Abstract
Purpose
68Ga-labeled 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid-d-Phe1-Tyr3-octreotide (68Ga-DOTATOC) is taken up by activated macrophages, which accumulate in active inflammatory lesions. The purpose of this study was to investigate the feasibility of 68Ga-DOTATOC PET/CT for assessment of vulnerable plaque, by evaluating correlation between aortic uptake of 68Ga-DOTATOC and cardiovascular risk factors.
Methods
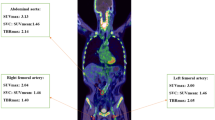
Fifty patients with neuroendocrine tumors who underwent 68Ga-DOTATOC PET/CT were retrospectively enrolled. The uptakes in the thoracic aorta were measured by two methods: multi-sample region-of-interest (ROI) method and single volume-of-interest (VOI) method. TBRmax-avg, TBRmean-avg, TBRmax-VOI, and TBRmean-VOI were defined by maximum and mean target-to-background ratio (TBR) from the multi-sample ROI method and the single VOI method, respectively.
Results
Framingham risk score (FRS) exhibited significant correlations with TBRmax-avg and TBRmean-avg, as well as TBRmax-VOI (r = 0.3389–0.4593, P < 0.05 for all). TBRmax-avg and TBRmax-VOI were significantly higher in high FRS group than in low FRS group (1.48 ± 0.21 vs. 1.70 ± 0.17, P < 0.001 for TBRmax-avg and 1.90 ± 0.33 vs. 2.25 ± 0.36, P = 0.002 for TBRmax-VOI). TBR exhibited high correlations between the two measuring methods (r = 0.9684, P < 0.001 for TBRmean-avg and TBRmean-VOI and r = 0.8681, P < 0.001 for TBRmax-avg and TBRmax-VOI).
Conclusions
68Ga-DOTATOC uptake in the thoracic aorta exhibited a significant correlation with cardiovascular risk factors, which suggests the feasibility of 68Ga-DOTATOC PET for vulnerable plaque imaging, with a simple measurement of the single VOI method that is comparable to the multi-sample ROI-based approach.





Similar content being viewed by others
References
Writing Group M, Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, et al. Heart disease and stroke statistics—2016 update: a report from the American Heart Association. Circulation. 2016;133:e38–360.
Naghavi M, Libby P, Falk E, Casscells SW, Litovsky S, Rumberger J, et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: part II. Circulation. 2003;108:1772–8.
Tarkin JM, Joshi FR, Rudd JH. PET imaging of inflammation in atherosclerosis. Nat Rev Cardiol. 2014;11:443–57.
Ross R. Atherosclerosis—an inflammatory disease. N Engl J Med. 1999;340:115–26.
Li X, Bauer W, Kreissl MC, Weirather J, Bauer E, Israel I, et al. Specific somatostatin receptor II expression in arterial plaque: (68)Ga-DOTATATE autoradiographic, immunohistochemical and flow cytometric studies in apoE-deficient mice. Atherosclerosis. 2013;230:33–9.
Graebe M, Pedersen SF, Borgwardt L, Hojgaard L, Sillesen H, Kjaer A. Molecular pathology in vulnerable carotid plaques: correlation with [18]-fluorodeoxyglucose positron emission tomography (FDG-PET). Eur J Vasc Endovasc Surg. 2009;37:714–21.
Pedersen SF, Graebe M, Fisker Hag AM, Hojgaard L, Sillesen H, Kjaer A. Gene expression and 18FDG uptake in atherosclerotic carotid plaques. Nucl Med Commun. 2010;31:423–9.
Rudd JH, Warburton EA, Fryer TD, Jones HA, Clark JC, Antoun N, et al. Imaging atherosclerotic plaque inflammation with [18F]-fluorodeoxyglucose positron emission tomography. Circulation. 2002;105:2708–11.
Rominger A, Saam T, Wolpers S, Cyran CC, Schmidt M, Foerster S, et al. 18F-FDG PET/CT identifies patients at risk for future vascular events in an otherwise asymptomatic cohort with neoplastic disease. J Nucl Med. 2009;50:1611–20.
Rudd JH, Myers KS, Bansilal S, Machac J, Rafique A, Farkouh M, et al. (18)Fluorodeoxyglucose positron emission tomography imaging of atherosclerotic plaque inflammation is highly reproducible: implications for atherosclerosis therapy trials. J Am Coll Cardiol. 2007;50:892–6.
D'Agostino RB Sr, Vasan RS, Pencina MJ, Wolf PA, Cobain M, Massaro JM, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. 2008;117:743–53.
McCollough CH, Ulzheimer S, Halliburton SS, Shanneik K, White RD, Kalender WA. Coronary artery calcium: a multi-institutional, multimanufacturer international standard for quantification at cardiac CT. Radiology. 2007;243:527–38.
Lee SJ, Paeng JC. Nuclear molecular imaging for vulnerable atherosclerotic plaques. Korean J Radiol. 2015;16:955–66.
Li X, Samnick S, Lapa C, Israel I, Buck AK, Kreissl MC, et al. 68Ga-DOTATATE PET/CT for the detection of inflammation of large arteries: correlation with18F-FDG, calcium burden and risk factors. EJNMMI Res. 2012;2:52.
Pedersen SF, Sandholt BV, Keller SH, Hansen AE, Clemmensen AE, Sillesen H, et al. 64Cu-DOTATATE PET/MRI for detection of activated macrophages in carotid atherosclerotic plaques: studies in patients undergoing endarterectomy. Arterioscler Thromb Vasc Biol. 2015;35:1696–703.
Malmberg C, Ripa RS, Johnbeck CB, Knigge U, Langer SW, Mortensen J, et al. 64Cu-DOTATATE for noninvasive assessment of atherosclerosis in large arteries and its correlation with risk factors: head-to-head comparison with 68Ga-DOTATOC in 60 patients. J Nucl Med. 2015;56:1895–900.
Rominger A, Saam T, Vogl E, Ubleis C, la Fougere C, Forster S, et al. In vivo imaging of macrophage activity in the coronary arteries using 68Ga-DOTATATE PET/CT: correlation with coronary calcium burden and risk factors. J Nucl Med. 2010;51:193–7.
Tarkin JM, Joshi FR, Evans NR, Chowdhury MM, Figg NL, Shah AV, et al. Detection of atherosclerotic inflammation by 68Ga-DOTATATE PET compared to [18F]FDG PET imaging. J Am Coll Cardiol. 2017;69:1774–91.
Rudd JH, Myers KS, Bansilal S, Machac J, Pinto CA, Tong C, et al. Atherosclerosis inflammation imaging with 18F-FDG PET: carotid, iliac, and femoral uptake reproducibility, quantification methods, and recommendations. J Nucl Med. 2008;49:871–8.
Khoury Z, Schwartz R, Gottlieb S, Chenzbraun A, Stern S, Keren A. Relation of coronary artery disease to atherosclerotic disease in the aorta, carotid, and femoral arteries evaluated by ultrasound. Am J Cardiol. 1997;80:1429–33.
Funding
This study was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea (Grant Number HI14C1277).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Lee Reeree, Jihyun Kim, Jin Chul Paeng, Jung Woo Byun, Gi Jeong Cheon, Dong Soo Lee, June-Key Chung, and Keon Wook Kang declare that they have no conflict of interest.
Ethical Approval
All procedures followed were performed in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975, as revised in 2013. The study design was approved by the Institutional Review Board of the Seoul National University Hospital (H-1711-112-901).
This manuscript has not been published before or is not under consideration for publication anywhere else and has been approved by all co-authors.
Informed Consent
The study design of the retrospective analysis and exemption of informed consent were approved by the Institutional Review Board of the Seoul National University Hospital.
Electronic Supplementary Material
Supplemental Table 1
(DOCX 27.1 kb)
Supplemental Table 2
(DOCX 27.5 kb)
Supplemental Figure 1
(TIF 19.4 mb)
Supplemental Figure 2
(TIF 18.9 mb)
Rights and permissions
About this article
Cite this article
Lee, R., Kim, J., Paeng, J.C. et al. Measurement of 68Ga-DOTATOC Uptake in the Thoracic Aorta and Its Correlation with Cardiovascular Risk. Nucl Med Mol Imaging 52, 279–286 (2018). https://doi.org/10.1007/s13139-018-0524-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-018-0524-y