Abstract
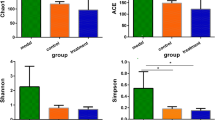
To probe into the mechanism of antibiotic-associated diarrhea (AAD), the bacterial diversity and composition in the intestinal mucosa of AAD mice were investigated. Twelve specific pathogen-free Kunming mice were divided into control group and model group. The mouse model of AAD was established by gavaging with antibiotics (mixture of gentamycin sulfate and cefradine) at a total dose of 23.33 ml kg−1 day−1 for 5 days continuously, twice a day. The mice in the control group were given with an equal amount of sterile water. Then, the intestinal mucosa DNA was extracted for 16S rRNA gene sequence analysis by high-throughput sequencing. The results showed that the alpha diversity of the two groups did not differ significantly from each other, while the composition of intestinal mucosa bacteria differed dramatically between the two groups. The model group showed a higher abundance of Proteobacteria and Actinobacteria. More importantly, Lactobacillus was significantly less abundant (p = 0.000), while Enterococcus was significantly more abundant (p = 0.019) in the model group than in the control group. Furthermore, antibiotic treatment increased the abundance of Citrobacter, Stenotrophomonas, and Glutamicibacter,whereas antibiotics decreased the abundance of Mycoplasma and Helicobacter. In addition, 6 and 11 unique genera were found in the control group and model group, respectively. The combination of gentamycin sulfate and cefradine changed the intestinal mucosa bacterial composition, reduced colonization resistance and damaged the intestinal mucosal barrier by reducing the abundance of Lactobacillus.






Similar content being viewed by others
References
Abbott IJ, Peleg AY (2015) Stenotrophomonas, Achromobacter, and nonmelioid Burkholderia species: antimicrobial resistance and therapeutic strategies. Semin Respir Crit Care Med 36(1):99–110. https://doi.org/10.1055/s-0034-1396929
Anand A, Bharadwaj R, Pol S (2017) Antibiotic associated diarrhea with special reference to Clostridium difficile. IJTDH 24(4):1–10. https://doi.org/10.9734/IJTDH/2017/34541
Borgo F, Garbossa S, Riva A, Severgnini M, Luigiano C, Benetti A (2018) Body mass index and sex affect diverse microbial niches within the gut. Front Microbiol 9:213–227. https://doi.org/10.3389/fmicb.2018.00213
Brandtzaeg P (2003) Mucosal immunity: integration between mother and the breast-fed infant. Vaccine 21(24):3382–3388. https://doi.org/10.1016/s0264-410x(03)00338-4
Calder P, Hall V (2012) Understanding gut-immune interactions in management of acute infectious diarrhea. Nurs Older People 24(9):29–37. https://doi.org/10.7748/nop2012.11.24.9.29.c9367
De Kivit S, Tobin MC, DeMeo MT, Fox S, Garssen J, Forsyth CB et al (2014) In vitro evaluation of intestinal epithelial TLR activation in preventing food allergic responses. Clin Immunol 154(2):91–99. https://doi.org/10.1016/j.clim.2014.07.002
Fitzgerald C (2015) Campylobacter. Clin Lab Med 35(2):289–298. https://doi.org/10.1016/j.cll.2015.03.001
Francino MP (2016) Antibiotics and the human gut microbiome: dysbiosis and accumulation of resistances. Front Microbiol 6:1543–1553. https://doi.org/10.3389/fmicb.2015.01543
Fuccio L, Guido A (2013) Probiotics supplementation for the prevention of gastrointestinal radiation-induced side effects: the time is now. Am J Gastroenterol 108(2):277. https://doi.org/10.1038/ajg.2012.418
Goh HMS, Yong MHA, Chong KKL, Kline KA (2017) Model systems for the study of Enterococcal colonization and infection. Virulence 8(8):1525–1562. https://doi.org/10.1080/21505594.2017.1279766
Guo KX, Yin KK, Wang H, Guo C, Zhao XP, Cao R (2014) The effect of modelling dysbacterial diarrhea with antibiotics on molecular diversity of intestinal microbiota in mice. Chin J Microecol 26(3):249–252. https://doi.org/10.13381/j.cnki.cjm.201403001
Hellming S, Ott S, Musfeldt M, Kosmahl M, Rosenstiel P, Stüber E et al (2005) Life-threatening chronic enteritis due to colonization of the small bowel with Stenotrophomonas maltophilia. Gastroenterology 129(2):706–712. https://doi.org/10.1016/j.gastro.2005.01.011
Hogenauer C, Hammer HF, Krejs GJ, Reisinger EC (1998) Mechanisms and management of antibiotic-associated diarrhea. Clin Infect Dis 27(4):702–710. https://doi.org/10.1086/514958
Jiang XT, Peng X, Deng GH, Sheng HF, Wang Y, Zhou HW et al (2013) Illumina sequencing of 16S rRNA tag revealed spatial variations of bacterial communities in a mangrove wetland. Microb Ecol 66(1):96–104. https://doi.org/10.1007/s00248-013-0238-8
Jin L, Yang XH, Ren JL, Li JL, Guo XY, Cao P et al (2012) Effect of dietary compound probiotics on disaccharidase in small intestine mucosa of layer breeders. Chin Poultry 34(12):14–17
Jin Y, Wu Y, Zeng Z, Jin C, Wu S, Wang Y et al (2016) From the cover: exposure to oral antibiotics induces gut microbiota dysbiosis associated with lipid metabolism dysfunction and low-grade inflammation in mice. Toxicol Sci 154(1):140–152. https://doi.org/10.1093/toxsci/kfw150
Jirillo E, Jirillo F, Magrone T (2012) Healthy effects exerted by prebiotics, probiotics and symbiotics with special reference to their impact on the immune system. Int J Vitam Nutr Res 82(3):200–208. https://doi.org/10.1024/0300-9831/a000112
Kim YJ, Choi YS, Baek KJ, Yoon SH, Park HK, Choi Y (2016) Mucosal and salivary microbiota associated with recurrent aphthous stomatitis. BMC Microbiol 16(Suppl 1):57. https://doi.org/10.1186/s12866-016-0673-z
Larcombe S, Hutton ML, Lyras D (2016) Involvement of bacteria other than Clostridium difficile in antibiotic-associated diarrhoea. Trends Microbiol 24(6):463–476. https://doi.org/10.1016/j.tim.2016.02.001
Lee Y, Eun CS, Lee AR, Park CH, Han DS (2016) Fusobacterium isolates recovered from colonic biopsies of inflammatory bowel disease patients in Korea. Ann Lab Med 36(4):387–389. https://doi.org/10.3343/alm.2016.36.4.387
Long CX, He L, Guo YF, Liu YW, Xiao NQ, Tan ZJ (2017) Diversity of bacterial lactase genes in intestinal contents of mice with antibiotics-induced diarrhea. World J Gastroenterol 23(42):7584–7593. https://doi.org/10.3748/wjg.v23.i42.7584
Long CX, Liu YW, He L, Tan Q, YU Z, Xiao NQ et al (2018) Bacterial lactase genes diversity in intestinal mucosa of mice with dysbacterial diarrhea induced by antibiotics. 3 Biotech 8(3):176
Lu YY, Zeng Y, Hu GY, Wang XP (2017) High-throughput sequencing for analysis of structural change of intestinal microbiota in patients with colorectal adenoma. J South Med Univ 37(9):1156–1163. https://doi.org/10.3969/j.issn.1673-4254.2017.09.03
Nishino K, Nishida A, Inoue R, Kawada Y, Ohno M, Sakai S (2018) Analysis of endoscopic brush samples identified mucosa-associated dysbiosis in inflammatory bowel disease. J Gastroenterol 53(1):95–106. https://doi.org/10.1007/s00535-017-1384-4
Noval Rivas M, Burton OT, Wise P, Zhang YQ, Hobson SA, Garcia Llore M et al (2013) A microbiota signature associated with experimental food allergy promotes allergic sensitization and anaphylaxis. J Allergy Clin Immunol 131(1):201–212. https://doi.org/10.1016/j.jaci.2012.10.026
Puhl NJ, Uwiera RR, Yanke LJ, Selinger LB, Inglis GD (2012) Antibiotics conspicuously affect community profiles and richness, but not the density of bacterial cells associated with mucosa in the large and small intestines of mice. Anaerobe 18(1):67–75. https://doi.org/10.1016/j.anaerobe.2011.12.007
Rodrigues RR, Greer RL, Dong X, DSouza KN, Gurung M, Gurung Y et al (2017) Antibiotic-induced alterations in gut microbiota are associated with changes in glucose metabolism in healthy mice. Front Microbiol 8:2306. https://doi.org/10.3389/fmicb.2017.02306
Schloss PD, Gevers D, Westcott SL (2011) Reducing the effects of PCR amplification and sequencing artifacts on 16S rRNA-based studies. PLoS One 6(12):e27310. https://doi.org/10.1371/journal.pone.0027310
Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS et al (2011) Metagenomic biomarker discovery and explanation. Genome Biol 12(6):R60. https://doi.org/10.1186/gb-2011-12-6-r60
Shin NR, Whon TW, Bae JW (2015) Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol 33(9):496–503. https://doi.org/10.1016/j.tibtech.2015.06.011
Silverman MA, Konnikova L, Gerber JS (2017) Impact of antibiotics on necrotizing enterocolitis and antibiotic-associated diarrhea. Gastroenterol Clin North Am 46(1):61–76. https://doi.org/10.1016/j.gtc.2016.09.010
Spigaglia P, Mastrantonio P, Barbanti F (2018) Antibiotic resistances of Clostridium difficile. In: Mastrantonio P, Rupnik M (eds) Updates on Clostridium difficile in Europe. Springer, Berlin
Stecher B, Maier L, Hardt WD (2013) ‘Blooming’ in the gut: how dysbiosis might contribute to pathogen evolution. Nat Rev Microbiol 11(4):277–284. https://doi.org/10.1038/nrmicro2989
Su L, Zhang YB, Zhou RB (2013) Treatment and prevention of antibiotic associated diarrhea. J Clin Emerg 14(5):240–242. https://doi.org/10.1093/toxsci/kfw150
Tian H, Zhao HL, Yang L, Wang N (2018) Research progress of the microecology and intestinal mucosal barrier. Basic Clin Med 38(3):418–421. https://doi.org/10.16352/j.issn.1001-6325.2018.03.026
Wang Y, Sheng HF, He Y, Wu JY, Jiang YX, Tam NF et al (2012) Comparison of the levels of bacterial diversity in fresh water, intertidal wetland, and marine sediments by using millions of Illumina tags. Appl Environ Microbiol 78(23):8264–8267. https://doi.org/10.1128/AEM.01821-12
Wu H, Zhou SN, Guo C, Tan ZJ, Cai GX, Zeng A et al (2012) A metagenome DNA extracting method of intestinal flora in mice for molecular diversity analysis based on PCR technology. Chin J Microecol 24(7):648–651. https://doi.org/10.13381/j.cnki.cjm.2012.07.003
Zeng A, Zhang HL, Tan ZJ, Cai Y, Cai GX, Zhou SN (2012) The construction of mice diarrhea model due to dysbacteriosis and curative effect of ultra-micro Qiweibaizhusan. Microbiol Chin 39(9):1341–1348. https://doi.org/10.13344/j.microbiol.china.2012.09.012
Zhang HL, Zhou SN, Cai R, Guo KX, She Y, Tan ZJ et al (2013) The effect of ultra-micro Qiweibaizhusan on the mucous membrane of small intestine of diarrheal mice with dysbacteriosis. Chin J Microecol 25(1):9–14. https://doi.org/10.13381/j.cnki.cjm.2013.01.007
Zhang HL, Cai Y, Tan ZJ, Zhou SN, Guo KX, She Y (2014) Effects of ultra-micro powder Qiweibaizhusan on metabolism diversity of intestinal microflora in diarrhea mice with dysbacteriosis. Chin J Appl Environ Biol 20(1):93–100. https://doi.org/10.3724/SP.J.1145.2014.00093
Zhang Q, Zhou ZK, Ren XC, Wang XF (2017) Comparision of faecal microbiota in rats with type 2 diabetes and non-diabetic rats using MiSeq high-throughput sequencing. J Chin Instit Food Sci Technol 17(6):232–239. https://doi.org/10.16429/j.1009-7848.2017.06.031
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81573951; 81804076).
Author information
Authors and Affiliations
Contributions
ZT designed the study; KT and MP performed the experiments; GX and CL analyzed the data; GX wrote the paper; DL checked the paper. The decision to submit the manuscript for publication was made by all the authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Human and animal rights
The study was approved by the Animal Ethics and Welfare Committee of Hunan University of Chinese Medicine.
Rights and permissions
About this article
Cite this article
Xie, G., Tan, K., Peng, M. et al. Bacterial diversity in intestinal mucosa of antibiotic-associated diarrhea mice. 3 Biotech 9, 444 (2019). https://doi.org/10.1007/s13205-019-1967-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1967-2