class IIa bacteriocin resistance in Listeria monocytogenes B73. Lett Appl Microbiol 26(1):5–8
Fabretti F, Theilacker C, Baldassari L, Kaczynski Z, Kropec A, Holst O, Huebner J (2006) Alanine esters of enterococcal lipoteichoic acid play a role in biofilm formation and resistance to antimicrobial peptides. Infect Immun 74(7):4164–4171
Fimland G, Johnsen L, Axelsson L, Brurberg MB, Nes IF, Eijsink VGH, Nissen-Meyer A (2000) A C-terminal disulfide bridge in pediocin-like bacteriocins renders bacteriocin activity less temperature dependent and is a major determinant of the antimicrobial spectrum. J Bacteriol 198(9):2643–2648
Gomez JAL, Hendrickx APA, Willems RJ, Top J, Sava I, Huebner J, Witte W, Werner G (2011) Intra- and interspecies genomic transfer of the Enterococcus faecalis pathogenicity island. PLoS One 6(4):e16720
Gravesen A, Jydegaard Axelsen AM, Mendes da Silva J, Hansen TB, Knochel S (2002a) Frequency of bacteriocin resistance development and associated fitness costs in Listeria monocytogenes. Appl Environ Microbiol 68:756–764
Gravesen A, Ramnath M, Rechinger KB, Andersen N, Jansch L, Hechard Y, Hastings JW, Knochel S (2002b) High-level resistance to class IIa bacteriocins is associated with one general mechanism in Listeria monocytogenes. Microbiology 148:2361–2369
Hancock RE, Chapple DS (1999) Peptide antibiotics. Antimicrob Agents Chemother 43:1317–1323
Hayat MA (2000) Principles and techniques of electron microscopy: biological applications, 4th edn. Cambridge University Press, Cambridge, United Kingdom
Hechard Y, Pelletier C, Cenatiempo Y, Frere J (2001) Analysis of σ54-dependent genes in Enterococcus faecalis: a mannose PTS permease (EIIMan) is involved in sensitivity to a bacteriocin, mesentericin Y105. Microbiology 147:1575–1580
Jacquet T, Cailliez-Grimal C, Borges F, Gaiani C, Francius G, Duval JFL, Waldvogel Y, Revol-Junelles (2012) Surface properties of bacteria sensitive and resistant to the class IIa carnobacteriocin Cbn BM1. J Appl Microbiol 112(2):372–382
Johnsen L, Fimland G, Nissen-Meyer J (2005) The C-terminal domain of pediocin-like antimicrobial peptides (class IIa bacteriocins) is involved in specific recognition of the C-terminal part of cognate immunity proteins and in determining the antimicrobial spectrum. J Biol Chem 280(10):9243–9250
Kahovcova J, Odavic R (1969) A simple method for the quantitative analysis of phospholipids separated by thin layer chromatography. J Chromatogr A 40:90–96
Koch S, Hufnagel M, Theilacker C, Huebner J (2004) Enterococcal infections: host response, therapeutic, and prophylactic possibilities. Vaccine 22:822–830
McBride SM, Sonenshein AL (2011) The dlt operon confers resistance to cationic antimicrobial peptides in Clostridium difficile. Microbiology 157:1457–1465
McBride SM, Fischetti VA, LeBlanc DJ, Moellering RC, Gilmore MS (2007) Genetic diversity among Enterococcus faecalis. PLoS One 7:e582
Mehla J, Sood SK (2011) Substantiation of Enterococcus faecalis of dose-dependent resistance and cross-resistance to pore-forming antimicrobial peptides by use of a polydiacetylene-based colorimetric assay. Appl Environ Microbiol 77(3):786–793
Mehla J, Sood SK (2013) Connecting membrane fluidity and surface charge to pore-forming antimicrobial peptides resistance by an ANN-based predictive model. Appl Microbiol Biotechnol 97(10):4377–4384
Mishra NN, Bayer AS, Tran TT, Shamoo Y, Mileykovskaya E, Dowhan W, Guan Z, Arias CA (2012) Daptomycin resistance in enterococci is associated with distinct alterations of cell membrane phospholipid content. PLoS One 7(8):e49358
Moll GN, Clark J, Chan WC, Bycroft BW, Roberts GC, Konings WN, Driessen AJ (1997) Role of transmembrane pH gradient and membrane binding in nisin pore formation. J Bacteriol 179(1):135–140
Naghmouchi K, Kheadr E, Lacroix C, Fliss I (2007) Class I/class IIa bacteriocin cross-resistance phenomenon in Listeria monocytogenes. Food Microbiol 24:718–727
Nes IF, Diep DB, Holo H (2007) Bacteriocin diversity in Streptococcus and Enterococcus. J Bacteriol 189(4):1189–1198
Opsata M, Nes IF, Holo H (2010) Class IIa bacteriocin resistance in Enterococcus faecalis V583: The mannose PTS operon mediates global transcriptional responses. BMC Microbiol 10:224
Park SC, Kim JY, Shin SO, Jeong CY, Kim MH, Shin SY, Cheong GW, Park Y, Hahm KS (2006) Investigation of toroidal pore and oligomerization by melittin using transmission electron microscopy. Biochem Biophys Res Commun 343:222–228
Paulsen IT, Banerjei L, Myers GS, Nelson KE, Seshadri R, Read TD, Fouts DE, Eisen JA, Gill SR, Heidelberg JF, Tettelin H, Dodson RJ, Umayam L, Brinkac L, Beanan M, Daugherty S, DeBoy RT, Durkin S, Kolonay J, Madupu R, Nelson W, Vamathevan J, Tran B, Upton J, Hansen T, Shetty J, Khouri H, Utterback T, Radune D, Ketchum KA, Dougherty BA, Fraser CM (2003) Role of mobile DNA in the evolution of vancomycin-resistant Enterococcus faecalis. Science 299:2071–2074
Peschel A, Sahl HG (2006) The co-evolution of host cationic antimicrobial peptides and microbial resistance. Nat Rev Microbiol 4:529–536
Peschel A, Otto M, Jack RW, Kalbacher H, Jung G, Gotz F (1999) Inactivation of the dlt operon in Staphylococcus aureus confers sensitivity to defensins, protegrins and other antimicrobial peptides. J Biol Chem 274:8405–8410
Pollack JH, Neuhaus FC (1994) Changes in wall teichoic acid during the rod-sphere transition of Bacillus subtilis. J Bacteriol 176:7252–7259
Ramnath M, Arous S, Gravesen A, Hastings JW, Hechard Y (2004) Expression of mptC of Listeria monocytogenes induces sensitivity to class IIa bacteriocins in Lactococcus lactis. Microbiology 150(8):2663–2668
Reifsteck F, Wee S, Wilkinson BJ (1987) Hydrophobicity-hydrophilicity of staphylococci. J Med Microbiol 24:65–73
Rekhif N, Atrih A, Lefebvre G (1994) Selection and properties of spontaneous mutants of Listeria monocytogenes ATCC 15313 resistant to different bacteriocins produced by lactic acid bacteria strains. Curr Microbiol 28:237–241
Sakayori Y, Muramatsu M, Hanada S, Kamagata Y, Kawamoto S, Shima J (2003) Characterization of Enterococcus faecium mutants resistant to mundticin KS, a class IIa bacteriocin. Microbiology 149:2901–2908
Sansom MS (1991) The biophysics of peptide models of ion channels. Prog Biophys Mol Biol 55:139–235
Shepard BD, Gilmore MS (1999) Identification of aerobically and anaerobically induced genes in Enterococcus faecalis by random arbitrarily primed PCR. Appl Environ Microbiol 65(4):1470–1476
Sood SK, Sinha PR (2003) Analysis of structure of YGNGV motif containing bacteriocins: A model for pore formation. Ind J Biotechnol 2:227–235
Sood SK, Vijay Simha B, Kumariya R, Garsa AK, Mehla J, Meena S, Lather P (2013) Highly specific culture-independent detection of YGNGV motif-containing pediocin-producing strains. Probiotics Antimicrob Prot 5:37–42
Tessema GT, Moretro T, Snipen L, Axelsson L, Naterstad K (2011) Global transcriptional analysis of spontaneous sakacin P-resistant mutant strains of Listeria monocytogenes during growth on different sugars. PLoS One 6(1):e16192
Thippeswamy HS, Sood SK, Venkateswarlu R, Raj I (2009) Membranes of five-fold alamethicin-resistant Staphylococcus aureus, Enterococcus faecalis and Bacillus cereus show decreased interactions with alamethicin due to changes in membrane fluidity and surface charge. Ann Microbiol 59:593–601
Vadyvaloo V, Arous S, Gravesen A, Hechard Y, Chauhan-Haubrock R, Hastings JW, Rautenbach M (2004a) Cell-surface alterations in class IIa bacteriocin-resistant Listeria monocytogenes strains. Microbiology 150(9):3025–3033
Vadyvaloo V, Snoep JL, Hastings JW, Rautenbach M (2004b) Physiological implications of class IIa bacteriocins resistance in Listeria monocytogenes. Microbiology 150(2):335–340
Vijay Simha B, Sood SK, Kumariya R, Garsa AK (2012) Simple and rapid purification of pediocin PA-1 from Pediococcus pentosaceus NCDC 273 suitable for industrial application. Microbiol Res 167:544–549
Yang L, Harroun TA, Weiss TM, Ding L, Huang HW (2001) Barrel-stave model or toroidal model? A case study on melittin pores. Biophys J 81:1475–1485
Zasloff M (2002) Antimicrobial peptides of multicellular organisms. Nature 415(6870):389–395
Acknowledgments
The authors thank the Director, National Dairy Research Institute, Karnal, India for providing general support during this period of research study. The study was supported by the National Initiative on Climate Resilient Agriculture (NICRA) Project, ICAR. We thank Dr. S. K. Tomar, Principal Scientist, Dairy Microbiology Division, NDRI, Karnal, India for helping us in carrying out scanning electron microscopy, and we are also thankful to All India Institute of Medical Sciences, India for allowing us to use the central facility for transmission electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumariya, R., Sood, S.K., Rajput, Y.S. et al. Gradual pediocin PA-1 resistance in Enterococcus faecalis confers cross-protection to diverse pore-forming cationic antimicrobial peptides displaying changes in cell wall and mannose PTS expression. Ann Microbiol 65, 721–732 (2015). https://doi.org/10.1007/s13213-014-0912-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-014-0912-1