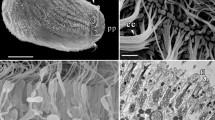
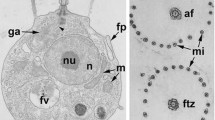
Abstract
The kinetid (flagellar/ciliary apparatus) of eukaryotic cells is an important source of phylogenetic information. It was found to be a prospective morphological phylogenetic marker in sponges, since its arrangement in choanocytes is congruent with the topology of the phylogenetic trees. However, investigation of the kinetid of sponge larval cells remains fragmentary. Here, we report the results of an ultrastructural study on the larval kinetids of the freshwater sponges Eunapius fragilis and Lubomirskia baikalensis (Demospongiae: Spongillida). Their kinetids were found to comprise a kinetosome associated with an accessory centriole and linked to the nucleus by a simple fibrillar root. The kinetosome bears a transverse cytoskeleton: filamentous train and microtubules which radiate from a microtubule organising centre (MTOC) shaped as a large hollow foot. In the short transition zone, between the central axonemal microtubules and kinetosome, a transverse plate with an axosome (central thickening) occurs. We have reviewed the kinetids of different sponge larvae to reconstruct the ancestral state of these traits. Thus, we suggest that spongillids retain the plesiomorphic characteristics of roots and an accessory centriole. But they possess the peculiarities of the transition zone, transverse cytoskeleton and MTOC structure.






Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Amano, S., & Hori, I. (1992). Metamorphosis of calcareous sponges I. Ultrastructure of free-swimming larvae. Invertebrate Reproduction and Development, 21(2), 81–90. https://doi.org/10.1080/07924259.1992.9672223.
Amano, S., & Hori, I. (1994). Metamorphosis of a demosponge i. Cells and structure of swimming larva. Invertebrate Reproduction and Development, 25(3), 193–204. https://doi.org/10.1080/07924259.1994.9672386.
Amano, S., & Hori, I. (1996). Transdifferentiation of larval flagellated cells to choanocytes in the metamorphosis of the demosponge Haliclona permollis. Biological Bulletin, 190(2), 161–172. https://doi.org/10.2307/1542536.
Amano, S., & Hori, I. (2001). Metamorphosis of coeloblastula performed by multipotential larval flagellated cells in the calcareous sponge Leucosolenia laxa. Biological Bulletin, 200(1), 20–32. https://doi.org/10.2307/1543082.
Andersen, R. A., Barr, D. J. S., Lynn, D. H., Melkonian, M., Moestrup, & Sleigh, M. A. (1991). Terminology and nomenclature of the cytoskeletal elements associated with the flagellar/ciliary apparatus in protists. Protoplasma, 164, 1–8. https://doi.org/10.1007/BF01320809.
Berquist, P. R. (1978). Sponges. London: Hutchinson.
Borojevic, R. (1969). Etude du développement et de la differentiation cellulaire d’éponges calcaires Calcinées (genres Clathrina et Ascandra). Annales d'Embryologie et de Morphogenèse, 2, 15–36.
Boury-Esnault, N. (1976). Ultrastructure de la larve parenchymella d’Hamigera hamigera (Poecilosclerida). Origine des cellules grises. Cahiers de Biologie Marine, 17, 9–20.
Boury-Esnault, N., Ereskovsky, A., Bézac, C., & Tokina, D. (2003). Larval development in the Homoscleromorpha (Porifera, Demospongiae). Invertebrate Biology, 122(3), 187–202. https://doi.org/10.1111/j.1744-7410.2003.tb00084.x.
Cárdenas, P., Pérez, T., & Boury-Esnault, N. (2012). Sponge systematics facing new challenges. Advances in Marine Biology, 61, 79–209. https://doi.org/10.1016/B978-0-12-387787-1.00010-6.
Dingle, A. D., & Larson, D. E. (1981). Structure and protein composition of the striated flagellar rootlets of some protists. BioSystems, 14, 345–358.
Edgar, R. (2004). MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research, 32(5), 1792–1797. https://doi.org/10.1093/nar/gkh340.
Efremova, S., Sukhodolskaya, A., & Alekseeva, N. (1988). The different structure of kinetosome rootlet systems in flagellated cells of the larvae and the choanocytes of sponges. In Modern and Perspective Investigations. Porifera and Cnidaria (pp. 22–23). Leningrad: USSR Academy of Sciences, Zoological institute.
Ereskovsky, A. V., & Tokina, D. B. (2004). Morphology and fine structure of the swimming larvae of Ircinia oros (Porifera, Demospongiae, Dictyoceratida). Invertebrate Reproduction and Development, 45(2), 137–150. https://doi.org/10.1080/07924259.2004.9652583.
Ereskovsky, A. V., & Willenz, P. (2008). Larval development in Guancha arnesenae (Porifera, Calcispongiae, Calcinea). Zoomorphology, 127, 175–187. https://doi.org/10.1007/s00435-008-0061-9.
Ereskovsky, A. V., Tokina, D. B., Bezac, C., & Boury-Esnault, N. (2007). Metamorphosis of Cinctoblastula larvae (Homoscleromorpha, Porifera). Journal of Morphology, 268, 518–528. https://doi.org/10.1002/jmor.10506.
Evans, C. W. (1977). The ultrastructure of larvae from the marine sponge Halichondria moorei Bergquist (Porifera, Desmospongiae). Cahiers de Biologie Marine, 18(1), 427–433.
Flammang, P., Demeulenaere, S., & Jangoux, M. (1994). The role of podial secretions in adhesion in two species of sea stars (Echinodermata). Biological Bulletin, 187, 35–47. https://doi.org/10.2307/1542163.
Galissian, M.-F., & Vacelet, J. (1992). Ultrastructure of the oocyte and embryo of the calcified sponge, Petrobiona massiliana (Porifera, Calcarea). Zoomorphology, 112, 133–141.
Gallissian, M.-F. (1983). Etude ultrastructurale du developpement embryonnaire chez Grantia compressa F (Porifera, Calcarea). Archives d’Anatomie Microscopique, 72(1), 59–75.
Gonobobleva, E. (2007). Basal apparatus formation in external flagellated cells of Halisarca dujardini larvae (Demospongiae: Halisarcida) in the course of embryonic development. Porifera Research: Biodiversity, Innovation and Sustainability (pp. 345–351).
Gonobobleva, E., & Ereskovsky, A. (2004). Metamorphosis of the larva of Halisarca dujardini (Demospongiae, Halisarcida). Bulletin de l’Institut Royal des Sciences naturelles de Belgique, Biologie, 74, 101–115.
Gonobobleva, E., & Maldonado, M. (2009). Choanocyte ultrastructure in Halisarca dujardini (Demospongiae, Halisarcida). Journal of Morphology, 270, 615–627. https://doi.org/10.1002/jmor.10709.
Hartman, W. D. (1958). A re-examination of Bidder’s classification of the Calcarea. Systematic Zoology, 7, 55–109. https://doi.org/10.2307/2411971.
Hill, M., Hill, A., Lopez, J., Peterson, K., Pomponi, S., Diaz, M., et al. (2013). Reconstruction of family-level phylogenetic relationships within Demospongiae (Porifera) using nuclear encoded housekeeping genes. PLoS One, 8(1), e50437.
Hooper, J. N. A., & van Soest, R. W. M. (2002). Class Demospongiae Sollas, 1885. In J. N. A. Hooper & R. W. M. van Soest (Eds.), Systema Porifera: A guide to the classification of sponges (pp. 15–18). New York: Kluwer. https://doi.org/10.1007/978-1-4615-0747-5_3.
Ivanova, L. V. (1997a). New data about morphology and metamorphosis of the spongillid larvae (Porifera, Spongillidae). 1. Morphology of the free-swimming larvae. In A. V. Ereskovsky, H. Keupp, & H. R. Kohring (Eds.), Modern problems of Poriferan biology (pp. 55–71). Berlin: Berliner Geowiss Abh, Freie University.
Ivanova, L. V. (1997b). New data about morphology and metamorphosis of the spongillid larvae (Porifera, Spongillidae). 2. The metamorphosis of the spongillid larva. In A. V. Ereskovsky, H. Keupp, & H. R. Kohring (Eds.), Modern problems of Poriferan biology (pp. 73–91). Berlin: Berliner Geowiss Abh, Freie University.
Karpov, S. A. (2000). Flagellate phylogeny: Ultrastructural approach. In J. Green & B. Leadbeater (Eds.), The flagellates (pp. 336–360). London: Taylor and Francis.
Karpov, S. A. (2016). Flagellar apparatus structure of choanoflagellates. Cilia, 5(1), 1–5. https://doi.org/10.1186/s13630-016-0033-5.
Karpov, S. A., & Fokin, S. I. (1995). The structural diversity of flagellar transitional zone in heterotrophic flagellates and other protists. In S. A. Karpov (ed.), The biology of free-living heterotrophic flagellates. Tsitologia (vol. 37, pp. 1038–1052).
Lanna, E., & Klautau, M. (2012). Embryogenesis and larval ultrastructure in Paraleucilla magna (Calcarea, Calcaronea), with remarks on the epilarval trophocyte epithelium (‘placental membrane’). Zoomorphology, 131, 277–292. https://doi.org/10.1007/s00435-012-0160-5.
Lévi, C. (1964). Ultrastructure de la larve parenchymella de démosponge. I: Mycale contarenii. Cahiers de Biologie Marine, 5, 97–104.
Lévi, C., & Lévi, P. (1976). Embryogenese de Chondrosia reniformis (Nardo), demosponge vipare, et transmission des bacteries symbiotiques. Annales des Sciences Naturelles. Zoologie et biologie animale, 18, 367–380.
Leys, S. P., & Degnan, B. M. (2001). Cytological basis of photoresponsive behavior in a sponge larva. Biological Bulletin, 201(3), 323–338. https://doi.org/10.2307/1543611.
Leys, S. P., & Degnan, B. M. (2002). Embryogenesis and metamorphosis in a haplosclerid demosponge: Gastrulation and transdifferentiation of larval ciliated cells to choanocytes. Invertebrate Biology, 121(3), 171–189.
Lynn, D. H., & Small, E. G. (1981). Protist kinetids: Structural conservatism, kinetid structure and ancestral states. BioSystems, 14, 377–385. https://doi.org/10.1016/0303-2647(81)90044-7.
Maddison, W., & Maddison, D. (2019). Mesquite: a modular system for evolutionary analysis. Version 3.61. http://www.mesquiteproject.org
Maldonado, M. (2006). The ecology of the sponge larva. Canadian Journal of Zoology., 84(2), 175–194. https://doi.org/10.1139/Z05-177.
Maldonado, M. (2009). Embryonic development of verongid demosponges supports the independent acquisition of spongin skeletons as an alternative to the siliceous skeleton. Biological Journal of the Linean Society, 97, 427–447. https://doi.org/10.1111/j.1095-8312.2009.01202.x.
Maldonado, M., & Riesgo, A. (2008). Reproductive output in a Mediterranean population of the homosclerophorid Corticium candelabrum (Porifera, Demospongiae), with notes on the ultrastructure and behavior of the larva. Marine Ecology, 29(2), 298–316. https://doi.org/10.1111/j.1439-0485.2008.00244.x.
Maldonado, M., Durfort, M., McCarthy, D. A., & Young, C. M. (2003). The cellular basis of photobehavior in the tufted parenchymella larva of demosponges. Marine Biology, 143(3), 427–441. https://doi.org/10.1007/s00227-003-1100-1.
Manconi, R., & Pronzato, R. (2002). Suborder Spongillina subord. Nov.: Freshwater sponges. In J. N. A. Hooper & R. W. M. van Soest (Eds.), Systema Porifera: A guide to the classification of sponges (pp. 921–1021). New York: Kluwer. https://doi.org/10.1007/978-1-4615-0747-5_97.
Melkonian, M. (1982). Structural and evolutionary aspects of the Flagellar apparatus in Green algae and land plants. Taxon, 31(2), 255–265. https://doi.org/10.2307/1219989.
Moestrup, Ø. (1982). Phycological reviews 7: Flagellar structure in algae: A review, with new observations particularly on the Chrysophyceae, Phaeophyceae (Fucophyceae), Euglenophyceae, and Reckertia. Phycologia, 21(4), 427–528. https://doi.org/10.2216/i0031-8884-21-4-427.1.
Moestrup, Ø. (2000). The Flagellar cytoskeleton: Introduction of general terminology for microtubular Flagellar roots in Protists. In B. S. C. Leadbeater & J. C. Green (Eds.), The flagellates. Systematics association special publications (pp. 69–94). London: Taylor & Francis.
Morrow, C., & Cárdenas, P. (2015). Proposal for a revised classification of the Demospongiae (Porifera). Frontiers in Zoology, 12, 7. https://doi.org/10.1186/s12983-015-0099-8.
Morrow, C. C., Picton, B. E., Erpenbeck, D., Boury-Esnault, N., Maggs, C. A., & Allcock, A. L. (2012). Congruence between nuclear and mitochondrial genes in Demospongiae: A new hypothesis for relationships within the G4 clade (Porifera: Demospongiae). Molecular Phylogenetics and Evolution, 62(1), 174–190. https://doi.org/10.1016/j.ympev.2011.09.016.
Morrow, C. C., Redmond, N. E., Picton, B. E., Thacker, R. W., Collins, A. G., Maggs, C. A., Sigwart, J. D., & Allcock, A. L. (2013). Molecular phylogenies support homoplasy of multiple morphological characters used in the taxonomy of Heteroscleromorpha (Porifera: Demospongiae). Integrative and Comparative Biology, 53(3), 428–446. https://doi.org/10.1093/icb/ict065.
Nielsen, C. (2019). Early animal evolution: A morphologist’s view. Royal Society Open Science, 6, 190638. https://doi.org/10.1098/rsos.190638.
Pozdnyakov, I. R., Sokolova, A. M., Ereskovsky, A. V., & Karpov, S. A. (2017). Kinetid structure of choanoflagellates and choanocytes of sponges does not support their close relationship. Protistology, 11(4), 248–264. https://doi.org/10.21685/1680-0826-2017-11-4-6.
Pozdnyakov, I., Sokolova, A., Ereskovsky, A., & Karpov, S. (2018). Kinetid structure in sponge choanocytes of Spongillida in the light of evolutionary relationships within Demospongiae. Zoological Journal of the Linnean Society, 184(2), 255–272. https://doi.org/10.1093/zoolinnean/zlx109/4905843.
Pozdnyakov, I., Sokolova, A., Ereskovsky, A., & Karpov, S. (2020). The kinetid structure of two Oscarellid sponges (class Homoscleromorpha) unveils plesiomorphies in kinetids of Homoscleromorpha-Calcarea lineage. Invertebrate Biology in press.
Riesgo, A., Taylor, C., & Leys, S. P. (2007). Reproduction in a carnivorous sponge: The significance of the absence of an aquiferous system to the sponge body plan. Evolution and Development, 9(6), 618–631. https://doi.org/10.1111/j.1525-142X.2007.00200.x.
Ronquist, F., & Huelsenbeck, J. (2003). MRBAYES 3: Bayesian phylogenetic inference under mixed models. Bioinformatics, 19, 1572–1574. https://doi.org/10.1093/bioinformatics/btg180.
Schuster, A., Vargas, S., Knapp, I. S., Pomponi, S. A., Toonen, R. J., Erpenbeck, D., & Wörheide, G. (2018). Divergence times in demosponges (Porifera): First insights from new mitogenomes and the inclusion of fossils in a birth-death clock model. BMC Evolutionary Biology, 18(1), 114. https://doi.org/10.1186/s12862-018-1230-1.
Simion, P., Philippe, H., Baurain, D., Jager, M., Richter, D. J., Di Franco, A., et al. (2017). A large and consistent phylogenomic dataset supports sponges as the sister group to all other animals. Current Biology, 27(7), 958–967. https://doi.org/10.1016/j.cub.2017.02.031.
Sogabe, S., Nakanishi, N., & Degnan, B. (2016). The ontogeny of choanocyte chambers during metamorphosis in the demosponge Amphimedon queenslandica. Evodevo, 7, 6. https://doi.org/10.1186/s13227-016-0042-x.
Sokolova, A. M., Pozdnyakov, I. R., Ereskovsky, A. V., & Karpov, S. A. (2019). Kinetid structure in larval and adult stages of the demosponges Haliclona aquaeductus (Haplosclerida) and Halichondria panicea (Suberitida). Zoomorphology, 138(2), 171–184. https://doi.org/10.1007/s00435-019-00437-5.
Sperling, E., Peterson, K., & Pisani, D. (2009). Phylogenetic-signal dissection of nuclear housekeeping genes supports the paraphyly of sponges and the monophyly of Eumetazoa. Molecular Biology and Evolution, 26, 2261–2274.
Stamatakis, A. (2014). RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics, 30(9), 1312–1313. https://doi.org/10.1093/bioinformatics/btu033.
Stephens, K. M., Ereskovsky, A., Lalor, P., & Mccormack, G. P. (2013). Ultrastructure of the ciliated cells of the free-swimming larva, and sessile stages, of the marine sponge Haliclona indistincta (Demospongiae: Haplosclerida). Journal of Morphology, 274(11), 1263–1276. https://doi.org/10.1002/jmor.20177.
Tamm, S. L., & Tamm, S. (2002). Novel bridge of axon-like processes of epithelial cells in the aboral sense organ of ctenophores. Journal of Morphology, 254, 99–120. https://doi.org/10.1002/jmor.10019.
Uriz, M. J., Turon, X., & Becerro, M. A. (2001). Morphology and ultrastructure of the swimming larvae of Crambe crambe (Demospongiae, Poecilosclerida). Invertebrate Biology, 120(4), 295–307. https://doi.org/10.1111/j.1744-7410.2001.tb00039.x.
Uriz, M. J., Turon, X., & Mariani, S. (2008). Ultrastructure and dispersal potential of sponge larvae: Tufted versus evenly ciliated parenchymellae. Marine Ecology, 29, 280–297. https://doi.org/10.1111/j.1439-0485.2008.00229.x.
Usher, K. M., & Ereskovsky, A. V. (2005). Larval development, ultrastructure and metamorphosis in Chondrilla australiensis Carter, 1873 (Demospongiae, Chondrosida, Chondrillidae). Invertebrate Reproduction and Development, 47(1), 51–62. https://doi.org/10.1080/07924259.2005.9652146.
Van Soest, R., Boury-Esnault, N., Hooper, J., Rützler, K., de Voogd, N., Alvarez, B., Hajdu E, Pisera, A., Manconi, R., Schönberg, C., Klautau, M., Kelly, M., Vacelet, J., Dohrmann, M., Díaz, M.-C., Cárdenas, P., Carballo, J., Ríos, P., Downey, R., & Morrow, C. (2020) World Porifera Database. http://www.marinespecies.org/porifera Accessed on 2020-05-29.
Voigt, O., Wülfing, E., & Wörheide, G. (2012). Molecular phylogenetic evaluation of classification and scenarios of character evolution in calcareous sponges (Porifera, class Calcarea). PLoS One, 7(3), e33417. https://doi.org/10.1371/journal.pone.0033417.
Woollacott, R. M., & Pinto, R. L. (1995). Flagellar basal apparatus and its utility in phylogenetic analyses of the Porifera. Journal of Morphology, 226(3), 247–265. https://doi.org/10.1002/jmor.1052260302.
Acknowledgements
The research was supported by the Russian Foundation for Basic Research (projects nos. 18-04-01314 and 19-34-90084). The work of AMS was conducted under the IDB RAS GBRP № 0088-2019-0005. IP was supported by the ZIN RAS research project АААА-А19-119031200042-9. SK was supported by the ZIN RAS research project AAAA-A19-119020690109-2. We thank Research Resource Center for Molecular and Cell Technologies (RRC MCT) at St. Petersburg State University for access to the EM facilities and the Morphology Service of the Mediterranean Institute of Marine and Terrestrial Biodiversity and Ecology (IMBE). We are grateful to N. Kovalchuk for Lubomirskia larvae collection and fixation and Dr. Barry S. C. Leadbeater for editing the language of the text.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Suppl. Fig. 1
Bayesian phylogenetic tree of the sponges based on the 18S dataset. Node support is indicated as branch labels, scale according to GTR + G + I model distances. (PNG 2995 kb)
Suppl. Fig. 2
Maximum likelihood phylogenetic tree of sponges based on the 18S dataset. Node support is indicated as branch labels, scale according to GTR + G + I model distances. (PNG 2386 kb)
Suppl. Fig. 3
(PNG 127 kb)
Suppl. Table 1
Standard Categorical matrix of morphological characters used in the analysis. Polymorphisms are listed separated by the ‘&’ symbol; uncertain states are indicated by the ‘/’ symbol. Clades included in analysis are highlighted in blue. (DOCX 26 kb)
Rights and permissions
About this article
Cite this article
Sokolova, A.M., Pozdnyakov, I.R., Schepetov, D.M. et al. Kinetid in larval cells of Spongillida (Porifera: Demospongiae): tracing the ancestral traits. Org Divers Evol 20, 669–680 (2020). https://doi.org/10.1007/s13127-020-00460-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-020-00460-1