Abstract
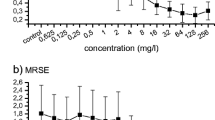
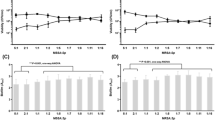
Staphylococcus aureus and Staphylococcus lugdunensis are often associated with pathogenic biofilms ranging from superficial mucosal to life-threatening systemic infections. Recent studies have reported that chelerythrine (CHE) displays antimicrobial activities against a few microorganisms, but its effects on dual-species biofilms of S. aureus and S. lugdunensis have never been reported. The purpose of this study was to investigate how dual-species biofilms of S. aureus and S. lugdunensis respond when challenged with CHE. Minimum inhibitory concentration (MIC) of CHE against planktic cells in dual-species culture was 8 μg/mL. CHE also suppressed dual-species biofilm formation at minimal biofilm inhibitory concentration (MBIC90, 4 μg/mL). Further, confocal laser scanning microscope (CLSM) using five fluorescent dyes revealed the dose-dependent reduction of the levels of three key biofilm matrix components, and reduced tolerance to gatifloxacin, of biofilms exposed to CHE. Moreover, CHE efficiently eradicated preformed dual-species biofilms at minimal biofilm eradication concentration (MBEC, 256 μg/mL). Hence, CHE has the potential to address biofilm infections of clinical course and other biofilm-related diseases caused by S. aureus and S. lugdunensis.








Similar content being viewed by others
References
Dai J et al (2019) Prevalence and characterization of Staphylococcus aureus isolated from pasteurized milk in China. Front Microbiol 10:641. https://doi.org/10.3389/fmicb.2019.00641
de Oliveira A, Cataneli Pereira V, Pinheiro L, Moraes Riboli DF, Benini Martins K, Ribeiro de Souza da Cunha Mde L (2016) Antimicrobial resistance profile of planktonic and biofilm cells of Staphylococcus aureus and coagulase-negative Staphylococci. Int J Mol Sci 17 Doi: 10.3390/ijms17091423
Frank KL, Del Pozo JL, Patel R (2008) From clinical microbiology to infection pathogenesis: how daring to be different works for Staphylococcus lugdunensis. Clin Microbiol Rev 21:111–133. https://doi.org/10.1128/CMR.00036-07
Guo X et al (2019) Effect of D-cysteine on dual-species biofilms of Streptococcus mutans and Streptococcus sanguinis. Sci Rep 9:6689. https://doi.org/10.1038/s41598-019-43081-1
He N, Wang P, Wang P, Ma C, Kang W (2018) Antibacterial mechanism of chelerythrine isolated from root of Toddalia asiatica (Linn) Lam. BMC Complement Altern Med 18:261. https://doi.org/10.1186/s12906-018-2317-3
Hobley L, Harkins C, MacPhee CE, Stanley-Wall NR (2015) Giving structure to the biofilm matrix: an overview of individual strategies and emerging common themes. FEMS Microbiol Rev 39:649–669. https://doi.org/10.1093/femsre/fuv015
Lee DH, Klinkova O, Kim JW, Nanjappa S, Greene JN (2019) A case series of Staphylococcus lugdunensis infection in cancer patients at an academic cancer institute in the United States. Infect Chemother 51:45–53. https://doi.org/10.3947/ic.2019.51.1.45
Li M, Huang R, Zhou X, Zhang K, Zheng X, Gregory RL (2014) Effect of nicotine on dual-species biofilms of Streptococcus mutans and Streptococcus sanguinis. FEMS Microbiol Lett 350:125–132. https://doi.org/10.1111/1574-6968.12317
Montazeri A, Salehzadeh A, Zamani H (2019) Effect of silver nanoparticles conjugated to thiosemicarbazide on biofilm formation and expression of intercellular adhesion molecule genes, icaAD, in Staphylococcus aureus. Folia Microbiol 65:153–160. https://doi.org/10.1007/s12223-019-00715-1
Olson ME, Ceri H, Morck DW, Buret AG, Read RR (2002) Biofilm bacteria: formation and comparative susceptibility to antibiotics. Can J Vet Res 66:86–92
Olszewska MA, Nynca A, Bialobrzewski I, Kocot AM, Laguna J (2019) Assessment of the bacterial viability of chlorine- and quaternary ammonium compounds-treated Lactobacillus cells via a multi-method approach. J Appl Microbiol 126:1070–1080. https://doi.org/10.1111/jam.14208
Oniciuc EA, Cerca N, Nicolau AI (2016) Compositional analysis of biofilms formed by Staphylococcus aureus isolated from food sources. Front Microbiol 7:390. https://doi.org/10.3389/fmicb.2016.00390
Qian W et al (2019) Antimicrobial activity of eugenol against carbapenem-resistant Klebsiella pneumoniae and its effect on biofilms. Microb Pathogenesis 139:103924. https://doi.org/10.1016/j.micpath.2019.103924
Rashed K, Ćirić A, Glamočlija J, Soković M (2014) Antibacterial and antifungal activities of methanol extract and phenolic compounds from Diospyros virginiana L. Ind Crops Prod 59:210–215. https://doi.org/10.1016/j.indcrop.2014.05.021
Ribeiro SM, de la Fuente-Nunez C, Baquir B, Faria-Junior C, Franco OL, Hancock RE (2015) Antibiofilm peptides increase the susceptibility of carbapenemase-producing Klebsiella pneumoniae clinical isolates to beta-lactam antibiotics. Antimicrob Agents Chemother 59:3906–3912. https://doi.org/10.1128/AAC.00092-15
Salehzadeh A, Zamani H, Langeroudi MK, Mirzaie A (2016) Molecular typing of nosocomial Staphylococcus aureus strains associated to biofilm based on the coagulase and protein A gene polymorphisms. Iran J Basic Med Sci 19:1325–1330. https://doi.org/10.22038/ijbms.2016.7919
Shen S, Zhang T, Yuan Y, Lin S, Xu J, Ye H (2015) Effects of cinnamaldehyde on Escherichia coli and Staphylococcus aureus membrane. Food Control 47:196–202. https://doi.org/10.1016/j.foodcont.2014.07.003
Shi C et al (2016) Antimicrobial activity and possible mechanism of action of citral against Cronobacter sakazakii. PLoS ONE 11:e0159006. https://doi.org/10.1371/journal.pone.0159006
Shin DS, Eom YB (2019) Efficacy of zerumbone against dual-species biofilms of Candida albicans and Staphylococcus aureus. Microb Pathog 137:103768. https://doi.org/10.1016/j.micpath.2019.103768
Tong SY, Davis JS, Eichenberger E, Holland TL, Fowler VG Jr (2015) Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev 28:603–661. https://doi.org/10.1128/CMR.00134-14
Van der Waal SV, de Almeida J, Krom BP, de Soet JJ, Crielaard W (2017) Diffusion of antimicrobials in multispecies biofilms evaluated in a new biofilm model. Int Endod J 50:367–376. https://doi.org/10.1111/iej.12634
Van der Mee-Marquet N, Achard A, Mereghetti L, Danton A, Minier M, Quentin R (2003) Staphylococcus lugdunensis infections: high frequency of inguinal area carriage. J Clin Microbiol 41:1404–1409. https://doi.org/10.1128/jcm.41.4.1404-1409.2003
Xu Y et al (2017) Antimicrobial activity of punicalagin against Staphylococcus aureus and Its effect on biofilm formation. Foodborne Pathog Dis 14:282–287. https://doi.org/10.1089/fpd.2016.2226
Yang XJ et al (2012) In vitro antifungal activity of sanguinarine and chelerythrine derivatives against phytopathogenic fungi. Molecules 17:13026–13035. https://doi.org/10.3390/molecules171113026
Acknowledgements
This study was funded by the Key Research and Development Project of Shaanxi Province (2019JM-184), and the Industry Cultivation Project of Education Department of Shaanxi Provincial Government (18JC006, 18JC007).
Author information
Authors and Affiliations
Contributions
Design of the work was performed by WQ. Acquisition of data, analysis and interpretation, drafting and critical revision, approval of the final version and agreement to be accountable for the work were performed by ZS, YF, MY, TW, and YL.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Qian, W., Sun, Z., Fu, Y. et al. Efficacy of chelerythrine against dual-species biofilms of Staphylococcus aureus and Staphylococcus lugdunensis. 3 Biotech 10, 427 (2020). https://doi.org/10.1007/s13205-020-02401-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02401-3