Abstract
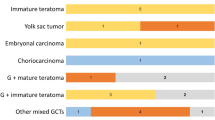
Germ cell tumors (GCT) are an intriguing group of neoplasm having myriad clinical and morphological presentation. More and more transcription factors are being evaluated for identification of same. To study the spectrum of GCTs in a tertiary care center and the use of a stem cell marker OCT4 as a diagnostic adjunct, a retrospective 5-year (2008–2013) study was carried out. Immunohistochemistry (IHC) with OCT4 was performed on all cases and IHC for α feto protein (AFP), CD30, and epithelial membrane antigen (EMA) as per requirement. Cohort included 73 cases (23 males and 50 females). Testicular and ovarian GCTs accounted for 95.83% and 35.71% respectively. In males, seminoma was the commonest (34.78%) followed by mixed GCT (26%). 17.85% of ovarian GCTs were malignant mostly constituted by dysgerminoma (18%). Benign mature cystic teratoma (MCT) constituted 50% of ovarian GCTs. OCT4 immunoexpression was seen in all cases of seminoma/dysgerminoma, embryonal carcinoma, immature teratoma, and seminomatous/embryomatous component of mixed GCTs. Pure yolk sac tumor (YST) and MCT were consistently negative. OCT4 was especially helpful in identification of mixed GCT. A panel of immunohistochemical markers would be a more ideal way to identify and clarify the components because correct identification of the components is important for therapeutic intervention and prognostication. OCT4 being a primordial germ cell marker predicts aggressive behavior and targeted therapy against this should be investigated.



Similar content being viewed by others
Code Availability
Not applicable.
References
Pierce GB (1967) Teratocarcinoma: model for a developmental concept of cancer. Curr Top Dev Biol 2:223–246. https://doi.org/10.1016/s0070-2153(08)60289-6
Anderson R, Copeland TK, Schöler H, Heasman J, Wylie C (2000) The onset of germ cell migration in the mouse embryo. Mech Dev 91(1–2):61–68. https://doi.org/10.1016/s0925-4773(99)00271-3
Cheng L, Sung MT, Cossu-Rocca P, Jones TD, MacLennan GT, De Jong J, Lopez-Beltran A, Montironi R, Looijenga LH (2007) OCT4: biological functions and clinical applications as a marker of germ cell neoplasia. J Pathol 211(1):1–9. https://doi.org/10.1002/path.2105
Gershenson DM (2007) Management of ovarian germ cell tumors. J Clin Oncol 25(20):2938–2943. https://doi.org/10.1200/JCO.2007.10.8738
Looijenga LH, Stoop H, de Leeuw HP, de Gouveia Brazao CA, Gillis AJ, van Roozendaal KE, van Zoelen EJ, Weber RF, Wolffenbuttel KP, van Dekken H, Honecker F, Bokemeyer C, Perlman EJ, Schneider DT, Kononen J, Sauter G, Oosterhuis JW (2003) POU5F1 (OCT3/4) identifies cells with pluripotent potential in human germ cell tumors. Cancer Res 63(9):2244–2250
Nichols J, Zevnik B, Anastassiadis K, Niwa H, Klewe-Nebenius D, Chambers I, Schöler H, Smith A (1998) Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell. 95(3):379–91. https://doi.org/10.1016/s0092-8674(00)81769-9
Berney DM, Looijenga LH, Idrees M, Oosterhuis JW, Rajpert-De Meyts E, Ulbright TM, Skakkebaek NE (2016) Germ cell neoplasia in situ (GCNIS): evolution of the current nomenclature for testicular pre-invasive germ cell malignancy. Histopathology 69(1):7–10. https://doi.org/10.1111/his.12958
Nogales FF, Dulcey I, Preda O (2014) Germ cell tumors of the ovary: an update. Arch Pathol Lab Med 138(3):351–362. https://doi.org/10.5858/arpa.2012-0547-RA
Sankaranarayanan R, Ferlay J (2006) Worldwide burden of gynaecological cancer: the size of the problem. Best Pract Res Clin Obstet Gynaecol 20(2):207–225. https://doi.org/10.1016/j.bpobgyn.2005.10.007
Mankar DV, Jain GK (2015) Histopathological profile of ovarian tumours: a twelve year institutional experience. Muller J Med Sci Res 6:107–111
Bosl GJ, Motzer RJ (1997) Testicular germ-cell cancer. N Engl J Med 337(4):242–253. https://doi.org/10.1056/NEJM199707243370406
Bhattacharyya NK, De A, Bera P, Sristidhar M, Chakraborty S, Bandopadhyay R (2010) Ovarian tumors in pediatric age group - a clinicopathologic study of 10 years’ cases in West Bengal. India Indian J Med Paediatr Oncol 31(2):54–57. https://doi.org/10.4103/0971-5851.71656
Atlasi Y, Mowla SJ, Ziaee SAM, Bahrami A-R (2007) OCT-4, an embryonic stem cell marker, is highly expressed in bladder cancer. Int J Cancer 120:1598–1602
Wu YC, Ling TY, Lu SH, Kuo HC, Ho HN, Yeh SD, Shen CN, Huang YH (2012) Chemotherapeutic sensitivity of testicular germ cell tumors under hypoxic conditions is negatively regulated by SENP1-controlled sumoylation of OCT4. Cancer Res 72(19):4963–4973. https://doi.org/10.1158/0008-5472.CAN-12-0673
Lakshmanan M, Gupta S, Kumar V, Akhtar N, Chaturvedi A, Misra S, Jain K, Garg S (2018) Germ cell tumor ovary: an institutional experience of treatment and survival outcomes. Indian J Surg Oncol 9(2):215–219. https://doi.org/10.1007/s13193-018-0742-x
Joshi A, Zanwar S, Shetty N, Patil V, Noronha V, Bakshi G et al (2016) Epidemiology of male seminomatous and nonseminomatous germ cell tumors and response to first-line chemotherapy from a tertiary cancer center in India. Indian J Cancer 53:313–316. https://doi.org/10.4103/0019-509X.197741
Ulbright TM (2005) Germ cell tumors of the gonads: a selective review emphasizing problems in differential diagnosis, newly appreciated, and controversial issues. Mod Pathol 18(Suppl 2):S61-79. https://doi.org/10.1038/modpathol.3800310
Cheng L (2004) Establishing a germ cell origin for metastatic tumors using OCT4 immunohistochemistry. Cancer 101(9):2006–2010. https://doi.org/10.1002/cncr.20566
Sung MT, Jones TD, Beck SD, Foster RS, Cheng L (2006) OCT4 is superior to CD30 in the diagnosis of metastatic embryonal carcinomas after chemotherapy. Hum Pathol 37(6):662–667. https://doi.org/10.1016/j.humpath.2006.01.019
Talerman, Aleksander, and Russell Vang. Blaustein’s pathology of the female genital tract. 6th ed., Springer, 2011.Chapter 16 ,Germ cell tumor. P 848–897. https://doi.org/10.1007/978-1-4419-0489-8
Sung MT, Jones TD, Beck SD Foster RR, Cheng L ( 2006) OCT3/4 is superior to CD30 in the diagnosis of metastatic EC after chemotherapy. Hum Pathol 37(6) 662–7
McKenney JK, Heerema-McKenney A, Rouse RV (2007) Extragonadal germ cell tumors: a review with emphasis on pathologic features, clinical prognostic variables, and differential diagnostic considerations. Adv AnatPathol 14(2):69–92. https://doi.org/10.1097/PAP.0b013e31803240e6
Baker PM, Oliva E (2005) Immunohistochemistry as a tool in the differential diagnosis of ovarian tumors: an update. Int J GynecolPathol 24(1):39–55
Ismail S. Mohiuddin, Sung-Jen Wei, Min H. Kang (2020) Role of OCT4 in cancer stem-like cells and chemotherapy resistance. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 1866(4) 165432 https://doi.org/10.1016/j.bbadis.2019.03.005.
Rajeswari B, Nair M, Ninan A, Parukuttyamma K (2016) Ovarian tumors in children: 10-year experience from a tertiary care center in South India. Indian J Cancer 53:292–295. https://doi.org/10.4103/0019-509X.197726
Hemanth Kumar, S.V. Saju, Venkatraman Radhakrishnan, Anand Raja, Trivadi S ( 2020) Ganesan, Manikandan Dhanushkodi et al. Analysis of extra-cranial germ cell tumors in male children: experience from a single centre in India. Pediatr Hematol Oncol J 5(2):37–42. https://doi.org/10.1016/j.phoj.2020.03.012
Shen H, Shih J, Hollern DP, Wang L, Bowlby R, Tickoo SK et al (2018) Integrated molecular characterization of testicular germ cell tumors. Cell Rep 23(11):3392–3406. https://doi.org/10.1016/j.celrep.2018.05.039
Biermann K, Göke F, Nettersheim D, Eckert D, Zhou H, Kahl P, Gashaw I, Schorle H, Büttner R (2007) c-KIT is frequently mutated in bilateral germ cell tumours and down-regulated during progression from intratubular germ cell neoplasia to seminoma. J Pathol 213(3):311–318. https://doi.org/10.1002/path.2225
Looijenga LH, de Leeuw H, van Oorschot M, van Gurp RJ, Stoop H, Gillis AJ et al (2003) Stem cell factor receptor (c-KIT) codon 816 mutations predict development of bilateral testicular germ-cell tumors. Cancer Res 63(22):7674–7678
Gilbert D, Rapley E, Shipley J (2011) Testicular germ cell tumours: predisposition genes and the male germ cell niche. Nat Rev Cancer 11(4):278–288. https://doi.org/10.1038/nrc3021
Isabella Syring, Joanna Bartels, Stefan Holdenrieder, Glen Kristiansen, Stefan C. Müller, Jörg Ellinger (2015) Circulating serum miRNA (miR-367-3p, miR-371a-3p, miR-372-3p and miR-373-3p) as biomarkers in patients with testicular germ cell cancer, The Journal of Urology 193(1) 331-7, ISSN 0022-5347. https://doi.org/10.1016/j.juro.2014.07.010
Albany C, Hever-Jardine M. P, von Herrmann K. M, Yim C. Y, Tam J, Warzecha J. M et al. (2017) Refractory testicular germ cell tumors are highly sensitive to the second generation DNA methylation inhibitor guadecitabine. Oncotarget. 8: 2949–59.https://doi.org/10.18632/oncotarget.13811
Cavallo F, Graziani G, Antinozzi C, Feldman DR, Houldsworth J, Bosl GJ et al (2012) Reduced proficiency in homologous recombination underlies the high sensitivity of embryonal carcinoma testicular germ cell tumors to cisplatin and poly (ADP-ribose) polymerase inhibition. PLoS ONE 7(12):e51563. https://doi.org/10.1371/journal.pone.0051563
Fernández-Cabezudo MJ, Faour I, Jones K, Champagne DP, Jaloudi MA, Mohamed YA, Bashir G et al (2016) Deficiency of mitochondrial modulator MCJ promotes chemoresistance in breast cancer. JCI Insight 1(7):e86873. https://doi.org/10.1172/jci.insight.86873
Faulkner SW, Friedlander ML (2000) Microsatellite instability in germ cell tumors of the testis and ovary. Gynecol Oncol 79(1):38–43. https://doi.org/10.1006/gyno.2000.5906.
King BL, Carcangiu ML, Carter D, et al (1995) Microsatellite instability in ovarian neoplasms. Br J Cancer. 1995;72: 376 –382 King, B., Carcangiu, ML., Carter, D. et al. Microsatellite instability in ovarian neoplasms. Br J Cancer 72 376–382. https://doi.org/10.1038/bjc.1995.341
JorgensenN,Muller J, Jaubert F, Clausen OP, Skakkebaek NE (1997) Heterogeneity of gonadoblastoma germ cells: similarities with immature germ cells, spermatogonia and testicular carcinoma in situ cells. Histopathology. 30: 177–86. https://doi.org/10.1046/j.1365-2559.1997.d01-580.x
Cools M, Looijenga LH, Wolffenbuttel KP, Drop SL (2009) Disorders of sex development: update on the genetic background, terminology and risk for the development of germ cell tumors. World J Pediatr 5(2):93–102. https://doi.org/10.1007/s12519-009-0020-7
Acknowledgements
Authors are thankful to King Georges Medical University, Lucknow, for providing necessary resources. We would like to acknowledge Prof Raj Mehrotra, former Professor, dept of Pathology and Dean, KGMU, who had guided us in our study and is no longer with us. We want to thank our technical team members especially Mr. Kamlesh Sharma and Miss Nidhi Varma for their support. The authors have no other relevant affiliations or financial involvements.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
This is a retrospective study on the paraffin blocks stored in the Department of Pathology, KGMU, Lucknow, and consent for use of the stored tissue is taken from the patients at the time of surgery. Institute ethics committee has a waiver for these retrospective studies on paraffin blocks.
Conflict of Interest
The authors declare no competing interests.
Transparency Declaration
The authors affirm that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned have been explained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Paramita, P., Preeti, A., Mili, J. et al. Spectrum of Germ Cell Tumor (GCT): 5 Years’ Experience in a Tertiary Care Center and Utility of OCT4 as a Diagnostic Adjunct. Indian J Surg Oncol 13, 533–541 (2022). https://doi.org/10.1007/s13193-022-01522-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-022-01522-w