Abstract
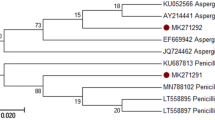
The objective of the present work was to evaluate the water hyacinth (WH) as a substrate for the production of hydrolytic enzymes (cellulases and hemicellulases) of 100 strains of filamentous fungi under conditions of solid growth. Five fungal strains, identified as Trichoderma harzianum, Trichoderma atroviride, Penicillium griseofulvum, Penicillium commune and Aspergillus versicolor, were selected and studied for their ability to grow on water hyacinth as a substrate and carbon source only, evaluating hydrolytic enzymatic activities (α-l-arabinofuranosidase, cellulase, xylanase and β-d-xylopyranosidase) and extracellular protein per g of water hyacinth dry matter (gdm). The five strains selected were able to produce the four enzymes studied; however, T. harzianum strain PBCA produces the highest xylanase (149.3 ± 14.3 IU/gdm at 108 h), cellulase (16.4 ± 0.6 IU/gdm at 84 h) and β-d-xylopyranosidase (127.7 ± 14.8 IU/gdm at 48 h). In contrast, the fungus with the highest α-l-arabinofuranosidase activity was A. versicolor, with 129.8 ± 13.3 IU/gdm after 108 h. In conclusion, T. harzianum showed the best production of the hydrolytic enzymes studied, using as a matrix and carbon source, water hyacinth. In addition, catalytic activities of arabinofuranosidase and xylopyranosidase were reported for the first time in T. versicolor and T. harzianum.



Similar content being viewed by others
References
Abraham M, Kurup M (1996) Bioconversion of tapioca (Manihot esculenta) waste and water hyacinth (Eichhornia crassipes)—Influence of various physico-chemical factors. J Ferment Bioeng 82(3):259–263. https://doi.org/10.1016/0922-338X(96)88817-9
Ahmed S, Imdad SS, Jamil A (2012) Comparative study for the kinetics of extracellular xylanases from Trichoderma harzianum and Chaetomium thermophilum. Electron J Biotechnol 15:3–3. https://doi.org/10.2225/vol15-issue3-fulltext-2
Baffi MA, Romo-Sánchez S, Úbeda-Iranzo J, Briones-Pérez AI (2012) Fungi isolated from olive ecosystems and screening of their potential biotechnological use. New Biotechnol 29:451–456. https://doi.org/10.1016/S0141-0229(96)00236-0
Balasubramanian D, Arunachalam K, Das AK, Arunachalam A (2012) Decomposition and nutrient release of Eichhornia crassipes (Mart.) Solms under different trophic conditions in wetlands of eastern Himalayan foothills. Ecol Eng 44:111–122. https://doi.org/10.1016/j.ecoleng.2012.03.002
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Browning BL (1967) Methods of wood chemistry, vol I & II. Wiley, New York, USA
Cervera-Tison M, André-Leroux G, Lafond M, Georis J, Juge N, Berrin JG (2009) Molecular determinants of substrate and inhibitor specificities of the Penicillium griseofulvum family 11 xylanase. Biochim Biophys Acta 1794:438–445. https://doi.org/10.1016/j.bbapap.2008.11.024
Chutani P, Sharma KK (2015) Biochemical evaluation of xylanases from various filamentous fungi and their application for the deinking of ozone treated newspaper pulp. Carbohidrate Polymers 127:54–63. https://doi.org/10.1016/j.carbpol.2015.03.053
De La Cruz J, Pintor-Toro JA, Benitez T, LLobell A (1995) Purification and characterization of an endo-beta-1, 6-glucanase from Trichoderma harzianum that is related to its mycoparasitism. J Bacteriol 177(7):1864–1871. https://doi.org/10.1128/jb.177.7.1864-1871-1995
Cruz-Ramírez MG, Rivera-Ríos JM, Téllez-Jurado A, Maqueda Gálvez AP, Mercado-Flores Y, Arana-Cuenca A (2102) Screening of thermotolerant ligninolytic fungi with laccase, lipase, and protease activity isolated in México. J Environ Manag 95:S256–S259. https://doi.org/10.1016/j.jenvman.2010.10.045
Das SP, Gupta A, Das D, Goyal A (2016) Enhanced bioethanol production from water hyacinth (Eichhornia crassipes) by statistical optimization of fermentation process parameters using Taguchi orthogonal array design. Int Biodeterior Biodegrad 109:174–184. https://doi.org/10.1016/j.ibiod.201601.008
da Silva Delabona P, Cota J, Hoffmam ZB, Paixão DAA, Farinas CS, Cairo JPLF, Lima DJ, Squina FM, Ruller R, da Cruz Pradella JG (2013) Understanding the cellulolytic system of Trichoderma harzianum P49P11 and enhancing saccharification of pretreated sugarcane bagasse by supplementation with pectinase and α-l-arabinofuranosidase. Bioresour Technol 131:500–507. https://doi.org/10.1016/j.biortech.2012.12.105
de Souza MF, da Silva ASA, Bon EP (2018) A novel Trichoderma harzianum strain from the Amazon Forest with high cellulolytic capacity. Biocatal Agric Biotechnol 14:183–188. https://doi.org/10.1016/j.bcab.2018.03.008
Do Vale LH, Gómez-Mendoza DP, Kim KS, Pandey A, Ricart CA, Edivaldo XF, Sousa MV (2012) Secretome analysis of the fungus Trichoderma harzianum grown on cellulose. Proteomics 12:2716–2728. https://doi.org/10.1002/pmic.201200063
El-Katatny M, Gudelj M, Robra KH, Elnaghy M, Gübitz G (2001) Characterization of a chitinase and an endo-β-1, 3-glucanase from Trichoderma harzianum Rifai T24 involved in control of the phytopathogen Sclerotium rolfsii. Appl Microbiol Biotechnol 56(1–2):137–143. https://doi.org/10.1007/s002530100646
Grootaert C, Delcour JA, Courtin CM, Broekaert WF, Verstraete W, Van de Wiele T (2007) Microbial metabolism and prebiotic potency of arabinoxylan oligosaccharides in the human intestine. Trends Food Sci Technol 18:64–71. https://doi.org/10.1016/j.tifs.2006.08.004
Gunnarsson CC, Petersen CM (2007) Water hyacinth as a resource in agriculture and energy production: a literature review. Waste Manag 27(1):117–129. https://doi.org/10.1016/j.wasman.2005.12.011
Guragain YN, Coninck JD, Husson F, Durand A, Rakshit SK (2011) Comparison of some new pretreatment methods for second generation bioethanol production from wheat straw and water hyacinth. Bioresour Technol 102:4416–4424. https://doi.org/10.1016/j.biortech.2010.11.125
Ikram-Ul-Haq UH, Shahzadi K, Javed MSA, Qadeer MA (2005) Cotton saccharifying activity of cellulases by Trichoderma harzianum UM-11 in shake flask. J Bot 1(1):19–22
Jafari NG (2010) Ecological and socio-economic utilization of water hyacinth (Eichhornia crassipes Mart Solms). J Appl Sci Environ Manag 14:43–49. https://doi.org/10.4314/jasem.v14i2.57834
Juárez-Luna G, Perraud-Gaime I, Juárez-Cárdenas I, Favela-Torres MT, Toribio-Cueva H, Pedraza-Segura L, Favela-Torres E (2011) Composición de la fracción lignocelulósica del lirio acuático (Eichhornia crassipes) extenso memory, AMIDIQ, Quintana Roo México, pp 2162–2166
Karmakar M, Ray RR (2011) A statistical approach for optimization of simultaneous production of β-glucosidase and endoglucanase by Rhizopus oryzae from solid-state fermentation of water hyacinth using central composite design. Biotechnol Res Int 2011:574983. https://doi.org/10.4061/2011/574983
Kasana RC, Salwan R, Dhar H, Dutt S, Gulati A (2008) A rapid and easy method for the detection of microbial cellulases on agar plates using gram’s iodine. Curr Microbiol 57:503–507. https://doi.org/10.1007/s00284-008-9276-8
Kovács K, Megyeri L, Szakacs G, Kubicek CP, Galbe M, Zacchi G (2008) Trichoderma atroviride mutants with enhanced production of cellulase and B-glucosidase on pretreated willow. Enzyme Microb Technol 43:48–55. https://doi.org/10.1016/j.enzmictec.2008.02.006
Kumar A, Singh LK, Ghosh S (2009) Bioconversion of lignocellulosic fraction of water hyacinth (Eichhornia crassipes) hemicellulose hydrolysate to ethanol by Pichia stipitis. Bioresour Technol 100:3293–3297. https://doi.org/10.1016/j.biortech.2009.02.023
Lee S, Jang Y, Lee YM, Lee J, Lee H, Kim GH, Kim JJ (2011) Rice straw-decomposing fungi and their cellulolytic and xylanolytic enzymes. J Microbiol Biotechnol 21:1322–1329. https://doi.org/10.4014/jmb.1107.07022
López-Ramírez N, Volke-Sepúlveda T, Gaime-Perraud I, Saucedo-Castañeda G, Favela-Torres E (2018) Effect of stirring on growth and cellulolytic enzymes production by Trichoderma harzianum in a novel bench-scale solid-state fermentation bioreactor. Bioresour Technol 265:291–298. https://doi.org/10.1016/j.biortech.2018.06.015
Muñoz-Gutiérrez I, Martinez A (2013) Polysaccharide hydrolysis with engineered Escherichia coli for the production of biocommodities. J Ind Microbiol Biotechnol 40:401–410. https://doi.org/10.1007/S10295-013.1245-y
Nigam JN (2002) Bioconversion of water-hyacinth (Eichornia crassipes) hemicellulose acid hydrolysate to motor fuel ethanol by xylose fermenting yeast. J Biotechnol 97:107–116. https://doi.org/10.1016/S0168-1656(02)00013-5
Okunowo WO, Gbenle GO, Osuntoki AA, Adekunie AA, Ojokuku SA (2010) Production of cellulolytic and xylanolytic enzyme by a phytopathogenic Myrothecium roridum and some avirulent fungal isolates from water hyacinth. Afr J Biotech 9:1074–1078. https://doi.org/10.4314%2Fajb.v9i7
Pathak P, Bhardwaj NK, Singh AK (2014) Production of crude cellulase and xylanase from Trichoderma harzianum PPDDN10 NFCCI-295 and its application in photocopier waste paper recycling. Appl Biochem Biotechnol 172(8):3776–3797. https://doi.org/10.1007/s12010-014-0758-9
Pontecorvo G, Roper JA, Chemmons LM, MacDonald KD, Bufton AWJ (1953) The genetics of Aspergillus nidulans. Adv Genet 5:14–238. https://doi.org/10.1016/S0065-2660(08)60408-3
Páramo-Aguilera L, Ortega-Morales BO, Narváez-Zapata JA (2012) Culturable fungi associated with urban stone surfaces in Mexico City. Electron J Biotechnol 15:4–4. https://doi.org/10.2225/vol15-issue4-fulltext-6
Quintanar Gómez S, Arana-Cuenca A, Mercado Flores Y, Gracida Rodríguez JN, Téllez-Jurado A (2012) Effect of particle size and aeration on the biological delignification of corn straw using Trametes sp. 44. BioResources 7:327–344
Raeder U, Broda P (1985) Rapid preparation of DNA from filamentous fungi. Lett Appl Microbiol 1:17–20. https://doi.org/10.1111/j.1472-765X.1985.tb01479.x
Rahnama N, Mamat S, Shah MDU, Ling F, Rahman A, Ariff N A (2013) Effect of alkali pretreatment of rice straw on cellulase and xylanase production by local Trichoderma harzianum SNRS3 under solid state fermentation. BioResources 8(2):2881–2896. http://ojs.cnr.ncsu.edu/index.php/BioRes/article/view/3781
Rezende MI, Barbosa AM, Vasconcelos AFD, Endo AS (2002) Xylanase production by Trichoderma harzianum rifai by solid state fermentation on sugarcane bagasse. Braz J Microbiol 33(1):67–72. https://doi.org/10.1590/S1517-83822002000100014
Runa A, Krishman KP, Sabu T, Wilson PA, Thamban M (2009) Phenotypic and molecular identification of Cellulosimicrobium cellulans isolated from Antarctic snow. Anton Leeuw 96:627–634. https://doi.org/10.1007/s10482-009-9377-9
Samanta AK, Jayapal N, Kolte AP, Senani S, Sridhar M, Suresh KP, Sampath KT (2012) Enzymatic production of xylooligosaccharides from alkali solubilized xylan of natural grass (Sehima nervosum). Bioresour Technol 112:199–205. https://doi.org/10.1016/j.biortech.2012.02.036
Saravanakumar K, Yu C, Dou K, Wang M, Li Y, Chen J (2016) Synergistic effect of Trichoderma-derived antifungal metabolites and cell wall degrading enzymes on enhanced biocontrol of Fusarium oxysporum f. sp. Cucumerinum. Biolog Control 94:37–46. https://doi.org/10.1016/j.biocontrol.2015.12.001
Sedlmeyer FB (2011) Xylan as by-product of biorefineries: characteristics and potential use for food applications. Food Hydrocoll 25:1891–1898. https://doi.org/10.1016/j.foodhyd.2011.04.005
Seiboth B, Metz B (2011) Fungal arabinan and L-arabinose metabolism. Appl Microbiol Biotechnol 89:1665–1673. https://doi.org/10.1007/s00253-010-3071-8
Sornvoraweat B, Kongkiattikajorn J (2010) Separated hydrolysis and fermentation of water hyacinth leaves for ethanol production. KKU Res J 15(9):794–802
TAPPI T204 om-84 (1987) Solvent extractives of wood and pulp. TAPPI Press, Atlanta, GA
TAPPI T207 om-93 (1993) Water solubility of wood and pulp. TAPPI Press, Atlanta, GA
TAPPI T211 om-93 (1993) Ash in wood, pulp, paper and paperboard: combustion at 525 °C. TAPPI Press, Atlanta, GA
TAPPI T222 om-88 (1988) Acid insoluble lignin in wood and pulp. TAPPI Press, Atlanta, GA
Tagawa K, Kaji A (1988) α-l-Arabinofuranosidase from Aspergillus niger. Method Enzymol 160:707–712. https://doi.org/10.1016/0076-6879(88)60191-1
Tham HT (2012) Water hyacinth (Eichhornia crassipes)—Biomass production, ensilability and feeding value to growing cattle. Doctoral Thesis. Swedish University of Agricultural Sciences. Uppsala. Mekong Delta, Vietnam
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. A guide to methods and applications. Academic Press, San Diego, pp 315–322
Zhang X, Li Y, Zhao X, Bai F (2017) Constitutive cellulose production from glucose using the recombinant Trichoderma reesei strain over expressing an artificial transcription activator. Bioresour Technol 223:317–322. https://doi.org/10.1016/j.biortech.2016.10.083
Zhou P, Zhu H, Yan Q, Katrolia P, Jiang Z (2011) Purification and properties of a psychotrophic Trichoderma sp. xylanase and its gene sequence. Appl Biochem Biotechnol 164(6):944–956. https://doi.org/10.1007/s12010-011-9186-2
Zyani M, Mortabit D, Mostakim M, Iraqui M, Haggoud A, Ettayebi M, Koraichi SI (2009) Cellulolytic potential of fungi in wood degradation from an old house at the Medina of Fez. Ann Microbiol 59:699–704. https://doi.org/10.1007/BF03179211
Acknowledgements
This work was financially supported by the FONCICYT-C002-2008-1 ALA/127249 project. The support to collect water hyacinth by TEMA S.A. is acknowledged with thanks.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Arana-Cuenca, A., Tovar-Jiménez, X., Favela-Torres, E. et al. Use of water hyacinth as a substrate for the production of filamentous fungal hydrolytic enzymes in solid-state fermentation. 3 Biotech 9, 21 (2019). https://doi.org/10.1007/s13205-018-1529-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1529-z