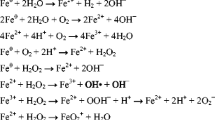Abstract
Metallic iron (Fe0) has been increasingly used to remove toxics from water over the past three decades. However, the idea that metallic iron (Fe0) is not an environmental reducing agent has been vigorously refuted. Researchers presenting their findings in a scientific journal have to accept the burden of proving that their argument has any validity. This 30-year-lasting discussion within the Fe0 remediation community is alien to electro-chemists, as it is a century-old knowledge. Nevertheless, the peer-reviewed literature on “remediation using Fe0” seems to be dominated by evaluators thinking that Fe0 is a reducing agent. This communication challenges the view that Fe0 donates any electron to any dissolved species. The sole goal is to reconcile a proven efficient technology with its scientific roots and enable the design of better Fe0 remediation systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Metallic iron for environmental remediation
Permeable reactive barriers (PRBs) as an effective remediation technology for polluted groundwater were introduced in the 1980s (McMurty and Elton 1985). Upon careful design and installation, PRBs remediate groundwater polluted with diverse contaminants, including chlorinated hydrocarbons, nutrients, and trace metals (Bigg and Judd 2000; Blowes et al. 2000; Thiruvenkatachari et al. 2008; Obiri-Nyarko et al. 2014; Courcelles 2015). Metallic iron (Fe0) has been tested the most as a filling material for PRBs (Warner 2015; Gheju 2018). The suitability of Fe0 for groundwater remediation is due to the negative value of the electrode potential of the FeII/Fe0 redox couple (E0 = − 0.44 V) (Nesic 2007). This makes Fe0 a potential reducing agent for many dissolved species (E0 > − 0.44 V), and even for water (E0 = 0.00 V) (Thiruvenkatachari et al. 2008; Courcelles 2015). Water is the solvent and thus stoichiometrically very abundant (55.5 mol L−1). At pH ranges above 5.0, the reaction between Fe0 and water produces an oxide layer (oxide scale) close to the Fe0 surface which is of fundamental importance for the efficiency of Fe0 PRBs. In other words, the oxide scale on Fe0 does act as a contaminant scavenger (Obiri-Nyarko et al. 2014; Gheju 2018). Despite three decades of intensive investigations, the mechanistic discussion on the operation mode of Fe0/H2O systems is still on-going (Warner 2015; Cai et al. 2021).
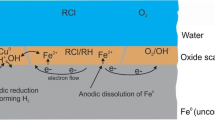
In 1994, Matheson and Tratnyek published highly counter-intuitive findings that metallic iron (Fe0) could be oxidized by dissolved contaminants at circum-neutral pH values. Their results were reported to be confirmed two years later by Weber (1996). Since then, environmental scientists are mostly considering Fe0 as a reducing agent for relevant aqueous species, including chlorinated organics (RCl) (O’Hannesin and Gillham 1998). RCl reductive transformation is considered to be a cathodic half-reaction occurring simultaneously with the anodic Fe0 oxidative dissolution (Eq. 1):
Any researcher familiar with the Fe0 remediation technology will confirm that this is the state-of-the-art knowledge (Obiri-Nyarko et al. 2014; Gheju 2018; Chen et al. 2019; Thakur et al. 2020). Any reader interested in following the path of the paradigm from Reynolds et al. (1990) is encouraged to read Gillham (2008). In fact, Reynolds et al. (1990) fortuitously found that trichloroethylene disappeared in Fe0-based vessels (Lee et al. 2004; Gillham 2008). This is virtually considered as the starting point for the Fe0 remediation technology which is now 30 years old and widely recognized as a competent alternative technology for remediation of contaminated groundwater (Lee et al. 2004; Gillham 2008; Chen et al. 2019; Thakur et al. 2020) and the provision of safe drinking water (Giles et al. 2011; Neumann et al. 2013; Huang et al. 2021a, b). In this paper, we contrast the misconception of Matheson and Tratnyek (1994) to the fundamental science of aqueous iron corrosion (Fe0/H2O system) (Whitney 1903).
The science of the Fe0/H2O system
In 1903, Willis Rodney Whitney established the science of the Fe0/H2O system which convincingly demonstrated that, at pH above 4.5 and under immersed conditions, only water can oxidize Fe0 by an electrochemical mechanism according to Eq. 2 (Whitney 1903):
In particular, Whitney (1903) demonstrated two important facts: (1) carbonic acid (H2CO3) accelerates corrosion by supplementing protons (H+) in Eq. 2 and intensifying water dissociation, and (2) dissolved oxygen (O2) accelerates corrosion by consuming Fe2+ and favoring the forward reaction in Eq. 2 (i.e., Le Chatelier's principle). In other words, all reports on direct aqueous Fe0 corrosion by O2 are faulty. In terms of chemistry, H2CO3 and O2 enhance iron corrosion by an indirect mechanism or following the Le Chatelier's principle. O2 reduction by Fe2+ is given by Eq. 3 (Stratmann and Müller 1994):
This is basic knowledge of a chemist. Similarly, the chemical reduction of RCl by FeII species should be written as in Eq. 4 (Fig. 1):
For the sake of simplicity, the pH-dependent speciation of Fe ions (FeII and FeIII) is not considered in Eqs. 3 and 4. To this point, the presentation demonstrates that the view that under environmental conditions (pH > 4.5) the reductive transformation of RCl in the presence of Fe0 is an electrochemical reaction (Fig. 1a) is contrary to basic chemistry textbook knowledge (Fig. 1b). Thus, the Fe0 remediation literature has distorted the basic science of aqueous iron corrosion. In this regard, the Fe0 remediation research community has perpetuated and propagated the mistake for years now despite various efforts drawing attention to this error as summarized by Hu et al. (2020). This mistake is attributed to insufficient literature review as the seminal paper by Whitney (1903) was then 91 years old. Recent trends in journal editorial and review processes emphasizing the review and citation of recent literature have also contributed to the current scenario where researchers exclude “old” but correct knowledge, while simultaneously introducing conceptual errors. Clearly, researchers trying to restore the integrity of science now bear the burden of proof and face resistance from experts (e.g., grant referees and journal reviewers). However, those researchers have basically no proofs to bring as they are just correcting an error which has been propagated over the years. The error has developed to be an “acceptable truth” by virtue of mere repeated citations, but it is common knowledge that repeating an error does not make it the truth (Noubactep 2019). In addition, active researchers may also have the tendency to become attached to their ideas no matter how flawed they might be. Such a situation has made publishing on “Fe0 for water remediation” becoming an irrational discussion among scientists, rather than a rational competition among ideas underpinned by fundamental scientific knowledge (Chamberlin 1897; Fudge 2014).
For contaminant removal in Fe0/H2O systems, there should be a recognizable and universal reality based on scientific principles. In this case, it is about the corrosion science of metallic iron in aqueous environments. To reach a new reality, “good explanations should rise out of the ashes of those (assumptions) shown to be false” (Fudge 2014). Accordingly, scientists seeking to reach the “recognizable and universal reality” on the fate of contaminants in Fe0/H2O systems should strive to entertain the two theories given above as concurrent hypotheses and find out which one enables a better explanation of their experimental results. This is currently not the case as even “critical” reviews are written without any hint on the controversy existing in the literature with respect to the mechanisms of contaminants removal in Fe0/H2O systems (Noubactep 2014; Thakur et al. 2020). This communication is not addressing the related controversies; interested readers are referred to the following five representative articles: Gheju and Balcu (2011), Ghauch (2015), Noubactep (2014), Hu et al. (2021b), and Konadu-Amoah et al. (2022).
The mistaken path of the recent Fe0 remediation research
Fe0 has been industrially employed for water treatment since the 1850s (Tucker 1892; Baker 1934, 1948; Van Craenenbroeck 1998; Mwakabona et al. 2017). Selected outstanding patents were: (1) Medlock (1857) producing iron oxide by merely suspending iron rods in water; (2) Spencer (1857) using magnetic carbide of iron in household filters; (3) Bischof (1870) using spongy iron initially in household filters; (4) Anderson (1880) using iron fillings in a revolving cylinder (revolving purifier); and (5) Sellers (1890) using suspended iron rods while making the rods revolve. In particular, the Bischof process and the Anderson process were put into use in some cities, but the total number of plants remained small and all were soon abandoned after the First World War (Baker 1948). In fact, no Fe0-based water treatment plant seems to have survived the First World War (Van Craenenbroeck 1998). This ancient use of Fe0 was not considered in the post-1990 literature until 2017 (Mwakabona et al. 2017; Antia 2020). The reason why the ancient Fe0 water treatment technology failed has never been scientifically investigated. From Baker (1948), it is clear that filtration systems failed because of cementation of gravel/sand and iron causing clogging, while fluidized systems were initially very efficient but not sustainable.
The key result of the ancient use of Fe0 for water treatment is that it was an attempt at achieving coagulation (Baker 1948). Fe0 was used to generate flocs for the collection of dissolved pollutants and suspended substances. This technology was independently rediscovered in the 1950s by Lauderdale and Emmons (1951), who used steel wool to generate iron oxides for the removal of radionuclides from drinking water. In other words, before 1990, Fe0 has never been used as a reducing agent under environmentally relevant conditions (pH > 4.5). For lower pH values, the solubility of iron is higher and the Fe0 surface is accessible to dissolved species, but such conditions rarely occur in nature (Ghauch 2015). It is the pH range of cementation (Noubactep 2010a, b), which is a clearly distinct process. Cementation using Fe0 is a heterogeneous process in which dissolved metallic ions (e.g., Cu2+) are reduced to an element (e.g., Cu0) at the Fe0 surface (Khudenko 1991). Section 2 has already recalled that no single species other than H+ can initiate iron corrosion under environmental conditions. This was the starting point of our research group to establish the science of the Fe0/H2O system. Recent bibliometric analysis of “Fe0 research for environmental remediation from 2000 to 2019” suggests that our research group is the most prolific with respect to research on Fe0/H2O systems (Chen et al. 2019; Li et al. 2021). Table 1 summarizes the milestones of our contribution to establish the renewed aspects of Fe0/H2O systems.
Table 1 shows that our research group has presented a different path to systematically investigate the Fe0/H2O system. The focus was not on individual contaminants (e.g., biological, inorganic, and organic) and the extent of their removal. Rather, the time-dependent changes of the extent of the decontamination capacity of the systems were characterized (Noubactep 2014). Moreover, batch experiments were mostly performed under quiescent conditions and lasted for up to four months. Because only some two or three research groups had adopted quiescent conditions, results are not really comparable to other published data and no profound discussion was possible. A key aspect was the development of the methylene blue method (MB method) which is the sole available tool to characterize the dynamics within the Fe0/H2O system without relying on a specific contaminant (Btatkeu-K et al. 2016; Konadu-Amoah et al. 2022). The MB method assesses the availability of iron corrosion products (FeCPs), or the extent of iron corrosion as influenced by selected operational parameters. For example, for column studies, the time to contaminant breakthrough has never been a key evaluation parameter. Rather, systems were designed for rapid breakthrough such that their behavior after breakthrough can be characterized for the longest experimental duration (Miyajima and Noubactep 2013; Konadu-Amoah et al. 2022).
Before 2007, tools to characterize the Fe0 intrinsic reactivity were already presented as summarized in one of the papers (Btatkeu-K et al. 2016). It was also clear to us that there is no quantitative contaminant removal in Fe0/H2O systems until enough oxides are precipitated. Given that even recent review articles are still presenting Fe0 as a (strong) reducing agent (Huang et al. 2021a, b), it is fair to state that collaborators within the research community still fail to understand the fundamentals of iron corrosion under aqueous conditions. For sure, this includes all actors: researchers, reviewers, and editors. While this may sound harsh, it is critical that past errors are corrected to ensure that the industry of water treatment using Fe0 cannot continue to deny its flaws, while expecting interested funders and investors to commit resources to fund research on Fe0/H2O systems. In summary, researchers are still using a paradigm that attempts to violate the first law of thermodynamics (conservation of mass).
The real tragedy of the Fe0 remediation technology has been a wrong system analysis (Noubactep 2009a), as demonstrated by the electron efficiency concept. The expression “electron efficiency” was introduced in an effort to avoid Fe0 wastage by optimizing the proportion of electrons from Fe0 used for the reductive transformation of a target contaminant (e.g., RCl) (Hu et al. 2021b). Regarding RCl, the electron efficiency characterizes the relative quantity of electrons transferred from Fe0 to RCl, where relevant competing electron receptors are co-contaminants (e.g., NO3–), dissolved O2, and water. Before the advent of the Fe0 remediation technology, however, it was already clear that no electron transfer to NO3–, O2, and RCl is possible (Sect. 2). The fallacy of the electron efficiency concept was demonstrated in a recent paper (Hu et al. 2021b).
A good literature review (Sect. 2), coupled with a proper system analysis restores the scientific truth. The three major mistakes as extracted from Huang et al. (2021a, b) are:
-
1.
Acknowledging the non-stoichiometric nature of decontamination reactions while using pseudo-first-order rate constants (kobs, kM, kSA) or the corresponding half-lives (t1/2) to describe the systems.
-
2.
Considering that any Fe0/H2O interface exists, including within pits, crevices, or equivalent defects in the oxide scale on Fe0. Direct electron transfer from Fe0 to contaminants (electrochemical reduction) supposedly occurs at such sites. Because the formation of an oxide scale is spontaneous, it occurs also at defects such that there is always a Fe0/oxides interface. Note that the oxide scale acts as a conductive barrier for electrons from Fe0 and a diffusion barrier for contaminants. Thus, contaminants can accumulate at the oxides/H2O interface.
-
3.
Improperly considering the kinetics of iron corrosion which is non-constant, and nonlinearly varies with the time (Nesic 2007). While the dynamics of the Fe0/H2O system has been often addressed, very few experiments have lasted for more than three months (Westerhoff and James 2003; Jha and Bose 2005; Yamashita and Yamamoto-Ikemoto 2008, 2014; Zheng et al. 2019), meaning that available data just reflect the initial corrosion rate yet they are used to design and model field applications and long-term performance (Jeen et al. 2009).
-
4.
A typical feature of a redox reaction is an exchange of electrons between at least two reaction partners; oxidation and reduction always occur simultaneously. Table 2 illustrates this for the Daniell cell which involves Cu2+ reduction by Zn0. For the Fe0 remediation technology, the contaminants are the oxidizing agent while Fe0 is the reducing agent. Thus, contaminant and Fe0 together form the corresponding redox pair. The question arises: Is contaminant reduction a chemical (electrons from FeII or H species) or an electrochemical reaction (electrons from Fe0)? Both types of reactions are possible because of the negative value of the Gibbs energy (ΔG0 < 0). In other words, the thermodynamics alone cannot help to disclose the real mechanism of contaminant reductive degradation in Fe0/H2O systems. Chemical reduction requires that the reaction partners come close together (< 4 Å or 0.4 nm). On the other hand, electrochemical reactions occur between distant partners under the prerequisite that they are both electronically and ionically conductive and coupled together (Table 2). Clearly, for a contaminant to be electrochemically reduced by Fe0, electrons should flow from Fe0 to the contaminant. Unfortunately, the Fe0 surface is permanently covered by a non-conductive oxide scale (Lee et al. 2004; Nesic 2007; Gheju 2018; Hu et al. 2020) and no electrochemical interaction with any contaminant is possible (Hu et al. 2021b).
The future of Fe0-based filtration systems
The view that Fe0 is a reducing agent for contaminant reduction under environmental conditions is faulty as demonstrated in Sects. 2 and 3. The main problem of Fe0 filters is the time-dependent decrease in its permeability (Lauderdale and Emmons 1951; Antia 2020). There are many contributing factors to permeability loss, but the most important one is the volumetric expansive nature of iron corrosion. Volumetric expansion implies that each oxide is at least twice larger in volume than the parent metal (Voxide > 2 Viron). The question arises: How much oxide is generated in a Fe0 filter per unit time? Answering this question requires that the corrosion rate of the used Fe0 specimens is known. However, this is not the case, and too little efforts have been directed at characterizing the reactivity of Fe0 materials (Li et al. 2019; Lufingo et al. 2019). This is very unfortunate in a context where it is well-documented that the Fe0 corrosion rate can vary over several orders of magnitude (Melchers and Petersen 2018). Equations 1 and 4 demonstrate that twice more Fe0 is consumed for the reduction of RCl than it would have been in an electrochemical reaction (Eq. 1). Given the low solubility of FeII and FeIII in water at pH above 4.5, this implies at least twice more oxides to fill the pores and induce permeability loss as has been assumed until now. When finally considering that only protons (H+) oxidize Fe0, it becomes clear that pore filling by iron corrosion products must be considered before any other operating factors (e.g., foreign precipitates; suspended particles; and even H2 generation). In other words, despite three decades of intensive research efforts to characterize the long-term permeability of Fe0 filters, satisfactorily results are lacking because the system analysis underlying past efforts was not holistic. For instance, in the majority of the studies, the Fe0 materials used were only poorly or never characterized with respect to their intrinsic reactivity (e.g., long-term corrosion rate). In other studies, high agitation rates that are not representative of operating conditions of the Fe0 remediation systems were used (Lee et al. 2004; Gheju and Balcu 2011; Ghauch 2015). Thus, a systematic comparison of results among studies is problematic.
It may be surprising to read that investigating the contribution of expansive iron corrosion to the process of permeability loss in Fe0 permeable reactive barriers (Fe0 PRBs) is an innovation. This becomes obvious, however, when it is recognized that no single contaminant can oxidize Fe0, while current models for predicting the operation of Fe0 PRBs are mostly based on the stoichiometry of an electrochemical reaction between Fe0 and the contaminants of concern (Sarr 2001; Lee et al. 2004; Santisukkasaem and Das 2019). Thus, such models are premised on the wrong scientific principle. For more than a decade, admixing sand or non-expansive aggregates with Fe0 was considered as “material dilution” with possible negative impacts on the decontamination process (Bi et al. 2009; Konadu-Amoah et al. 2022). Around 2010, it was proved that only hybrid systems (e.g., Fe0/sand) can be sustainable, making “material dilution” rather a prerequisite for sustainability (Domga et al. 2015). In other words, Fe0 filtration systems are yet to be properly considered as a special case of “electrochemical dissolution of metals in porous media.” In such porous systems, the corrosion rate also depends on the pore size distribution (Bi et al. 2009).
The last important feature to be considered for the next generation Fe0 PRBs is the validity of the design prerequisite that PRBs must have a higher hydraulic conductivity than the surrounding aquifer (Lauderdale and Emmons 1951; Gillham 2008). It is certain that the hydraulic conductivity of any Fe0 PRB will decrease with time (Stefanoni et al. 2019). This raises the question, what will happen when the wall and the surrounding porous environment have the same permeability? To the best of the authors’ knowledge, this issue has never been addressed in the Fe0 remediation literature.
A call to change course
The credibility of Fe0-based remediation system as a technology depends essentially on the quality of the work that researchers produce. The fact that a distortion of corrosion science has been prevailing in the Fe0 literature for three decades must be a concern to the scientific community. During the last decade, the situation has worsened to the extent that journal editors and grant referees rejected manuscripts or proposals questioning the current paradigm with the summary comment “not relevant” or “lack of novelty.” For manuscripts, the authors can migrate from one journal to another until they find more understanding and open-minded editors and reviewers. For grant proposals, however, there is almost no such chance, because the “best experts” or grant reviewers could be the same scientists who introduced and perpetuated the named mistakes. In a context of stiff competition for grant money, a single negative referee is often enough for a proposal rejection. Therefore, researchers or research groups submitting novel findings or proposals anchored on the fundamental principles of iron corrosion science either have to: (1) find alternative publication outlets or funding agencies (i.e., flee), or (2) conform to the current flawed or erroneous concepts (i.e., adapt). This is a fundamental problem which cannot be resolved by a few individuals (Tien 2007; Hu et al. 2021b; Konadu-Amoah et al. 2022). By making the problem better known to the public and the research community, the present authors hope to contribute to its solution. It is also our proposal that journal editorial boards and reviewers should avoid further acceptance of manuscripts further perpetuating or propagating the fundamental mistake highlighted here. This approach could redeem the Fe0 remediation technology from its current conceptual errors (Hu and Noubactep 2018, 2019; Hu et al. 2018, 2019; 2020; Noubactep 2022). In fact, that was the motivation for writing this short communication. It should be recalled that good results have refuted the reductive transformation concept in its initial phase (from 1994 to 2001) (Lipczynska-Kochany et al. 1994; Qiu et al. 2000; Lavine et al. 2001; Lee et al. 2004), but were collectively ignored. In particular, Lavine et al. (2001) used differential pulse polarography to simultaneously monitor the disappearance of nitrobenzene and the appearance of Fe2+. Their results could not confirm any electrochemical mechanism for the observed nitrobenzene reduction in the investigated Fe0/H2O system. Lavine et al.’s (2001) study was published in Microchemical Journal (Elsevier) which is mainly devoted to analytical chemistry. Yet 21 years later, this excellent article has been referenced only some 131 times according to Google Scholar (scholar.google.com, accessed: 13/03/2022). Clearly, Lavine et al.’s (2001) study has not been really considered within the Fe0 remediation literature. Several other thought-provoking articles have experienced the same fate (Lipczynska-Kochany et al. 1994; Farrell et al. 1999; Furukawa et al. 2002; Purenović et al. 2004; Lee et al. 2004; Jiao et al. 2009; Noubactep 2010a, b; Giles et al. 2011; Gheju and Balcu 2011, Fudge 2014). In particular, Furukawa et al. (2002) suggested that the abundance of FeCPs in Fe0/H2O systems implies their systematic consideration in all mechanistic discussions. On the other hand, Jiao et al. (2009) traceably demonstrated that the reductive transformation of carbon tetrachloride (CCl4) in the presence of Fe0 was the result of chemical interactions of CCl4 with primary iron corrosion products (e.g., H2 and Fe2+).
The way forward is to consider that the following statement has never been proven false “In 1888, Crum Brown pointed out that iron remained free from rust in an atmosphere of oxygen, carbon dioxide, and water vapor so long as liquid water was prevented from condensing on its surface” (Friend 1906). Moreover, this knowledge is yet to be properly considered in the Fe0 remediation literature where Fe0 is constantly immersed, meaning that less O2 and CO2 are available compared to atmospheric conditions.
Questioning the prevailing flawed concept
The present work is a further attempt to question the premise that “Fe0-induced contaminant removal is initiated by the direct electron transfer from Fe0 to substrates” (Kang and Choi 2009). The named premise was proved inconsistent many years ago by Noubactep (2007, 2008), but this has been largely ignored by active researchers while journal and grant referees are accusing the authors for citing only their own papers to support the new concept. Kang and Choi (2009) anticipated resistance for the new concept using the following wording “this argument is hardly acceptable since the role of the direct electron transfer in Fe0-mediated reactions is well-established and generally accepted among the research community.” It seems that there is no other way to demonstrate the fallacy of the reductive transformation concept than to question the experimental design of Matheson and Tratnyek (1994) and its ability to support their conclusions.
Matheson and Tratnyek (1994) recognized that determining the relative importance of three reductive mechanisms is essential to predict the performance of related remediation systems: (1) reduction by electrons from Fe0 (direct reduction), (2) reduction by electrons from FeII species (indirect reduction), and (3) reduction by electrons from H2 (indirect reduction). Unfortunately, already in their theoretical analysis, Matheson and Tratnyek (1994) had under-estimated or overlooked the importance of indirect reduction because there was a need for an effective catalyst to support reduction through FeII and H2. The same authors further considered that Fe0 is oxidized by dissolved O2 under oxic conditions and by H2O under anoxic conditions. Yet it was known that even under oxic conditions, O2 cannot diffuse to the Fe0 surface and is oxidized within the oxide scale by FeII species (Stratmann and Müller 1994).
Other inconsistencies of the demonstration by Matheson and Tratnyek (1994) include the following considerations: (1) that water acts as proton donor to enable the reduction of RCl, (2) that because the addition of external Fe2+ or H2 had not significantly affected the rate of CCl4 dehalogenation under their experimental condition, indirect reduction can be ruled out, and (3) that complexing FeII with ethylenediaminetetraacetic acid (EDTA) would impact CCl4 reduction by Fe0. EDTA is a ligand which forms very stable complexes with FeIII, meaning that a proper discussion should consider the electrode potential of the redox couple FeIIIEDTA/FeIIEDTA. All these important aspects were not considered by Matheson and Tratnyek (1994) in the mechanistic discussion of their findings.
The development of concepts for understanding processes leading to decontamination in Fe0/H2O systems requires a careful analysis of all the contributing physicochemical processes (Lee et al. 2004; Noubactep 2009a; Taylor and Ke 2021; Hu et al. 2021b). Relevant processes include: (1) mass transport from the polluted water to the Fe0 vicinity, (2) chemical and electrochemical reactions at the Fe0/oxide interface, within the oxide scale, and at the oxide/H2O interface, (3) compositional and microstructural changes in Fe0 and the in-situ generated and transforming oxide scale, and (4) the dynamic interactions of solutes, including pollutants with the oxide scale (Nesic 2007; Wen et al. 2012; Gheju 2018; Barrera et al. 2018; Hu et al. 2021b; Taylor and Ke 2021). These processes are highly dynamic, while some maybe close to equilibrium, and heavily dependent upon environmental variables such as hydrodynamic conditions, pH value, the presence of impurities in the used Fe0 material, and the ambient temperature. Clearly, designing laboratory experiments to support field installations of Fe0 PRBs is a very challenging task that shall not be further complicated by avoidable biases (Lee et al. 2004; Hu et al. 2021b). In particular, mixing conditions (mass transfer rate) used in the large majority of batch experiments in treatability studies influence mass transfer in a manner that is not representative of field Fe0 PRBs (Lee et al. 2004; Noubactep 2008; Noubactep et al. 2012). The existence of such a fundamental vagueness imposes difficulties in determining an appropriate Fe0 amount and a thickness for the PRB (Sarr 2001).
Finally, Table 3 summarizes and illustrates four key perceptions in favor of the reductive transformation concept and the corresponding disproving counter-arguments. Table 3 is also regarded as the summary of this communication.
Conclusions
This communication recalls that under environmental conditions the following occur in the Fe0/H2O system:
-
1.
Fe0 is oxidized by water and by water alone,
-
2.
Fe2+, H2, and other species like green rust or Fe3O4 reduce contaminants (and O2),
-
3.
Twice more Fe0 is needed to produce the number of electrons for the reduction by Fe2+ compared to the case where Fe0 would have been the reducing agent, and
-
4.
Because of the very low solubility of FeII and FeIII species, iron oxides and hydroxides must precipitate close to the Fe0 surface and cannot migrate far. Iron oxides are larger in volume than Fe0 and will progressively fill the pores. For these reasons, expansive iron corrosion should be considered first when discussing the process of permeability loss.
All these aspects were strongly neglected in the Fe0 remediation research in the past three decades.
A survey of publications on “groundwater remediation” (Chen et al. 2019) and “remediation using Fe0” (Thakur et al. 2020) shows increasing publication numbers. This confirms the importance of the subject of study and its relevance to industrial technology. The impressive publication record should have made any member of the community proud in this robust state of affairs (Tien 2007). However, the evidence that the large majority of these publications are propagating a mistake and that a circular reasoning is deveoped in the community is very disappointing. The present communication is a call for the Fe0 remediation research community to look critically at their past and future results, and then embark a new path anchored on well-established scientific principles (i.e., aqueous corrosion of Fe0, iron corrosion in porous media).
References
Antia DDJ (2020) Water treatment and desalination using the eco-materials n-Fe0 (ZVI); n-Fe3O4; n-FexOyHz[mH2O]; and n-Fex[Cation]nOyHz[Anion]m [rH2O]. In: Kharissova OV et al (ed) Handbook of nanomaterials and nanocomposites for energy and environmental applications. Springer Nature, Switzerland
Baker MN (1934) Sketch of the history of water treatment. J Am Water Works Assoc 26:902–938
Baker MN (1948) The quest for pure water: The history of water purification from the earliest records to the twentieth century. American Water Works Association, New York, p 527
Barrera O, Bombac D, Chen Y, Daff TD, Galindo-Nava E, Gong P, Haley D, Horton R, Katzarov I, Kermode JR, Liverani C (2018) Understanding and mitigating hydrogen embrittlement of steels: a review of experimental, modelling and design progress from atomistic to continuum. J Mater Sci 53:6251–6290
Bi E, Devlin JF, Huang B (2009) Effects of mixing granular iron with sand on the kinetics of trichloroethylene reduction. Groundw Monit Remediat 29:56–62
Bigg T, Judd SJ (2000) Zero-valent iron for water treatment. Environ Technol 21:661–670
Blowes DW, Ptacek CJ, Benner SG, McRae CW, Bennett TA, Puls RW (2000) Treatment of inorganic contaminants using permeable reactive barriers. J Contam Hydrol 45:123–137
Btatkeu-K BD, Olvera-Vargas H, Tchatchueng JB, Noubactep C, Caré S (2014) Characterizing the impact of MnO2 on the efficiency of Fe0-based filtration systems. Chem Eng J 250:416–422
Btatkeu-K BD, Tchatchueng JB, Noubactep C, Caré S (2016) Designing metallic iron based water filters: light from methylene blue discoloration. J Environ Manag 166:567–573
Cai S, Chen B, Qiu X, Li J, Tratnyek PG, He F (2021) Sulfidation of zero-valent iron by direct reaction with elemental sulfur in water: efficiencies, mechanism, and dechlorination of trichloroethylene. Environ Sci Technol 55:645–654
Cao V, Alyoussef G, Gatcha-Bandjun N, Gwenzi W, Noubactep C (2021) Characterizing the impact of MnO2 addition on the efficiency of Fe0/H2O systems. Sci Rep 11:1–12
Caré S, Crane R, Calabrò PS, Ghauch A, Temgoua E, Noubactep C (2013) Modeling the permeability loss of metallic iron water filtration systems. Clean Soil Air Water 41:275–282
Chamberlin TC (1897) The method of multiple working hypotheses. J Geol 5:837–848
Chen Q, Fan G, Na W, Liu J, Cui J, Li H (2019) Past; present; and future of groundwater remediation research: a scientometric analysis. Int J Environ Res Public Health 16:3975
Courcelles B (2015) Guidelines for preliminary design of funnel-and-gate reactive barriers. Int J Environ Pollut Remed 3:1–11
Domga R, Togue-Kamga F, Noubactep C, Tchatchueng JB (2015) Discussing porosity loss of Fe0 packed water filters at ground level. Chem Eng J 263:127–134
Farrell J, Bostick WD, Jarabek RJ, Fiedor JN (1999) Uranium removal from ground water using zero valent iron media. Ground Water 34:618–624
Friend JN (1906) The rusting of iron. Nature 74:540
Fudge DS (2014) Fifty years of J. R. Platt’s strong inference. J Exper Biol 217:1202–1204
Furukawa Y, Kim JW, Watkins J, Wilkin RT (2002) Formation of ferrihydrite and associated iron corrosion products in permeable reactive barriers of zero-valent iron. Environ Sci Technol 36:5469–5475
Ghauch A (2015) Iron-based metallic systems: an excellent choice for sustainable water treatment. Freiberg Online Geosci 32:1–80
Gheju M (2018) Progress in understanding the mechanism of CrVI Removal in Fe0-based filtration systems. Water 10:651
Gheju M, Balcu I (2011) Removal of chromium from Cr(VI) polluted wastewaters by reduction with scrap iron and subsequent precipitation of resulted cations. J Hazard Mater 196:131–138
Giles DE, Mohapatra M, Issa TB, Anand S, Singh P (2011) Iron and aluminium based adsorption strategies for removing arsenic from water. J Environ Manage 92:3011–3022
Gillham RW (2008), Development of the granular iron permeable reactive barrier technology (good science or good fortune). In: Chen Y, Tang X, Zhan L (eds) Advances in environmental geotechnics: proceedings of the international symposium on geoenvironmental engineering in Hangzhou; China; September 8–10; 2007. Springer Berlin, pp 5–15
Henderson AD, Demond AH (2007) Long-term performance of zero-valent iron permeable reactive barriers: a critical review. Environ Eng Sci 30:401–423
Hu R, Noubactep C (2018) Iron corrosion: scientific heritage in jeopardy. Sustainability 10:4138
Hu R, Noubactep R (2019) Redirecting research on Fe0 for environmental remediation: the search for synergy. Int J Environ Res Public Health 16:4465
Hu R, Cui X, Gwenzi W, Wu S, Noubactep C (2018) Fe0/H2O systems for environmental remediation: the scientific history and future research directions. Water 10:1739
Hu R, Gwenzi W, Sipowo-Tala VR, Noubactep C (2019) Water treatment using metallic iron: a tutorial review. Processes 7:622
Hu R, Yang H, Tao R, Cui X, Xiao M, Amoah BK, Cao V, Lufingo M, Soppa-Sangue NP, Ndé-Tchoupé AI, Gatcha-Bandjun N (2020) Metallic iron for environmental remediation: starting an overdue progress in knowledge. Water 12:641
Hu R, Cui X, Xiao M, Gwenzi W, Noubactep C (2021a) Characterizing the impact of pyrite addition on the efficiency of Fe0/H2O systems. Sci Rep 11:2326
Hu R, Ndé-Tchoupé AI, Cao V, Gwenzi W, Noubactep C (2021b) Metallic iron for environmental remediation: the fallacy of the electron efficiency concept. Front Environ Chem 2:677813
Huang J, Jones A, Waite TD, Chen Y, Huang X, Rosso KM, Kappler A, Mansor M, Tratnyek PG, Zhang H (2021a) Fe(II) redox chemistry in the environment. Chem Rev 121:8161–8233
Huang Z, Cao V, Nya EL, Gwenzi W, Noubactep C (2021b) Kanchan arsenic filters and the future of Fe0-based filtration systems for single household drinking water supply. Processes 9:58. https://doi.org/10.3390/pr9010058
Jeen SW, Gillham RW, Gui L (2009) Effects of initial iron corrosion rate on long-term performance of iron permeable reactive barriers: Column experiments and numerical simulation. J Contam Hydrol 103:145–156
Jha D, Bose P (2005) Use of pyrite for pH control during hydrogenotrophic denitrification using metallic iron as the ultimate electron donor. Chemosphere 61:1020–1031
Jiao Y, Qiu C, Huang L, Wu K, Ma H, Chen S, Ma L, Wu D (2009) Reductive dechlorination of carbon tetrachloride by zero-valent iron and related iron corrosion. Appl Catal B Environ 91:434–440
Kang S-H, Choi W (2009) Response to comment on “Oxidative degradation of organic compounds using zero-valent iron in the presence of natural organic matter serving as an electron shuttle.” Environ Sci Technol 43:3966–3967
Khudenko BM (1991) Feasibility evaluation of a novel method for destruction of organics. Water Sci Technol 23:1873–1881
Konadu-Amoah B, Ndé-Tchoupé AI, Hu R, Gwenzi W, Noubactep C (2022) Investigating the Fe0/H2O systems using the methylene blue method: validity, applications and future directions. Chemosphere 291:132913
Lauderdale RA, Emmons AH (1951) A method for decontaminating small volumes of radioactive water. J Am Water Works Assoc 43:327–331
Lavine BK, Auslander G, Ritter J (2001) Polarographic studies of zero valent iron as a reductant for remediation of nitroaromatics in the environment. Microchem J 70:69–83
Lee G, Rho S, Jahng D (2004) Design considerations for groundwater remediation using reduced metals. Korean J Chem Eng 21:621–628
Li J, Dou X, Qin H, Sun Y, Yin D, Guan X (2019) Characterization methods of zerovalent iron for water treatment and remediation. Water Res 148:70–85
Li X, Li Z, Du C, Tian Z, Zhu Q, Li G, Shen Q, Li C, Li J, Li W, Zhao C (2021) Bibliometric analysis of zerovalent iron particles research for environmental remediation from 2000 to 2019. Environ Sci Pollut Res 28:4200–34210
Lipczynska-Kochany E, Harms S, Milburn R, Sprah G, Nadarajah N (1994) Degradation of carbon tetrachloride in the presence of iron and sulphur containing compounds. Chemosphere 29:1477–1489
Liu H, Wang Q, Wang C, Li XZ (2013) Electron efficiency of zero-valent iron for groundwater remediation and wastewater treatment. Chem Eng J 215–216:90–95
Lufingo M, Ndé-Tchoupé AI, Hu R, Njau KN, Noubactep C (2019) A novel and facile method to characterize the suitability of metallic iron for water treatment. Water 11:2465
Matheson LJ, Tratnyek PG (1994) Reductive dehalogenation of chlorinated methanes by iron metal. Environ Sci Technol 28:2045–2053
McMurty DC, Elton RO (1985) New approach to in-situ treatment of contaminated groundwaters. Environ Progr 4:168–170
Melchers RE, Petersen RB (2018) A reinterpretation of the Roman off NBS data for corrosion of steels in soils. Corros Eng Sci Technol 53:131–140
Miyajima K, Noubactep C (2012) Effects of mixing granular iron with sand on the efficiency of methylene blue discoloration. Chem Eng J 200–202:433–438
Miyajima K, Noubactep C (2013) Impact of Fe0 amendment on methylene blue discoloration by sand columns. Chem Eng J 217:310–319. https://doi.org/10.1016/j.cej.2012.11.128
Mwakabona HT, Ndé-Tchoupé AI, Njau KN, Noubactep C, Wydra KD (2017) Metallic iron for safe drinking water provision: Considering a lost knowledge. Water Res 117(2017):127–142
Nesic S (2007) Key issues related to modelling of internal corrosion of oil and gas pipelines—a review. Corros Sci 49:4308–4338
Neumann A, Kaegi R, Voegelin A, Hussam A, Munir AK, Hug SJ (2013) Arsenic removal with composite iron matrix filters in Bangladesh: a field and laboratory study. Environ Sci Technol 47:4544–4554
Njaramba LK, Park JB, Lee CS, Nzioka AM, Kim YJ (2021) Permeable reactive barriers with zero-valent iron and pumice for remediation of groundwater contaminated with multiple heavy metals. Environ Eng Sci 38:245–255
Noubactep C (2007) Processes of contaminant removal in “Fe0–H2O” systems revisited. the importance of co-precipitation. Open Environ Sci 1:9–13
Noubactep C (2008) A critical review on the mechanism of contaminant removal in Fe0–H2O systems. Environ Technol 29:909–920
Noubactep C (2009a) An analysis of the evolution of reactive species in Fe0/H2O systems. J Hazard Mater 168:1626–1631
Noubactep C (2009b) Characterizing the effects of shaking intensity on the kinetics of metallic iron dissolution in EDTA. J Hazard Mater 170:1149–1155
Noubactep C (2010a) Elemental metals for environmental remediation: learning from cementation process. J Hazard Mater 181:1170–1174
Noubactep C (2010b) The fundamental mechanism of aqueous contaminant removal by metallic iron. Water SA 36:663–670
Noubactep C (2011) Aqueous contaminant removal by metallic iron: Is the paradigm shifting? Water SA 37:419–426
Noubactep C (2014) Metallic iron for environmental remediation: a review of reviews. Water Res 85:114–123
Noubactep C (2016) Predicting the hydraulic conductivity of metallic iron filters: modeling gone astray. Water 8:162
Noubactep C (2019) The operating mode of Fe0/H2O systems: Hidden truth or repeated nonsense? Fresenius Environ Bull 28:8328–8330
Noubactep C (2022) Should the term “metallic iron” appear in the title of a research paper? Chemosphere 287:132314
Noubactep C, Caré S (2010) Enhancing sustainability of household water filters by mixing metallic iron with porous materials. Chem Eng J 162:635–642
Noubactep C, Licha T, Scott TB, Fall M, Sauter M (2009) Exploring the influence of operational parameters on the reactivity of elemental iron materials. J Hazard Mater 172:943–951
Noubactep C, Schöner A, Sauter M (2012) Significance of oxide-film in discussing the mechanism of contaminant removal by elemental iron materials. In: "Photo-electrochemistry & photo-biology for the sustainability". Union Press, Osaka, Japan, pp 97–122. ISBN-10: 4946428615; ISBN-13: 978-4946428616
O’Hannesin SF, Gillham RW (1998) Long-term performance of an in situ “iron wall” for remediation of VOCs. Ground Water 36:164–170
Obiri-Nyarko F, Grajales-Mesa SJ, Malina G (2014) An overview of permeable reactive barriers for in situ sustainable groundwater remediation. Chemosphere 111:243–259
Phukan M, Noubactep C, Licha T (2015) Characterizing the ion-selective nature of Fe0-based filters using azo dyes. Chem Eng J 259:481–491
Phukan M, Noubactep C, Licha T (2016) Characterizing the ion-selective nature of Fe0-based filters using three azo dyes in batch systems. J Environ Chem Eng 4:65–72
Purenović M, Perović J, Bojić A, Anđelković T, Bojić D (2004) Cu and Cd removal from wastewater by a microalloyed aluminium composite. Environ Chem Lett 2:59–63
Qiu SR, Lai HF, Roberson MJ, Hunt ML, Amrhein C, Giancarlo LC, Flynn GW, Yarmoff JA (2000) Removal of contaminants from aqueous solution by reaction with iron surfaces. Langmuir 16:2230–2236
Reynolds GW, Hoff JT, Gillham RW (1990) Sampling bias caused by materials used to monitor halocarbons in groundwater. Environ Sci Technol 24:135–142
Santisukkasaem U, Das DB (2019) A non-dimensional analysis of permeability loss in zero-valent iron permeable reactive barrier (PRB). Transp Porous Media 126:139–159
Sarr D (2001) Zero-valent-iron permeable reactive barriers - how long will they last? Remediation 11:1–18
Stefanoni M, Angst UM, Elsener B (2019) Kinetics of electrochemical dissolution of metals in porous media. Nat Mater 18:942–947
Stratmann M, Müller J (1994) The mechanism of the oxygen reduction on rust-covered metal substrates. Corros Sci 36:327–359
Tao R, Yang H, Cui X, Xiao M, Gatcha-Bandjun N, Kenmogne-Tchidjo JF, Lufingo M, Konadu Amoah B, Tepong-Tsindé R, Ndé-Tchoupé AI, Touomo-Wouafo M (2022) The suitability of hybrid Fe0/aggregate filtration systems for water treatment. Water 14:260
Taylor CD, Ke H (2021) Investigations of the intrinsic corrosion and hydrogen susceptibility of metals and alloys using density functional theory. Corros Rev 39:177–209
Thakur AK, Vithanage M, Das DB, Kumar M (2020) A review on design; material selection; mechanism; and modelling of permeable reactive barrier for community-scale groundwater treatment. Environ Technol Innov 19:100917
Thiruvenkatachari R, Vigneswaran S, Naidu R (2008) Permeable reactive barrier for groundwater remediation. J Ind Eng Chem 14:145–156
Tien C (2007) Remarks on adsorption manuscripts revised and declined: an editorial. Sep Purif Technol 54:277–278
Tucker WG (1892) The purification of water by chemical treatment. Science 20:34–38
Van Craenenbroeck W (1998) Easton & Anderson and the water supply of Antwerp (Belgium). Ind Archaeol Rev 20:105–116
Warner SD (2015) Two decades of application of permeable reactive barriers to groundwater remediation. In: Naidu R, Birke V (eds) Permeable reactive barrier sustainable groundwater remediation. CRC Press, Boca Raton, pp 25–39
Weber EJ (1996) Iron-mediated reductive transformations: investigation of reaction mechanism. Environ Sci Technol 30:716–719. https://doi.org/10.1021/es9505210
Wen YH, Chen LQ, Hawk JA (2012) Phase-field modeling of corrosion kinetics under dual oxidants. Model Simulat Mater Sci Eng 20:035013
Westerhoff P, James J (2003) Nitrate removal in zero-valent iron packed columns. Water Res 37:1818–1830
Whitney WR (1903) The corrosion of iron. J Am Chem Soc 25:394–406
Xiao M, Cui X, Hu R, Gwenzi W, Noubactep C (2020a) Validating the efficiency of the FeS2 method for elucidating the mechanisms of contaminant removal using Fe0/H2O systems. Processes 8:1162
Xiao M, Hu R, Cui X, Gwenzi W, Noubactep C (2020) Understanding the operating mode of Fe0/Fe-sulfide/H2O systems for water treatment. Processes 8:409
Yamashita T, Yamamoto-Ikemoto R (2008) Phosphate removal and sulfate reduction in a denitrification reactor packed with iron and wood as electron donors. Water Sci Technol 58:1405–1413
Yamashita T, Yamamoto-Ikemoto R (2014) Nitrogen and phosphorus removal from wastewater treatment plant effluent via bacterial sulfate reduction in an anoxic bioreactor packed with wood and iron. Int J Environ Res Public Health 11:9835–9853
Yang H, Hu R, Ruppert H, Noubactep C (2021) Modeling porosity loss in Fe0-based permeable reactive barriers with Faraday’s law. Sci Rep 11:16998
Zheng X, Jin M, Xu H, Chen W, Zhang Y, Yang M, Shao X, Xu Z, Wang W (2019) Enhanced simultaneous nitrogen and phosphorus removal in a denitrifying biological filter using waterworks sludge ceramsite coupled with iron-carbon. Int J Environ Res Public Health 16:2646
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by the Ministry of Science and Technology of China through the Program “Driving process and mechanism of three dimensional spatial distribution of high risk organic pollutants in multi field coupled sites” (Project Code: 2019YFC1804303).
Author information
Authors and Affiliations
Contributions
H.Y., R.T., V.C., B.K.A., and A.I.N.T conceived the presented idea and developed the theory and presented the initial draft. R.H., W.G., H.R., and C.N. supervised this work. W.G. supervised the redaction of the first draft and proofread the final manuscript. All authors contributed to the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Institutional review board statement
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Konadu-Amoah, B., Hu, R., Cao, V. et al. Realizing the potential of metallic iron for the mitigation of toxics: flee or adapt?. Appl Water Sci 12, 217 (2022). https://doi.org/10.1007/s13201-022-01738-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-022-01738-9