Abstract
Co-occurrence of two devastating foliar-fungal diseases of peanut, viz., late leaf spot (LLS), and rust may cause heavy yield loss besides adversely affecting the quality of kernel and fodder. This study reports the mapping of seven novel stress-related candidate EST-SSRs in a region having major QTLs for LLS and rust diseases using an F2 mapping population (GJG17 × GPBD4) consisting of 328 individuals. The parental polymorphism using 1311 SSRs revealed 84 SSRs (6.4%) as polymorphic and of these 70 SSRs could be mapped on 14 linkage groups (LG). QTL analysis has identified a common QTL (LLSQTL1/RustQTL) for LLS and rust diseases in the map interval of 1.41 cM on A03 chromosome, explaining 47.45% and 70.52% phenotypic variations, respectively. Another major QTL for LLS (LLSQTL1), explaining a 29.06% phenotypic variation was also found on LG_A03. A major rust QTL has been validated which was found harboring R-gene and resistance-related genes having a role in inducing hypersensitive response (HR). Further, 23 linked SSRs including seven novel EST-SSRs were also validated in 177 diverse Indian groundnut genotypes. Twelve genotypes resistant to both LLS and rust were found carrying the common (rust and LLS) QTL region, LLS QTL region, and surrounding regions. These identified and validated candidate EST-SSR markers would be of great use for the peanut breeding groups working for the improvement of foliar-fungal disease resistance.




Similar content being viewed by others
Change history
10 November 2020
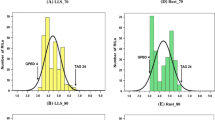
Inadvertently, Fig.��4 got published incorrectly as Fig.��2 in the original publication. The correct Fig.��2 and Fig.��4 are given in the following page.
References
Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y (2004) Role of resveratrol in prevention and therapy of cancer: preclinical and clinical studies. Anticancer Res 24:2783–2840 (PMID:15517885)
Alahmad S, Dinglasan E, Leung KM, Riaz A, Derbal N, Voss-Fels KP et al (2018) Speed breeding for multiple quantitative traits in durum wheat. Plant Methods 14:36. https://doi.org/10.1186/s13007-018-0302-y
Bala M, Radhakrishnan T, Kumar A, Mishra GP, Dobaria JR, Kirti PB (2016) Over-expression of a fusion gene of radish and fenugreek defensins improves the resistance to leaf spot diseases caused by Cercospora arachidicola and Phaeoisariopsis personata in transgenic peanut. Turkish J Biol 40:139–149. https://doi.org/10.3906/biy-1412-46
Bayer M, Milne I, Stephen G, Shaw P, Cardle L, Wright F et al (2011) Comparative visualization of genetic and physical maps with Strudel. Bioinformatics 27:1307–1308. https://doi.org/10.1093/bioinformatics/btr111
Bernoux M, Moncuquet P, Kroj T, Dodds PN (2014) A novel conserved mechanism for plant NLR protein pairs: the “integrated decoy” hypothesis. Front Plant Sci 5:606. https://doi.org/10.3389/fpls.2014.00606
Bhalani H, Thankappan R, Mishra GP, Sarkar T, Bosamia TC, Dobaria JR (2019) Regulation of antioxidant mechanisms by AtDREB1A improves soil-moisture deficit stress tolerance in transgenic peanut (Arachis hypogaea L). PLoS ONE 14(5):e0216706. https://doi.org/10.1371/journal.pone.0216706
Bosamia TC, Mishra GP, Thankappan R, Dobaria JR (2015) Novel and stress relevant EST-derived SSR markers developed and validated in peanut. PLoS ONE 10(7):e0133537. https://doi.org/10.1371/journal.pone.0129127
Bosamia TC, Dodia SM, Mishra GP, Ahmad S, Joshi B, Thirumalaisamy PP et al (2020) Unraveling the mechanisms of resistance to Sclerotium rolfsii in peanut (Arachis hypogaea L.) using comparative RNA-Seq analysis of resistant and susceptible genotypes. PLoS ONE 15(8):e0236823. https://doi.org/10.1371/journal.pone.0236823
Clevenger J, Chu Y, Chavarro C, Botton S, Culbreath A, Isleib TG et al (2018) Mapping late leaf spot resistance in peanut (Arachis hypogaea) using QTL-seq reveals markers for marker-assisted selection. Front Plant Sci 9:83. https://doi.org/10.3389/fpls.2018.00083
Dodia SM, Joshi B, Gangurde SG, Thirumalaisamy PP, Mishra GP, Kumar N et al (2019) Genotyping-by-sequencing based genetic mapping reveals a large number of epistatic interactions for stem rot resistance in groundnut. Theor Appl Genet 132:1001–1016. https://doi.org/10.1007/s00122-018-3255-7
Doerge RW (2002) Mapping and analysis of quantitative trait loci in experimental populations. Nat Rev Genet 3:43–52. https://doi.org/10.1038/nrg703
Dwivedi SL, Pande S, Rao JN, Nigam SN (2002) Components of resistance to late leaf spot and rust among interspecific derivatives and their significance in a foliar disease resistance breeding in groundnut (Arachis hypogaea L.). Euphytica 125:81–88. https://doi.org/10.1023/A:1015707301659
Eitas TK, Dangl JL (2010) NB-LRR proteins: pairs, pieces, perception, partners, and pathways. Curr Opin Plant Biol 13:472–477. https://doi.org/10.1016/j.pbi.2010.04.007
FAOSTAT (2018) Food and agricultural organization of the United Nations. https://www.fao.org/faostat/en/#data/QC. Accessed 7 Aug 2020
Gajjar K, Mishra GP, Radhakrishnan T, Dodia S, Rathnakumar A, Kumar N et al (2014) Validation of SSR markers linked to the rust and late leaf spot diseases resistance in diverse peanut genotypes. Aust J Crop Sci 86:927–936. https://www.cropj.com/thankapan_8_6_2014_927_936.pdf. Accessed 1 Mar 2020
Gautami B, Foncéka D, Pandey MK, Moretzsohn MC, Sujay V, Qin H et al (2012) An international reference consensus genetic map with 897 marker loci based on 11 mapping populations for tetraploid groundnut (Arachis hypogaea L). PLoS ONE 7:e41213. https://doi.org/10.1371/journal.pone.0041213
Gowda MV, Motagi BN, Naidu GK, Diddimani SB, Sheshagiri R (2002) GPBD 4: a Spanish bunch groundnut genotype resistant to rust and late leaf spot. Intl Arachis Newslett 22:29–32
He G, Meng R, Newman M, Gao G, Pittman RN, Prakash CS (2003) Microsatellites as DNA markers in cultivated peanut (Arachis hypogaea L). BMC Plant Biol 3:3. https://doi.org/10.1186/1471-2229-3-3
Hong YB, Liang XQ, Chen XP, Liu HY, Zhou GY, Li SX et al (2008) Construction of genetic linkage map based on SSR markers in peanut (Arachis hypogaea L.). Agric Sci China 7:915–921. https://doi.org/10.1016/S1671-2927(08)60130-3
Hong Y, Chen X, Liang X, Liu H, Zhou G, Li S et al (2010) A SSR based composite genetic linkage map for the cultivated peanut Arachis hypogaea L. genome. BMC Plant Biol 10:17–30. https://doi.org/10.1186/1471-2229-10-17
Keppler LD, Novacky A (1986) Involvement of membrane lipid peroxidation in the development of a bacterially induced hypersensitive reaction. Phytopathol 76:104–108
Khedikar YP, Gowda MV, Sarvamangala C, Patgar KV, Upadhyaya HD, Varshney RK (2010) A QTL study on late leaf spot and rust revealed one major QTL for molecular breeding for rust resistance in groundnut (Arachis hypogaea L.). Theor Appl Genet 121:971–984. https://doi.org/10.1007/s00122-010-1366-x
Kim SH, Hong JK, Lee SC, Sohn KH, Jung HW, Hyang BK (2004) CAZFP1, Cys2/His2-type zinc-finger transcription factor gene functions as a pathogen-induced early-defense gene in Capsicum annuum. Plant Mol Biol 55:883–904. https://doi.org/10.1007/s11103-004-2151-5
Ko JH, Sethi G, Um JY, Shanmugam MK, Arfuso F, Kumar AP et al (2017) The role of resveratrol in cancer therapy. Int J Mol Sci 18(12):2589. https://doi.org/10.3390/ijms18122589
Kolekar RM, Sujay V, Shirasawa K, Sukruth M, Khedikar YP, Gowda MV et al (2016) QTL mapping for late leaf spot and rust resistance using an improved genetic map and extensive phenotypic data on a recombinant inbred line population in peanut (Arachis hypogaea L.). Euphytica 209:147–156. https://doi.org/10.1007/s10681-016-1651-0
Kosambi DD (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175. https://doi.org/10.1111/j.1469-1809.1943.tb02321.x
Kwon SJ, Jin HC, Lee S, Nam MH, Chung JH, Kwon SI et al (2009) GDSL lipase-like 1 regulates systemic resistance associated with ethylene signaling in Arabidopsis. Plant J 58:235–245. https://doi.org/10.1111/j.1365-313X.2008.03772.x
Lee DS, Kim BK, Kwon SJ, Jin HC, Park OK (2009) Arabidopsis GDSL lipase 2 plays a role in pathogen defense via negative regulation of auxin signaling. Biochem Biophys Res Commun 379:1038–1042. https://doi.org/10.1016/j.bbrc.2009.01.006
Li H, Ye G, Wang J (2007) A modified algorithm for the improvement of composite interval mapping. Genetics 175:361–374. https://doi.org/10.1534/genetics.106.066811
Liao BS (2003) The groundnut, Hubei Press for Science and Technology, Wuhan, China, https://www.peanutscience.com/doi/pdf/10.3146/AT07-004.1. Accessed 1 Mar 2020
Mehan VK, Reddy PM, Subrahmanyam P, McDonald D, Singh AK (1996) Identification of new sources of resistance to rust and late leaf spot in peanut. Int J Pest Manag 42:267–271. https://doi.org/10.1080/09670879609372004
Mishra GP, Radhakrishnan T, Kumar A, Thirumalaisamy PP, Kumar N, Bosamia TC et al (2015) Advancements in molecular marker development and their applications in the management of biotic stresses in peanuts. Crop Prot 77:74–86. https://doi.org/10.1016/j.cropro.2015.07.019
Mondal S, Badigannavar AM (2010) Molecular diversity and association of SSR markers to rust and late leaf spot resistance in cultivated groundnut (Arachis hypogaea L). Plant Breed 129(1):68–71. https://doi.org/10.1111/j.1439-0523.2009.01635.x
Mondal S, Badigannavar AM (2018) Mapping of a dominant rust resistance gene revealed two R genes around the major Rust_QTL in cultivated peanut (Arachis hypogaea L.). Theor Appl Genet 131:1671–1681. https://doi.org/10.1007/s00122-018-3106-6
Mondal S, Badigannavar AM, Murty GSS (2008) RAPD markers linked to a rust resistance gene in cultivated groundnut (Arachis hypogaea L.). Euphytica 159:233–239. https://doi.org/10.1007/s10681-007-9482-7
Mondal S, Badigannavar AM, D’Souza SF (2012a) Development of genic molecular markers linked to a rust resistance gene in cultivated groundnut (Arachis hypogaea L.). Euphytica 188:163–173. https://doi.org/10.1007/s10681-011-0619-3
Mondal S, Badigannavar AM, D’Souza SF (2012b) Molecular tagging of a rust resistance gene in cultivated groundnut Arachis hypogaea L. introgressed from Arachis cardenasii. Mol Breed 29:467–476. https://doi.org/10.1007/s11032-011-9564-z
Monyo ES, Osiru MO, Kadyampakeni D, Mponda O, Chinyamunyamu B (2009) Improving food security and nutrition in Malawi and Tanzania through research on edible legumes. Proceedings of stakeholder workshops on groundnut production in Malawi and Tanzania held 1–2 March and 13 April 2007, Lilongwe Malawi and Mtwara Tanzania. Patancheru 502 324, Andhra Pradesh, India: ICRISAT, 96. (ISBN:978–92–9066–515–1)
Nawade B, Bosamia TC, Thankappan R, Rathnakumar AL, Kumar A, Dobaria JR et al (2016) Insights into the Indian peanut genotypes for ahFAD2 gene polymorphism regulating its oleic and linoleic acid fluxes. Front Plant Sci 7:1271. https://doi.org/10.3389/fpls.2016.01271
Nawade B, Mishra GP, Radhakrishnan T, Dodia SM, Ahmad S, Kumar A et al (2018) High oleic peanut breeding: achievements perspectives and prospects. Trends Food Sci Technol 78:107–119. https://doi.org/10.1016/j.tifs.2018.05.022
Nawade B, Mishra GP, Radhakrishnan T, Sangh C, Dobariya JR, Kundu R (2019) Development of high oleic peanut lines through marker-assisted introgression of mutant ahFAD2 alleles and its fatty acid profiles under open-field and controlled conditions. Biotech 9:243. https://doi.org/10.1007/s13205-019-1774-9
Oh S, Park JM, Joung YH, Lee S, Chung E, Kim S et al (2005) A plant EPF-type zinc-finger protein, CaPIF1, involved in defence against pathogens. Mol Plant Pathol 63:269–285. https://doi.org/10.1111/j.1364-3703.2005.00284.x
Oliveira KM, Pinto LR, Marconi TG, Margarido GR, Pastina MM, Teixeira LH et al (2007) Functional integrated genetic linkage map based on EST-markers for a sugarcane (Saccharum spp.) commercial cross. Mol Breed 20:189–208. https://doi.org/10.1007/s11032-007-9082-1
Pandey MK, Monyo E, Ozias-Akins P, Liang X, Guimarães P, Nigam SN et al (2012) Advances in Arachis genomics for peanut improvement. Biotechnol Adv 30:639–651. https://doi.org/10.1016/j.biotechadv.2011.11.001
Pandey MK, Khan AW, Singh VK, Vishwakarma MK, Shasidhar Y, Kumar V et al (2017) QTL-seq approach identified genomic regions and diagnostic markers for rust and late leaf spot resistance in groundnut (Arachis hypogaea L.). Plant Biotechnol J 15:927–941. https://doi.org/10.1111/pbi.12686
Rathnakumar AL, Singh R, Parmar DL, Misra JB (2013) A crop profile and compendium of varieties notified in India, Directorate of Groundnut research, PB No. 5 Junagadh-362001 Gujarat India, 118. https://www.dgr.org.in/wp-content/uploads/2015/07/Groundnut-a-crop-profile-and-compendium-of-notified-varieties-of-India.pdf. Accessed 1 Mar 2020
Schweiger W, Steiner B, Vautrin S, Nussbaumer T, Siegwart G, Zamini M et al (2016) Suppressed recombination and unique candidate genes in the divergent haplotype encoding Fhb1, a major Fusarium head blight resistance locus in wheat. Theor Appl Genet 129:1607–1623. https://doi.org/10.1007/s00122-016-2727-x
Serba DD, Uppalapati SR, Mukherjee S, Krom N, Tang Y, Mysore KS et al (2015) Transcriptome profiling of rust resistance in switchgrass using RNA-Seq analysis. Plant Genome 82:1–12. https://doi.org/10.3835/plantgenome2014.10.0075
Shirasawa K, Bhat RS, Khedikar YP, Sujay V, Kolekar RM, Yeri SB et al (2018) Sequencing analysis of genetic loci for resistance for late leaf spot and rust in peanut (Arachis hypogaea L). Front Plant Sci 9:1727. https://doi.org/10.3389/fpls.2018.01727
Shoba D, Manivannan N, Vindhiyavarman P, Nigam SN (2012) SSR markers associated for late leaf spot disease resistance by bulked segregant analysis in groundnut (Arachis hypogaea L.). Euphytica 188:265–272. https://doi.org/10.1007/s10681-012-0718-9
Singh AK, Mehan VK, Nigam SN (1997) Sources of resistance to groundnut fungal and bacterial disease: an update and appraisal. Technical Report, Information bulletin, No. 50, International Crops Research Institute for the Semi-Arid Tropics, Patencheru 502324, Andhra Pradesh, India. https://oar.icrisat.org/id/eprint/6676. Accessed 1 Mar 2020
Sourdille P, Cadalen T, Guyomarch H, Snape J, Perretant M, Charmet G et al (2003) An update of the Courtot 9 Chinese spring intervarietal molecular marker linkage map for the QTL detection of agronomic traits in wheat. Theor Appl Genet 106:530–538. https://doi.org/10.1007/s00122-002-1044-8
Subbarao PV, Subrahmanyam P, Reddy PM (1990) A modified nine point disease scale for assessment of rust and late leaf spot of groundnut. Second International Congress of French Phytopathological Society, Montpellier, France
Subrahmanyam P, Williams JH, McDonald D, Gibbons RW (1984) The influence of foliar diseases and their control by selective fungicides on a range of groundnut (Arachis hypogaea L.) genotypes. Ann Appl Bio 1043:467–476. https://doi.org/10.1111/j.1744-7348.1984.tb03029.x
Subrahmanyam P, Rao VR, McDonald D, Moss JP, Gibbons RW (1989) Origins of resistances to rust and late leaf spot in peanut (Arachis hypogaea, Fabaceae). Econ Bot 43:444. https://doi.org/10.1007/BF02935917
Subrahmanyam P, McDonald D, Waliyar F, Reddy LJ, Nigam SN, Gibbons RW (1995) Screening methods and sources of resistance to rust and late leaf spot of groundnut. In: Information bulletin, No 47, ICRISAT, Patancheru, India, https://oar.icrisat.org/id/eprint/3477. Accessed 1 Mar 2020
Sujay V, Gowda MV, Pandey MK, Bhat RS, Khedikar YP, Nadaf HL et al (2012) QTL analysis and construction of consensus genetic map for foliar diseases resistance based on two RIL populations in cultivated groundnut (Arachis hypogaea L.). Mol Breed 32:773–788. https://doi.org/10.1007/s11032-011-9661-z
Sukruth M, Paratwagh SA, Sujay V, Kumari V, Gowda MV, Nadaf HL et al (2015) Validation of markers linked to late leaf spot and rust resistance, and selection of superior genotypes among diverse recombinant inbred lines and backcross lines in peanut (Arachis hypogaea L.). Euphytica 204:343–351. https://doi.org/10.1007/s10681-014-1339-2
Sundaresha S, Kumar AM, Rohini S, Math S, Keshamma E, Chandrashekar S et al (2010) Enhanced protection against two major fungal pathogens of groundnut, Cercospora arachidicola and Aspergillus flavus in transgenic groundnut over expressing a tobacco β 1–3 glucanase. Eur J Plant Pathol 126:497–508. https://doi.org/10.1007/s10658-009-9556-6
Tian ZD, Zhang Y, Liu J, Xie CH (2010) Novel potato C2H2-type zinc finger protein gene, StZFP1, which responds to biotic and abiotic stress, plays role in salt tolerance. Plant Biol 12:689–697. https://doi.org/10.1111/j.1438-8677.2009.00276.x
Varman PA (1999) Foliar disease resistant line developed through interspecific hybridization in groundnut (Arachis hypogaea). Indian J Agr Sci 69:67–68
Varshney RK, Bertioli DJ, Moretzsohn MD, Vadez V, Krishnamurthy L, Aruna R et al (2009) The first SSR-based genetic linkage map for cultivated groundnut (Arachis hypogaea L.). Theor Appl Genet 118:729–739. https://doi.org/10.1007/s00122-008-0933-x
Varshney RK, Pandey MK, Janila P, Nigam SN, Sudini H, Gowda MV et al (2014) Marker-assisted introgression of a QTL region to improve rust resistance in three elite and popular varieties of peanut (Arachis hypogaea L.). Theor Appl Genet 127:1771–1781. https://doi.org/10.1007/2Fs00122-014-2338-3
Wang J, Li H, Zhang L, Meng L (2016) User’s Manual of QTL Genetic Resources IciMapping Version 4.0, The Quantitative Genetics Group, Institute of Crop Science, Chinese Academy of Agricultural Sciences CAAS), Beijing 100081, China, and Genetic Program. International Maize and Wheat Improvement Center CIMMYT, Mexico, Mexico
Xu PL, Wang J, Fincher GB (1992) Evolution and differential expression of the 1→3-beta-glucan endohydrolase-encoding gene family in barley, Hordeum vulgare. Gene 120:157–165. https://doi.org/10.1016/0378-1119(92)90089-8
Yeri SB, Shirasawa K, Pandey MK, Gowda MV, Sujay V, Shriswathi M et al (2014) Development of NILs from heterogeneous inbred families for validating the rust resistance QTL in peanut (Arachis hypogaea L.). Plant Breed 133:80–85. https://doi.org/10.1111/pbr.12130
Yol E, Upadhyaya HD, Uzun B (2016) Identification of rust resistance in groundnut using a validated SSR marker. Euphytica 210:405–411. https://doi.org/10.1007/s10681-016-1705-3
Young ND, Weeden NF, Kochert G (1996) Genome mapping in legumes. In: Austin A, Paterson AH (eds) Genome mapping in plants. Landes Company, USA, pp 211–227
Acknowledgment
Financial support received from the Indian Council of Agricultural Research (ICAR), New Delhi, India, and Maulana Azad National Fellowship of UGC to the first author is gratefully acknowledged. The technical assistance in maintaining the plant population rendered by M.B. Sheikh and M.B. Kandoliya is also thankfully acknowledged.
Author information
Authors and Affiliations
Contributions
Conceptualized, Investigation and Supervision: GPM, RT, HPG; Data curation and analysis: SA, BN, CS, TCB; Project administration, methodology, and resources: NK, JRD; Writing—original draft: SA, GPM; Writing—review and editing: GPM, RT.
Corresponding authors
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical statements
The corresponding author on behalf of all the co-authors testifies that this manuscript has not currently being considered for publication or published in whole or in part in any other journal. Further, we also affirm that there are no other ethical issues of any sort are involved while performing any experiment mentioned in this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.


Rights and permissions
About this article
Cite this article
Ahmad, S., Nawade, B., Sangh, C. et al. Identification of novel QTLs for late leaf spot resistance and validation of a major rust QTL in peanut (Arachis hypogaea L.). 3 Biotech 10, 458 (2020). https://doi.org/10.1007/s13205-020-02446-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02446-4