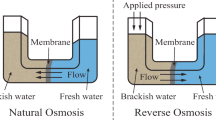
Abstract
Recently, water scarcity, world population rise and urbanization are causing desalting of seawater. Membrane separation processes such as reverse osmosis are applied for desalination over the conventional mass transfer techniques. For instance, reverse osmosis accounts for the treatment of nearly 1% of the world water supply and its applications are increasing with time. However, cost, technical management and efficiency are challenging aspects. Hence, membrane fouling, direct and indirect investment costs, and permeate quality are the major hurdles encountered. Toward solving these problems, various studies are being conducted including varying membrane operating conditions and separation methodologies whereby ion-exchange–reverse osmosis hybrids and the coupling of two or more other membrane filtration techniques can be mentioned. Apparently, most of the hybrid techniques targeted for the selective removal of a pollutant. Hence, ion exchange–reverse osmosis is the most applied hybrid process particularly for boron removal due to its public and environmental health impact. Despite that, there is no current and comprehensive review that bridged the piece by piece and fragmented reports. In this review, desalination–ion-exchange coupling with other membrane filtration is explored in detail and the recent advances in composite membrane material synthesis are uncovered. Further, the status, cost and future potential of hybrid membranes are discussed and gaps are identified for future research.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The water–population–energy nexus is increasingly a global issue for various reasons. Fundamentally, the world population is growing faster alongside urbanization and industrialization and so is water demand, especially in the last 30 years. Population boom is occurring in the developing world lead by China, India, Brazil and most African countries. In spite of the fact that 72% of the planet earth is covered with water, and the fresh and accessible portion is only 0.1% (du Plessis 2017), which is very limited and even it is getting scarcer vis-à-vis the continuing developmental activities that result in elevated water demand, release of huge wastewater and the consequent water pollution problems (Hand and German 2018).
Based on the information in Table 1 and Fig. 1, the total water budget of the earth is estimated to be 1396.5134 billion cubic meter of which almost the 96% is caught up in oceans and it is not fresh for use. Moreover, 30.1% of the freshest water is contained in aquifers, but the aquifers contain only 0.75% of the earth’s total water budget. In that regard, desalination appears as promising alternative for the ever-increasing water demand (El-Dessouky and Ettouney 2002).
Earth’s water sources and its fresh component (Data source:(El-Dessouky and Ettouney 2002))
Table 1 further shows the actual water volume of each reservoir. According to the quantities in the table, rivers are the most limited water resources. However, most process industries are installed adjacent to rivers (Chowdhary et al. 2018) which exacerbate scarcity due to pollution. In spite of the scarcity of freshwater reservoirs, the oceans contain the hugest proportion which is untapped. Consequently, there are opportunities to explore the oceans as source of useful water through desalination. Otherwise, exploring other water-rich planets may be considered as alternatives (Hand and German 2018).
In fact, desalination is applied not only for freshwater supply but also to get desalted water for agriculture, mining, removal/recovery of heavy metals and beyond toward water sustainability. Besides, for the industrial wastewater treatment membrane bioreactors are applied to pulp, metal, cosmetic and other industries whereby a chemical oxygen demand (COD) removal of up to 98% is reported. Further, diverse group of pharmaceutical compounds including persistent organic compounds (POPs), pesticides, pharmaceutically active compounds (PhACs) and endocrine disrupting chemicals (EDCs) removal is achieved with efficiency of > 90%. Further, ceramic membrane materials are being applied to food, municipal and alcohol distillery wastewaters treatment (Le and Nunes 2016).
In this regard, desalination technology is expected to increase freshwater access to the booming world population. Earlier desalination has been a synonym to desalting; however, it is getting broader in meaning and the level of its applications. Desalination is defined as the removal of dissolved salts and minerals in water (Le and Nunes 2016; Wang et al. 2018a) at different pore size/filtering capacity as desired (Fig. 2). Consequently, various thermal, membrane, chemical–physical and hybrid technologies (Fig. 3) are installed, tested and scaled-up as reported by researches in the field.
Schematic representation of various membrane processes, including reverse osmosis, NF, ultrafiltration, microfiltration and traditional particle filtration, in which the membrane pore size grows proportionally (Homaeigohar and Elbahri 2017)
In order to enhance desalination performance, diverse efforts are being applied with time. For instance, the coupling of membrane filtration (MF) techniques such as nanofiltration and reverse osmosis (NF/RO) is largely important to optimize cost and produced water quality whereby ion-exchange (IX)/MF coupling improved product quality and it is applied in the selective removal of particular pollutants (Ang et al. 2015; Esfahani et al. 2016; Talaeipour et al. 2017; Zaib and Fath 2013).
The coupling of IX–membrane technology is a relatively recent technology that has evolved to its large-scale applications such as in electrodialysis, electrodeionization, diffusion dialysis, membrane electrolysis, desalination of seawater and brackish water. Ion exchange is basically the process of replacing same charge ion between the medium and an electrolyte solution in which the medium can be a complex cross-linked polymer matrix that is widely used to produce ultrapure water. Such hybrid techniques are claimed to be energy efficient, quality relevant and waste valorizing tool, playing its role in the context of sustainability (Strathmann et al. 2013; Xu 2005).
Concurrent to the increase in desalinated water supply, the researches on diversification and enhancement of the desalination technology with respect to purpose, geographical conditions and the possible reduction in the associated cost and energy requirements are also advancing with time. As a result, hybrid systems are being investigated; of them is the integration of IX/RO which is less explored compared with other coupling techniques like adsorption, filtration and sedimentation, often to pretreat feed water (Tal 2018).
In this regard, today’s membrane processes brought opportunities in various industrial activities over the conventional mass transfer processes reaching far in uses such as in health sector for developing artificial organs through pollution removal. Membrane process as a separation technology evolved over time since its role as a laboratory tool 40 years back (Strathmann 2001). Despite its development and critical importance, membrane material often results in cationic fouling that is extremely difficult to remove. The problem is due to membrane chemistry; the surface of a polyamide membrane is usually negatively charged. In that regard, little is known or reported about coupling of membrane processes in general and the hybrid of membrane–ion-exchange techniques. Therefore, the objective of this scientific review is to explore the status and potential of hybrid desalination techniques with focus on IX/RO and its future.
Conventional membrane processes versus advances hybrid technology
So far, the conventional technology in desalination is RO accounting for nearly 1% of the world water supply. Following technological advances, use of RO increases with time mainly due to the increased investment and the growing profitability of the service. Optimization studies brought space and capital investment reduction as advantages in desalination over time. By the end of 2015, there were approximately 18,000 desalination plants worldwide, with a total installed capacity of 86.6 million m3/day that is increasing even at a faster rate. Application of RO is geographically uneven, dominated by the Middle East countries including Qatar and Kuwait (Le and Nunes 2016). Though, the hot spots for future desalination application include Asia, the USA, Latin America and North Africa mainly due to population rise and pollution. Along time, the cost of desalinated seawater is dramatically dropping due to the evolving efficient membrane technologies, and the direct and indirect capital cost remains to cover 50% of the overall on average (International Desalination Association 2016) which is consistently driving scientists to look for alternative and feasible hybrid technology.
Despite the ever-increasing application of desalination, the conventional membrane processes including RO are limited by fouling problems which worsens its existing cost implication. During operation, some foulants occur due to the use of metal salts for coagulation during feed water pretreatment like FeCl3. As a good approach to such limitations of membranes and for the selective removal of ions in water or wastewater, IX/MF hybrids came in to effect, to purify both the feed and the permeate (Abdulgader et al. 2013). Further advances use electrodeionization process to produce ultrapure water by applying separate beds of cation- and anion-exchange resins and a bipolar membrane while regenerating protons and hydroxide ions instead of the conventional use of mixed-bed ion exchange resins (Grabowski et al. 2006). Such attempts even lead to the development of a current thermal and mechanical pressure stable hybrid synthesized membranes of new kind (Afsar et al. 2017).
Moreover, ion-exchange membranes got wider applications including its use for dialysis, water and wastewater desalination, chlorine–alkaline production. Recently, these hybrid membranes got their potential application in electrical energy generation (Strathmann et al. 2013). These ion-exchange membranes are characterized based on their electrochemical properties that define the water in the membrane and the feed in addition to the physical membrane properties (Berezina et al. 2008).
Progress in membrane materials and their hybrid in desalination
Amy et al. (2017) highlighted that the existing desalination techniques are in transformation especially with focus on greening of the technology. Despite the huge potential for desalination of seawater resource, conventional techniques are limited by their significant specific energy consumption, membrane fouling, environmental impact and problem with brine disposal. Hence, there are progresses in the conventional desalination processes including modifications like closed-circuit RO, membrane distillation, forward osmosis, pressure retarded osmosis and reverse electrodialysis giving hybrids. Indeed, these developments occurred following advances in material science, process engineering and system integration techniques (Table 2).
An integrated/hybrid membrane system is a process which combines a membrane filtration unit (microfiltration/ultrafiltration/nanofiltration) with other processes such as coagulation, adsorption and ion exchange. It can also be a combination of different membranes in a system of a conventional process (Ang et al. 2015). Further, hybrid membrane desalination techniques are also required to get an enhanced quality of the treated water and reductions in the capital and operating costs of the plants which include to efficiently treat some solutes in water like boron (Zou et al. 2019). For instance, seawater RO permeate can further be treated by hybrid membrane systems and removes over 99% of boron, thereby meeting the desirable limit (5 mg/L) that otherwise could not be achieved by either osmosis or ion exchange alone. In a related study, Bryjak et al. (2008) explored the removal of boron from seawater by adsorption–membrane hybrid process using crushed boron-selective ion-exchange resin and MF in boron concentration reduced from 10 to 2 mg/L (Htun and Maung htun 2011). To know more about boron chemistry, its plant and animal health problems, a review by Wolska Bryjak has got relevant details (Wolska and Bryjak 2013).
Among the developments in material science which has got application through membrane-based desalination is the innovation of nanoparticles and nanocomposites that demonstrated an attractive desalination potential. These developments are being realized through the incorporation of conventional nanomaterials such as metal oxides, silica and zeolites to the conventional membrane systems. Further, the emergence of carbon nanotubes and graphene-family nanomaterials are hoped to contribute to the sustainability of membrane-based desalination technology through their water flux enhancing and exceptional salt rejection property.
Mathematically, water flux across a membrane is inversely proportional to its thickness. Thus, the atomic thinness of graphene (d ≈ 0.34 nm) can lead to larger water permeability than the polyamide active layer in thin-film composite membranes (d ≈ 100 nm). For instance, graphene oxide membrane flux of up to 276 LMH/MPa, with over 99% salt rejection, which is 10 times higher than that of the most commercialized nanofiltration membranes is reported. Consequently, sustainable membranes are envisaged to be produced from advances in engineered nanomaterials including graphene (Goh et al. 2016; Homaeigohar and Elbahri 2017). In addition, Kwon et al. (2010) synthesized a modified membrane called PEGA homopolymer-coated RO membranes which improved flux limitations by 100%.
In this regard, monolayer graphene membrane is produced and tested for separation efficiency (Fig. 4). According to Homaeigohar and Elbahri, the separation ability of the graphene membrane was examined using a forward osmosis process with different solutes, such as NaCl, MgSO4, Allura Red and dextran. The rejection efficiency of the membrane was as follows: ~ 70% of MgSO4, ~ 90% of Allura Red and ~ 83% of dextran while the NaCl showed a negative rejection (Homaeigohar and Elbahri 2017).
Schematic illustrating the process for the inclusion of controlled pores in a monolayer graphene membrane by ion bombardment followed by chemical oxidation (Homaeigohar and Elbahri 2017)
Aside from the polymeric membranes, inorganic membrane science and technology is another attraction in applied water science, water treatment and desalination. Despite the high synthesis cost, the desirability of membranes such as various ceramic membranes, carbon-based membranes, silica membranes and zeolite membranes is mainly for its resisting harsh chemical cleaning, high temperature and wear resistance, high chemical stability, long lifetime and autoclavable advantage (Fig. 5). (Kayvani Fard et al. 2018).
Scanning electron microscopy image of a porous, b dense, c asymmetric and d symmetric inorganic membrane (Kayvani Fard et al. 2018)
Regarding the efficiency of ceramic membranes in desalination, it is well reported in a range of values depending on the constituent materials of its fabrication. Table 3 summarizes produced water (PW), water that is produced as a by-product along with the oil and gas, treatment using various ceramic membranes whereby supported and unsupported Al2O3 ceramic is efficient. However, the flux advantage of the supported aluminum oxide ceramic is much flexible and it operates relatively at highest flux. In a related fact, zeolite membranes are also reported to show better removal/rejection capacity compared to polymer membranes (Kayvani Fard et al. 2018).
Yet, researches are in progress to advance membrane materials to fit to the existing challenges. Most of the researches are geared toward cost minimization, making it more environmentally friendly and efficient. Thus, surface coating on the active layer of an existing membrane, changing the synthesis material, enhancing antifouling and elevating surface hydrophilicity are some of the modifications to membranes (Zou et al. 2019). The inadequate rejection of ions and small neutral solutes by polyamide RO membranes necessitated extensive post-treatment. In a recent marvelous work, Werber and Elimelech (2018) reported defect-free biomimetic membranes that would have water/salt permselectivities of ~ 108-fold greater than current desalination membranes (Werber and Elimelech 2018).
Ion-exchange–membrane coupled technology configuration
Based on the literature, types of membrane fouling vary depending on the source of feed water (Sachit and Veenstra 2017). The categories of foulants so far reported include colloids, inorganic particles, biological as well as other chemical species like iron, calcium and manganese, oil, and natural organic matter (NOM), whose effect depends on the pH and electron potential of feed. Thus, such raw water properties rose the interest on the mode of coupling. In fact, coupling of IX/RO is not such recent technology; there are patents as old as over 4 decades that applied one or more ion-exchange resins followed by RO to desalt brackish water (Bresler 1972). Similarly, IX followed by NF is patented in 2008 (Sengupta and Sarkar 2008).
IX can be coupled with MFs in order to remove selectively a particular pollutant or just to produce ultrapure water as a product. Exchangeable ions in IX are applied as either anionic exchange resin (AXR) or cationic exchange resin (CXR) on an inert support material which preferable needs to be regenerable. The mode of integration of an IX can either be pre- or post-MFs as shown in Fig. 6 depending on the particular purpose for specified rejections. In this regard, RO, ultrafiltration (UF) or electrodialysis is typical integrable. In IX/RO integration, for instance, removal of boron from permeate and removal of salts after coagulation flocculation process for feeding RO have been applied (Eumine Suk and Matsuura 2006; Grabowski et al. 2006; Strathmann et al. 2013).
Continuous geometries of IXRs can be applied to pretreat water before membrane desalination. However, the most established industrial scale is the removal boron from RO permeate. In relation to that the regeneration of resin is relevant in coupling IX with membranes for desalination. In this regard, the type of resin used determines the regeneration potential of the resin. For instance, strong acid cation-exchange resin and weak acid cation-exchange resin showed regeneration efficiency of 33% and 100%, respectively (Bornak 2014).
Application maturity and status of the hybrid desalination technology with emphasis to boron removal
Generally, the intensity and selectivity of a separation process is influenced by the type and specification of the product required to fit to the end user. In water treatment, advanced technologies are sought over the conventional separation technologies so as to meet the desired quality of the product or to selectively remove trace pollutants from the feed water. For instance, the concentrations of boron and arsenic in geothermal water are much higher than the permissible limits for drinking purpose which required subsequent treatment to avoid the associated toxicity problem to consumers. According to Kabay et al. (2013), the concentration of boron in the geothermal water permeate could be lowered to the level below to 1.0 mg/L in 15 min by keeping the concentration of ion exchange resin in the suspension at 3 g resin L−1 of geothermal water. In addition to that, sorbents include zeolites also showed arsenic removal from geothermal water (Kabay et al. 2013).
Furthermore, emerging pollutants are not easily removed by the conventional wastewater treatment processes which include phenols and other pharmaceutical as well as cosmetic ingredients that are released to the wastewaters which ultimately end up in natural waters. These recalcitrant pollutants are increasingly in use either as therapeutic or personal care products such as antiepileptics (carbamazepine), tranquillizers (diazepam), analgesics (ibuprofen, naproxen, diclofenac), antibiotics (roxithromycin, erythromycin, sulfamethoxazole, trimethoprim) and three polycyclic musk fragrances (galaxolide, tonalide, celestolide) (Reif et al. 2008).
In view of that, there is a pressing need for developing novel technologies that can remove existing or emerging pollutants in water or wastewater and that are able to meet the stringent environmental discharge limits or the tightening water quality regulations. For instance, the selective removal of boron that usually occurs as boric acid H3BO3 cannot be removed by the conventional water treatment systems. As a result, the need for the application of hybrid systems is on the rise by both the industry and academia (Eumine Suk and Matsuura 2006). Consequently, there are numerous studies that evaluate either the performance or the cost of a particular membrane technique.
For instance, a recent review by Wang et al. (2018b) reported that a nanoporous material synthesized by phase inversion method from poly (acrylic acid)-grafted with polyvinylidene fluoride has efficiently (> 99.99%) removed hexadecane, toluene and diesel with a water flux of 15,500–23,200 L/m2 h bar. A related work by Miyoshi et al. (2018) reported a huge color reduction in surface water using MF-IX hybrid systems following physicochemical pretreatment. These researchers showed a reduction in surface water color to 0.05 mg/L on a Pt–Co scale (Miyoshi et al. 2018). Regarding the enhancement of transport or flux in desalination, Humplik et al. (2011) reviewed materials including zeolites, carbon nanotubes and graphene with potential applications to reverse osmosis, capacitive deionization and multistage flash, with respect to their cost and energy advantages (Humplik et al. 2011).
Choi et al. (2017) tested the coupled effect of electrocoagulation (EC) and ion concentration polarization (ICP) which is an electromembrane desalination process. By such hybrid laboratory-scale experiment, they demonstrated that continuous EC–ICP operation removed > 95% of suspended solids and reduced the salinity from brackish range (20 mM NaCl) to a potable level (< 8.6 mM NaCl) (Choi et al. 2017).
An earlier study by Taniguchi et al. (2004) proposed new seawater RO membrane by improving control between the membrane surface and boron and due to tight function layer to enhance the size exclusion performance. This new SWRO membrane, named TM820A, is believed to reduce the production cost and was able to reject 94–96% of boron from seawater (Taniguchi et al. 2004).
Abdulgader et al. (2013) conducted a review on the application of IX/MFs few years back. These scholars argued that the ion-exchange resin (IXR) coupled with RO and UF membranes showed a synergistic effect in removing trace pollutants in water through improving efficiency and operational costs. Further the authors disclosed that RO/IXR could be regarded as standard boron removal integration to RO permeate in seawater desalination. Though they directed at further research especially on robustness and feasibility of such technique, they declared the promising future of such integration for reduction in desalination costs and membrane scaling (Abdulgader et al. 2013).
Alharati et al. (2017) achieved the removal of boron to a level of 0.3 mg/L by combining IX pretreatment with NF on a batch experiment treating natural groundwater (Alharati et al. 2017). Advancing the earlier studied facts that integrating IX with membrane process can remove boron, these investigators went on to see the effect of applying sorption on IXR in batch and microfiltration on the frequency of resin application using model boron solution which is an issue in coupling IX/RO (Table 4).
Compared to use of UF alone which remove only 3% dissolved organic carbon (DOC), the AXR/UF hybrid method removes up to 80% minimizing the risk of irreversible membrane fouling by 15–20% (Table 4) (Cornelissen et al. 2009; Humbert et al. 2007, 2012; Ochoa et al. 2006). Further, an interesting result claimed by Venkatesan and Wankat (2011) states that IX/RO operation at higher recoveries decreased the cost of product water by 75% when brine disposal costs are high and by 20% when brine disposal costs are low in water softening process (Cappelle and Davis 2016; Venkatesan and Wankat 2011).
In fact, boron separation is not the only focus in desalination. Li et al. (2016) configured a laboratory-scale NF-RO-IX separation technology to remove sulfate and nitrate from a flue gas denitrification wastewater in which they achieved recovery of over 90% nitrate and sulfite (Li et al. 2016). While using NF in the presence of Na2SO4 and NaCl at high concentration, negative chloride rejection is observed which is an opportunity on one side. Thus, NF concentrate has been applied to regenerate an exhausted IX resin that reportedly reduced the energy and cost implications of membrane processes especially that of RO (Hilal et al. 2015).
Kim and Dempsey (2010) tested a nonionic microporous resin DAX-8 (Supelco), and three types of AXRs: a strong base macroporous IRA-958 (Rohm and Haas), a pre-swollen weak base gel resin called DEAE (Whatman International) and a strong base MIEX (magic IXR) resin (Orica Watercare) which they claimed DEAE and MIEX resins managed successfully to remove NOM from the wastewater effluent and prevent fouling (Fig. 7) (Kim and Dempsey 2010).
Fouling resistance of UF a and microfiltration (MF) membranes b during filtration of wastewaters after pretreatment with various IXR (Abdulgader et al. 2013)
In addition to foulant removal, IX/RO coupling is found to reduce operational and investment costs. Further, it is performing better and desirable compared to the double RO pass that takes up cost and also occupies more space as illustrated in Fig. 8 (Kabay et al. 2007; Yilmaz et al. 2006).
Integration of single stage RO and IX for boron removal (Lenntech 2018)
Further, use of the hybrid IX/RO systems has advantages to selectively remove heavy metals. In relation to that, IX/RO hybrid is almost serving as standard process to retain boron from surface water, brackish water and wastewater. Kabay et al. (2006) removed about 80% of boron from geothermal water using IX/RO hybrid systems. Further Kabay et al. (2007) showed the performance of nitrate removal from groundwater using IX/electrodialysis hybrid systems due to water polluted by intense agricultural activities (Kabay et al. 2007). Additionally, Kabay et al. (2013) conducted an experiment to evaluate boron removal from geothermal water, 10.3–11.4 mg/L, by evaluating effect of IX resin replacement with time on boron concentration in the permeate in a IX/UF coupled desalination system and it was possible to reduce the boron in the permeate lower to 1 mg/L using higher amount of resin (Kabay et al. 2013). Besides the effect of amount of resin, the functional group of the resin is a significant factor in IX desalination pretreatment processes together with the feed salt concentration based on laboratory- and pilot-scale studies (Hilal et al. 2015).
Over saturation of inorganic salt results in membrane scale usually deposited in the form of CaCO3, CaSO4_xH2O and Ca3(PO4)2 due to precipitation on its surface that again bring cost implication which together derived the application of coupling other methods like IX with RO or UF as pretreatment over the advantage of other antiscalants. IX post-treatment can also be applied to RO permeate for complete demineralization (Fig. 9). The energy demand of RO, though widely applied for desalination, is still challenging accounting 44% of the total water cost. In that regard, IXR-NF hybrid is a potential technology for seawater desalination (Venkatesan and Wankat 2011).
Future potential and challenges of the hybrid technology
The future of hybrid technology appears promising even beyond desalination. As time goes, ion-exchange membrane process is modifying especially depending on the particular type of pollutant to be removed. A recent study by Siekierka et al. (2017) modified the function of a polyvinyl chloride chromium removing membrane to modify membrane’s ion-exchange capacity, chloride and nitrogen contents, water regain and surface energetics and they brought promising chromium recovery potential (Siekierka et al. 2017).
Alternative to coupling of IX/RO for desalination of brackish water that reaches a concentration of over 35000 ppm of total dissolved solid, there are developments like the application of Ion Concentration Polarization Electrical Desalination that can deal with a concentration level of up to 10% total dissolved solid. These test level study demonstrated not only a reduction in energy but also membrane area consumption. It also showed that this technology is so robust and can treat shale oil/gas development wastes so-called fracking wastewater, salt and even other solids including brine waste with a salt rejection rate of 70% (Kim et al. 2016).
Ochoa et al. (2006) prepared hybrid UF polysulfone (PSf) membranes with resins’ particles deposited in the membranes’ porous structure, and at 20% resin both reversible and irreversible fouling were nearly eliminated (Abdulgader et al. 2013). The PSf-IXR membrane cross section is organized like the PSf- mixed matrix anion-exchange membranes (AEMs) (Fig. 10).
Scanning electron microscope cross sections of polysulfone membrane prepared silica AEMs: a 0%-wt, b 0.5%-wt, c 1.0%-wt and d 1.5%-wt (Khoiruddin and Wenten 2016)
From what has been done so far regarding IX/MF process, the challenge appears to be resin regeneration, process configuration and holistic feasibility of the hybrid technology. Further process optimization is not multivariate in most case, and much of the studies are conducted on a batch system at a laboratory scale. Therefore, multivariate optimization on a continuous flow mode, using regenerable resin is a challenge to be tackled in the future.
Furthermore, Al-Mamun et al. (2018) reviewed the microbial desalination cell technology aiming at reducing the cost of membrane technology with respect to the energy and improving suitability. The review reports that simultaneous electrical energy generation is possible during microbial desalination through bioelectrochemical oxidation of organics present in wastewater. However, practically membrane fouling problem persisted with other technical issues (Al-Mamun et al. 2018). Furthermore, a life cycle assessment of the microbial desalination cell technology argued the high environmental impact of the technology particularly due to its global warming potential (Yang et al. 2019; Zhang et al. 2018).
Conclusion
Ion-exchange–membrane hybrid separation is a full-scale process by application especially for potable water supply. Ion-exchange coupled membrane processes are effective in removing hardness, nitrate, heavy metals and other emerging pollutants from water and wastewater at least on a pilot scale including boron. The membrane–ion-exchange pairing studies done so far are encouraging with regard to the cost implication of such hybrid processes especially in solving membrane fouling problem. Further, the hybrid system is favorable regarding energy consumption and space use as compared to MFs alone including the double-pass RO. The widely tested IX/RO hybrid is taken as an operational scale specially to remove boron in water. However, the real implication of the laboratory-scale studies on sustainability and feasibility needs further investigation that includes the cost of IX resin regeneration.
References
Abdulgader HA, Kochkodan V, Hilal N (2013) Hybrid ion exchange—pressure driven membrane processes in water treatment: a review. Sep Purif Technol 116:253–264. https://doi.org/10.1016/j.seppur.2013.05.052
Afsar NU et al (2017) Fabrication of cation exchange membrane from polyvinyl alcohol using lignin sulfonic acid: applications in diffusion dialysis process for alkali recovery. Sep Sci Technol 52:1106–1113. https://doi.org/10.1080/01496395.2017.1279629
Alharati A, Swesi Y, Fiaty K, Charcosset C (2017) Boron removal in water using a hybrid membrane process of ion exchange resin and microfiltration without continuous resin addition. J Water Process Eng 17:32–39. https://doi.org/10.1016/j.jwpe.2017.03.002
Al-Mamun A, Ahmad W, Baawain MS, Khadem M, Dhar BR (2018) A review of microbial desalination cell technology: configurations, optimization and applications. J Clean Prod 183:458–480. https://doi.org/10.1016/j.jclepro.2018.02.054
Amy G, Ghaffour N, Li Z, Francis L, Linares RV, Missimer T, Lattemann S (2017) Membrane-based seawater desalination: present and future prospects. Desalination 401:16–21. https://doi.org/10.1016/j.desal.2016.10.002
Ang WL, Mohammad AW, Hilal N, Leo CP (2015) A review on the applicability of integrated/hybrid membrane processes in water treatment and desalination plants. Desalination 363:2–18. https://doi.org/10.1016/j.desal.2014.03.008
Berezina NP, Kononenko NA, Dyomina OA, Gnusin NP (2008) Characterization of ion-exchange membrane materials: properties versus structure. Adv Colloid Interface Sci 139:3–28. https://doi.org/10.1016/j.cis.2008.01.002
Bornak B (2014) Desalination by ion exchange. In: Kucera J (ed) Desalination: water from water, Chap 11, pp 503–520. https://doi.org/10.1002/9781118904855.ch11
Bresler SA (1972) Desalination Process. USA Patent
Bryjak M, Wolska J, Kabay N (2008) Removal of boron from seawater by adsorption–membrane hybrid process: implementation and challenges. Desalination 223:57–62. https://doi.org/10.1016/j.desal.2007.01.202
Cappelle MA, Davis TA (2016) Chapter 7—ion exchange membranes for water softening and high-recovery desalination A2—Hankins, Nicholas P. In: Singh R (ed) Emerging membrane technology for sustainable water treatment. Elsevier, Boston, pp 163–179. https://doi.org/10.1016/B978-0-444-63312-5.00007-3
Choi S, Kim B, Han J (2017) Integrated pretreatment and desalination by electrocoagulation (EC)–ion concentration polarization (ICP) hybrid. Lab Chip 17:2076–2084. https://doi.org/10.1039/c7lc00258k
Chowdhary P, Raj A, Bharagava RN (2018) Environmental pollution and health hazards from distillery wastewater and treatment approaches to combat the environmental threats: a review. Chemosphere 194:229–246. https://doi.org/10.1016/j.chemosphere.2017.11.163
Cornelissen ER, Beerendonk EF, Nederlof MN, van der Hoek JP, Wessels LP (2009) Fluidized ion exchange (FIX) to control NOM fouling in ultrafiltration. Desalination 236:334–341. https://doi.org/10.1016/j.desal.2007.10.084
du Plessis A (2017) Global water availability, distribution and use. In: Freshwater challenges of South Africa and its Upper Vaal River. Springer, Berlin, pp 3–11. https://doi.org/10.1007/978-3-319-49502-6
El-Dessouky HT, Ettouney HM (2002) Chapter 1—introduction. In: El-Dessouky HT, Ettouney HM (eds) Fundamentals of salt water desalination. Elsevier, Amsterdam, pp 1–17. https://doi.org/10.1016/B978-044450810-2/50003
Esfahani IJ, Rashidi J, Ifaei P, Yoo C (2016) Efficient thermal desalination technologies with renewable energy systems: a state-of-the-art review. Korean J Chem Eng 33:351–387. https://doi.org/10.1007/s11814-015-0296-3
Eumine Suk D, Matsuura T (2006) Membrane-based hybrid processes: a review. Sep Sci Technol 41:595–626. https://doi.org/10.1080/01496390600552347
Goh PS, Ismail AF, Hilal N (2016) Nano-enabled membranes technology: sustainable and revolutionary solutions for membrane desalination? Desalination 380:100–104. https://doi.org/10.1016/j.desal.2015.06.002
Grabowski A, Zhang G, Strathmann H, Eigenberger G (2006) The production of high purity water by continuous electrodeionization with bipolar membranes: influence of the anion-exchange membrane permselectivity. J Membr Sci 281:297–306. https://doi.org/10.1016/j.memsci.2006.03.044
Hand KP, German CR (2018) Exploring ocean worlds on Earth and beyond. Nat Geosci 11:2–4. https://doi.org/10.1038/s41561-017-0045-9
Hilal N, Kochkodan V, Al Abdulgader H, Johnson D (2015) A combined ion exchange–nanofiltration process for water desalination: II: membrane selection. Desalination 363:51–57. https://doi.org/10.1016/j.desal.2014.11.017
Homaeigohar S, Elbahri M (2017) Graphene membranes for water desalination. NPG Asia Mater 9:e427. https://doi.org/10.1038/am.2017.135
Htun O, Maung htun O (2011) Boron removal by reverse osmosis membranes. National University of Singapore, Singapore
Humbert H, Gallard H, Jacquemet V, Croué J-P (2007) Combination of coagulation and ion exchange for the reduction of UF fouling properties of a high DOC content surface water. Water Res 41:3803–3811. https://doi.org/10.1016/j.watres.2007.06.009
Humbert H, Gallard H, Croué JP (2012) A polishing hybrid AER/UF membrane process for the treatment of a high DOC content surface water. Water Res 46:1093–1100. https://doi.org/10.1016/j.watres.2011.12.010
Humplik T et al (2011) Nanostructured materials for water desalination. Nanotechnology 22:292001. https://doi.org/10.1088/0957-4484/22/29/292001
International Desalination Association (2016) Water reuse and recycling: turning vision into reality. https://idadesal.org/ida-international-conference-on-water-reuse-and-recycling-turning-vision-into-reality/
Kabay N, Yilmaz İ, Bryjak M, Yüksel M (2006) Removal of boron from aqueous solutions by a hybrid ion exchange–membrane process. Desalination 198:158–165. https://doi.org/10.1016/j.desal.2006.09.011
Kabay N, Yüksel M, Samatya S, Arar Ö, Yüksel Ü (2007) Removal of nitrate from ground water by a hybrid process combining electrodialysis and ion exchange processes. Sep Sci Technol 42:2615–2627. https://doi.org/10.1080/01496390701511374
Kabay N, Köseoğlu P, Yapıc D, Yüksel Ü, Yüksel M (2013) Coupling ion exchange with ultrafiltration for boron removal from geothermal water-investigation of process parameters and recycle tests. Desalination 316:17–22. https://doi.org/10.1016/j.desal.2013.01.027
Kayvani Fard A, McKay G, Buekenhoudt A, Al Sulaiti H, Motmans F, Khraisheh M, Atieh M (2018) Inorganic membranes: preparation and application for water treatment and desalination. Materials. https://doi.org/10.3390/ma11010074
Khoiruddin WI (2016) Investigation of electrochemical and morphological properties of mixed matrix polysulfone-silica anion exchange membrane. J Eng Technol Sci 48:1–11. https://doi.org/10.5614/j.eng.technol.sci.2016.48.1.1
Kim H-C, Dempsey BA (2010) Removal of organic acids from EfOM using anion exchange resins and consequent reduction of fouling in UF and MF. J Membr Sci 364:325–330. https://doi.org/10.1016/j.memsci.2010.08.032
Kim B et al (2016) Purification of high salinity brine by multi-stage ion concentration polarization desalination. Sci Rep 6:31850. https://doi.org/10.1038/srep31850
Kwon Y, Hong S, Choi H, Jung J, Moon J, Tak T (2010) Development of fouling-resistant RO membranes using PEGA macromer. Desalin Water Treat 15:54–61. https://doi.org/10.5004/dwt.2010.1667
Le NL, Nunes SP (2016) Materials and membrane technologies for water and energy sustainability. Sustain Mater Technol 7:1–28. https://doi.org/10.1016/j.susmat.2016.02.001
Lenntech (2018) Desalination Post-treatment: boron removal process. https://www.lenntech.com/processes/desalination/post-treatment/post-treatments/boron-removal.htm. Accessed 03 Jan 2018
Li H, Cao H, Yang C (2016) Pressure-driven membrane and ion exchange hybrid system for the treatment of wastewater containing sulfite and nitrite. Desalin Water Treat 57:4398–4405. https://doi.org/10.1080/19443994.2014.995137
Miyoshi T, Takahashi Y, Suzuki T, Nitisoravut R, Polprasert C (2018) Treatment of highly-colored surface water by a hybrid microfiltration membrane system incorporating ion-exchange. Water Sci Technol Water Supply. https://doi.org/10.2166/ws.2018.132
Ochoa NA, Masuelli M, Marchese J (2006) Development of charged ion exchange resin-polymer ultrafiltration membranes to reduce organic fouling. J Membr Sci 278:457–463. https://doi.org/10.1016/j.memsci.2005.11.032
Reif R, Suárez S, Omil F, Lema JM (2008) Fate of pharmaceuticals and cosmetic ingredients during the operation of a MBR treating sewage. Desalination 221:511–517. https://doi.org/10.1016/j.desal.2007.01.111
Sachit ED, Veenstra NJ (2017) Foulant analysis of three RO membranes used in treating simulated brackish water of the Iraqi marshes. Membranes 7:23. https://doi.org/10.3390/membranes7020023
Sengupta AK and Sarkar S (2008) Brackish and sea water desalination using a hybrid ion exchange-nanofiltration process. USA Patent
Siekierka A, Wolska J, Kujawski W, Bryjak M (2017) Modification of poly(vinyl chloride) films by aliphatic amines to prepare anion-exchange membranes for Cr(VI) removal. Sep Sci Technol. https://doi.org/10.1080/01496395.2017.1358746
Strathmann H (2001) Membrane separation processes: current relevance and future opportunities. AIChE J 47:1077–1087
Strathmann H, Grabowski A, Eigenberger G (2013) Ion-exchange membranes in the chemical process industry. Ind Eng Chem Res 52:10364–10379
Tal A (2018) Addressing desalination’s carbon footprint: the Israeli experience. Water 10:197. https://doi.org/10.3390/w10020197
Talaeipour M, Nouri J, Hassani AH, Mahvi AH (2017) An investigation of desalination by nanofiltration, reverse osmosis and integrated (hybrid NF/RO) membranes employed in brackish water treatment. J Environ Health Sci Eng 15:18. https://doi.org/10.1186/s40201-017-0279-x
Taniguchi M, Fusaoka Y, Nishikawa T, Kurihara M (2004) Boron removal in RO seawater desalination. Desalination 167:419–426. https://doi.org/10.1016/j.desal.2004.06.157
Venkatesan A, Wankat PC (2011) Simulation of ion exchange water softening pretreatment for reverse osmosis desalination of brackish water. Desalination 271:122–131. https://doi.org/10.1016/j.desal.2010.12.022
Venkatesan A, Wankat PC (2012) Desalination of the Colorado River water: a hybrid approach. Desalination 286:176–186. https://doi.org/10.1016/j.desal.2011.11.018
Wang Y, Zou X, Sun L, Rong H, Zhu G (2018a) A zeolite-like aluminophosphate membrane with molecular-sieving property for water desalination. Chem Sci 9:2533–2539. https://doi.org/10.1039/c7sc04974a
Wang Z, Wu A, Colombi Ciacchi L, Wei G (2018b) Recent advances in nanoporous membranes for water purification. Nanomaterials 8:65. https://doi.org/10.3390/nano8020065
Werber JR, Elimelech M (2018) Permselectivity limits of biomimetic desalination membranes. Sci Adv 4:eaar8266
Wolska J, Bryjak M (2013) Methods for boron removal from aqueous solutions—a review. Desalination 310:18–24. https://doi.org/10.1016/j.desal.2012.08.003
Xu T (2005) Ion exchange membranes: state of their development and perspective. J Membr Sci 263:1–29
Yang E, Chae K-J, Choi M-J, He Z, Kim IS (2019) Critical review of bioelectrochemical systems integrated with membrane-based technologies for desalination, energy self-sufficiency, and high-efficiency water and wastewater treatment. Desalination 452:40–67. https://doi.org/10.1016/j.desal.2018.11.007
Yilmaz İ, Kabay N, Brjyak M, Yüksel M, Wolska J, Koltuniewicz A (2006) A submerged membrane-ion-exchange hybrid process for boron removal. Desalination 198:310–315. https://doi.org/10.1016/j.desal.2006.01.031
Zaib Q, Fath H (2013) Application of carbon nano-materials in desalination processes. Desalin Water Treat 51:627–636. https://doi.org/10.1080/19443994.2012.722772
Zhang J, Yuan H, Deng Y, Zha Y, Abu-Reesh IM, He Z, Yuan C (2018) Life cycle assessment of a microbial desalination cell for sustainable wastewater treatment and saline water desalination. J Clean Prod 200:900–910. https://doi.org/10.1016/j.jclepro.2018.07.197
Zou S, Qin M, He Z (2019) Tackle reverse solute flux in forward osmosis towards sustainable water recovery: reduction and perspectives. Water Res 149:362–374. https://doi.org/10.1016/j.watres.2018.11.015
Acknowledgements
I would like to acknowledge the Digital Library of Addis Ababa University. I am indebted to thank Mrs. Alem Alemayehu for her immense support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Gebreeyessus, G.D. Status of hybrid membrane–ion-exchange systems for desalination: a comprehensive review. Appl Water Sci 9, 135 (2019). https://doi.org/10.1007/s13201-019-1006-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-019-1006-9