Abstract
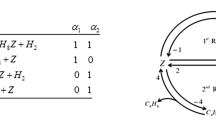
To study the impact of reaction rate coefficients, a two-step mechanism is considered in this article. The computational results are calculated using the different values of the rate coefficients. To inspect the behavior of the species for different values of the rate coefficients, their activation energy and transition period have been measured before attaining equilibrium. Comparison between the varying values of rate coefficients are graphically compared through MATLAB programing.







Similar content being viewed by others
References
Ali M, Shahzad M, Sultan F, Khan WA (2019) Physical assessments on the invariant region in multi-route reaction mechanism. Physica A Stat Mech Appl. https://doi.org/10.1016/j.physa.2019.122499
Aris R (1965) Prolegomena to the rational analysis of systems of chemical reactions. Arch Ration Mech Anal 19:81
Constales D, Yablonsky GS, D’hooge DR (2016) Advanced data analysis and modelling in chemical engineering. Elsevier, Amsterdam
Gorban AN, Shahzad M (2011) The Michaelis–Menten–Stueckelberg theorem. Entropy 13:966
Maxwell JC (1867) On the dynamical theory of gases. Philos Trans R Soc 157:49
Shahzad M, Sultan F (2018) Complex reactions and dynamics: advanced chemical kinetics. InTech, Rijeka. https://doi.org/10.5772/intechopen.70502
Shahzad M, Sajid M, Gulistan M, Arif H (2015) Initially approximated quasi equilibrium manifold. J Chem Soc Pak 37(2)
Shahzad M, Sultan F, Haq I, Ali M, Khan WA (2019a) C-matrix and invariants in chemical kinetics: a mathematical concept. Pramana J Phys. https://doi.org/10.1007/s12043-019-1723-5
Shahzad M, Sultan F, Shah SIA, Ali M, Khan HA, Khan WA (2019b) Physical assessments on chemically reacting species and reduction schemes for the approximation of invariant manifolds. J Mol Liq 285:237–243. https://doi.org/10.1016/j.molliq.2019.03.031
Shahzad M, Sultan F, Ali M, Khan WA, Irfan M (2019c) Slow invariant manifold assessments in multi-route reaction mechanism. J Mol Liq 284:265–270
Shahzad M, Sun H, Sultan F, Khan WA, Ali M, Irfan M (2019d) Transport of radiative heat transfer in dissipative cross nanofluid flow with entropy generation and activation energy. Phys Scr 94:115224
Shahzad M, Sultan F, Ali M, Khan WA, Mustafa S (2020a) Modeling multi-route reaction mechanism for surfaces: a mathematical and computational approach. Appl Nanosci. https://doi.org/10.1007/s13204-020-01275-4
Shahzad M, Sultan F, Ali M, Khan WA, Mustafa S (2020b) Modeling multi-route reaction mechanism for surfaces: a mathematical and computational approach. Appl Nanosci. https://doi.org/10.1007/s13204-020-01275.S
Sultan F, Khan WA, Shahzad M, Ali M, Shah SIA (2019a) Activation energy characteristics of chemically reacting species in multi-route complex reaction mechanism. Indian J Phys. https://doi.org/10.1007/s12648-019-01624-2
Sultan F, Shahzad M, Ali M, Khan WA (2019b) The reaction routes comparison with respect to Slow Invariant Manifold and equilibrium points. AIP Adv 9:015212. https://doi.org/10.1063/1.5050265
Yaublonsky GS, Branco D, Marin GB, Constales D (2020) New invariant expansions in chemical kinetics. Entropy 22:373
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conficts of interest.
Ethical approval
This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ali, M., Shahzad, M., Sultan, F. et al. Computational analysis on transition time-period in complex reaction mechanism. Appl Nanosci 12, 273–281 (2022). https://doi.org/10.1007/s13204-020-01645-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-020-01645-y