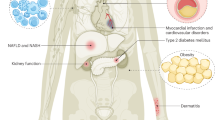
Abstract
Insulin resistance, the most important characteristic of the type 2 diabetes mellitus (T2DM), is mostly caused by impairment in the insulin receptor (IR) signal transduction pathway. Protein tyrosine phosphatase 1B (PTP1B), one of the main negative regulators of the IR signaling pathway, is broadly expressed in various cells and tissues. PTP1B decreases the phosphorylation of the IR resulting in insulin resistance in various tissues. The evidence for the physiological role of PTP1B in regulation of metabolic pathways came from whole-body PTP1B-knockout mice. Whole-body and tissue-specific PTP1B-knockout mice showed improvement in adiposity, insulin resistance, and glucose tolerance. In addition, the key role of PTP1B in the pathogenesis of T2DM and its complications was further investigated in mice models of PTP1B deficient/overexpression. In recent years, targeting PTP1B using PTP1B inhibitors is being considered an attractive target to treat T2DM. PTP1B inhibitors improve the sensitivity of the insulin receptor and have the ability to cure insulin resistance-related diseases. We herein summarized the biological functions of PTP1B in different tissues in vivo and in vitro. We also describe the effectiveness of potent PTP1B inhibitors as pharmaceutical agents to treat T2DM.



Similar content being viewed by others
Abbreviations
- ACC:
-
Acetyl-CoA carboxylase
- AgRP:
-
Agouti-related protein
- AMPK:
-
AMP-activated protein kinase
- BP:
-
Blood pressure
- BCR-ABL:
-
Breakpoint cluster region protein-Abelson murine leukemia
- BAT:
-
Brown adipose tissue
- CLK:
-
CDC-like kinase
- CeA:
-
Central nucleus of the amygdala
- DN:
-
Diabetic nephropathy
- DR:
-
Diabetic retinopathy
- Egr-1:
-
Early growth response 1
- ER:
-
Endoplasmic reticulum
- eNOS:
-
Endothelial nitric oxide synthase
- EGFR:
-
Epidermal growth factor receptor
- FAS:
-
Fatty acid synthase
- FFA:
-
Free fatty acid
- GLUT4:
-
Glucose transporter type 4
- GSIS:
-
Glucose-stimulated insulin secretion
- GK:
-
Goto–Kakizaki
- Grb 14:
-
Growth factor receptor bound protein 14
- HFD:
-
High fat diet
- IR:
-
Insulin receptor
- IRSI:
-
IR substrate 1
- RTK:
-
IR tyrosine kinase
- JAK2/STAT3:
-
Janus kinase 2/signal transducer and activator of transcription 3
- JNK:
-
Jun N-terminal kinase
- MCD:
-
Methionine/choline-deficient diet
- MAPK:
-
Mitogen-activated protein kinase
- MI:
-
Myocardial infarction
- NPY:
-
Neuropeptide Y
- NASH:
-
Nonalcoholic steatohepatitis
- NF-kB:
-
Nuclear factor kappa B
- PI3K:
-
Phosphatidylinositol 3-kinase
- PEPCK:
-
Phosphoenolpyruvate carboxykinase
- PDGF-R:
-
Platelet-derived growth factor receptors
- PTM:
-
Post-translational modification
- POMC:
-
Proopiomelanocortin
- PGI2:
-
Prostaglandin I2
- PKA:
-
Protein kinase A
- Akt:
-
Protein kinase B
- PKC:
-
Protein kinase C
- PTP1B:
-
Protein tyrosine phosphatase
- PTP:
-
Protein tyrosine phosphatases
- ROS:
-
Reactive oxygen species
- SIRT1:
-
Sirtuin 1
- SUMO:
-
Small ubiquitin-related modifier
- SREBP:
-
Sterol regulatory element-binding protein
- TEM:
-
Transendothelial migration
- T2DM:
-
Type 2 diabetes mellitus
- VCAM-1:
-
Vascular adhesion molecule 1
- YB1:
-
Y-box-binding protein 1
References
Surampudi PN, John-Kalarickal J, Fonseca VA (2009) Emerging concepts in the pathophysiology of type 2 diabetes mellitus. Mt Sinai J Med 76(3):216–226
Zaccardi F et al (2015) Pathophysiology of type 1 and type 2 diabetes mellitus: a 90-year perspective. Postgrad Med J 92(1084):63–69
Imperatore G et al (2012) Projections of type 1 and type 2 diabetes burden in the US population aged< 20 years through 2050. Diabetes Care 35(12):2515–2520
Tangvarasittichai S (2015) Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J Diabetes 6(3):456–480
Lebovitz H (2001) Insulin resistance: definition and consequences. Exp Clin Endocrinol Diabetes 109(Suppl 2):S135–S148
Alemzadeh I et al Clinical uses and survival study of free and encapsulated probiotic bacteria in fruit juices: a review. Appl Food Biotechnol 8(3): 161–180
Chang L, Chiang S-H, Saltlel A (2004) Insulin signaling and the regulation of glucose transport. Mol Med 10(7/12):65
Cheng A et al (2002) Coordinated action of protein tyrosine phosphatases in insulin signal transduction. Eur J Biochem 269(4):1050–1059
Xu E, Schwab M, Marette A (2014) Role of protein tyrosine phosphatases in the modulation of insulin signaling and their implication in the pathogenesis of obesity-linked insulin resistance. Rev Endocr Metab Disord 15(1):79–97
Gorgani-Firuzjaee S, Meshkani R (2015) SH2 domain-containing inositol 5-phosphatase (SHIP2) inhibition ameliorates high glucose-induced de-novo lipogenesis and VLDL production through regulating AMPK/mTOR/SREBP1 pathway and ROS production in HepG2 cells. Free Radical Biol Med 89:679–689
Knobler H, Elson A (2014) Metabolic regulation by protein tyrosine phosphatases. J Biomed Res 28(3):157
Egawa K et al (2001) Protein-tyrosine phosphatase-1B negatively regulates insulin signaling in l6 myocytes and Fao hepatoma cells. J Biol Chem 276(13):10207–10211
Rajala RV et al (2010) Enhanced retinal insulin receptor-activated neuroprotective survival signal in mice lacking the protein-tyrosine phosphatase-1B gene. J Biol Chem 285(12):8894–8904
Popov D (2011) Novel protein tyrosine phosphatase 1B inhibitors: interaction requirements for improved intracellular efficacy in type 2 diabetes mellitus and obesity control. Biochem Biophys Res Commun 410(3):377–381
Stuible M, Tremblay ML (2010) In control at the ER: PTP1B and the down-regulation of RTKs by dephosphorylation and endocytosis. Trends Cell Biol 20(11):672–679
Bakke J, Haj FG (2015) Protein-tyrosine phosphatase 1B substrates and metabolic regulation. Semin Cell Dev Biol. 37:58–65
Koren S, Fantus IG (2007) Inhibition of the protein tyrosine phosphatase PTP1B: potential therapy for obesity, insulin resistance and type-2 diabetes mellitus. Best Pract Res Clin Endocrinol Metab 21(4):621–640
Zhao Y et al (2015) The Role of PTP1B O-GlcNAcylation in Hepatic Insulin Resistance. Int J Mol Sci 16(9):22856–22869
Yip S-C, Saha S, Chernoff J (2010) PTP1B: a double agent in metabolism and oncogenesis. Trends Biochem Sci 35(8):442–449
Feldhammer M et al (2013) PTP1B: a simple enzyme for a complex world. Crit Rev Biochem Mol Biol 48(5):430–445
Fukada T, Tonks NK (2001) The reciprocal role of Egr-1 and Sp family proteins in regulation of the PTP1B promoter in response to the p210 Bcr-Abl oncoprotein-tyrosine kinase. J Biol Chem 276(27):25512–25519
Fukada T, Tonks NK (2003) Identification of YB-1 as a regulator of PTP1B expression: implications for regulation of insulin and cytokine signaling. EMBO J 22(3):479–493
Zabolotny JM et al (2008) Protein-tyrosine phosphatase 1B expression is induced by inflammation in vivo. J Biol Chem 283(21):14230–14241
Yu IC et al (2013) Neuronal androgen receptor regulates insulin sensitivity via suppression of hypothalamic NF-κB–mediated PTP1B expression. Diabetes 62(2):411–423
Sun C et al (2007) SIRT1 improves insulin sensitivity under insulin-resistant conditions by repressing PTP1B. Cell Metab 6(4):307–319
Parvaneh L et al (2010) Palmitate and inflammatory state additively induce the expression of PTP1B in muscle cells. Biochem Biophys Res Commun 396(2):467–471
MohammadTaghvaei N et al (2011) Palmitate enhances protein tyrosine phosphatase 1B (PTP1B) gene expression at transcriptional level in C2C12 skeletal muscle cells. Inflammation 34(1):43–48
MohammadTaghvaei N et al (2012) Palmitate-induced PTP1B expression is mediated by ceramide-JNK and nuclear factor κB (NF-κB) activation. Cell Signal 24(10):1964–1970
Lu X et al (2008) PTP1B is a negative regulator of interleukin 4–induced STAT6 signaling. Blood J Am Soc Hematol 112(10):4098–4108
Nasimian A et al (2013) Protein tyrosine phosphatase 1B (PTP1B) modulates palmitate-induced cytokine production in macrophage cells. Inflamm Res 62(2):239–246
Panzhinskiy E, Ren J, Nair S (2013) Protein tyrosine phosphatase 1B and insulin resistance: role of endoplasmic reticulum stress/reactive oxygen species/nuclear factor kappa B axis. PloS one 8(10):e77228
Yang YM et al (2012) Decrease of microRNA-122 causes hepatic insulin resistance by inducing protein tyrosine phosphatase 1B, which is reversed by licorice flavonoid. Hepatology 56(6):2209–2220
Cui S et al (2017) Folic acid supplementation delays atherosclerotic lesion development by modulating MCP1 and VEGF DNA methylation levels in vivo and in vitro. Int J Mol Sci 18(5):990
Zhang X et al (2015) miR-744 enhances type I interferon signaling pathway by targeting PTP1B in primary human renal mesangial cells. Sci Rep 11(5):12987
Christensen LL et al (2013) MiRNA-362-3p induces cell cycle arrest through targeting of E2F1, USF2 and PTPN1 and is associated with recurrence of colorectal cancer. Int J Cancer 133(1):67–78
Karolina DS et al (2011) MicroRNA 144 impairs insulin signaling by inhibiting the expression of insulin receptor substrate 1 in type 2 diabetes mellitus. PloS one 6(8):e22839
Yu M et al (2019) PTP 1B markedly promotes breast cancer progression and is regulated by miR‐193a‐3p. The FEBS J 286(6):1136–1153
Sun F et al (2018) miR-338–3p functions as a tumor suppressor in gastric cancer by targeting PTP1B. Cell Death Dis 9(5)
Ding XQ et al (2015) Curcumin protects against fructose-induced podocyte insulin signaling impairment through upregulation of miR-206. Mol Nutr Food Res 59(12):2355–2370
Yan Y et al (2020) The protective role of MiR-206 in regulating cardiomyocytes apoptosis induced by ischemic injury by targeting PTP1B. Biosci Rep 31;40(1)
Zhang J et al (2019) Upregulating microRNA-203 alleviates myocardial remodeling and cell apoptosis through downregulating protein tyrosine phosphatase 1B in rats with myocardial infarction. J Cardiovasc Pharmacol 74(5):474–481
Hess DT, Stamler JS (2012) Regulation by S-nitrosylation of protein post-translational modification. J Biol Chem 287(7):4411–4418
Lee D et al (2015) UBC9-dependent association between calnexin and protein tyrosine phosphatase 1B (PTP1B) at the endoplasmic reticulum. J Biol Chem 290(9):5725–5738
van Montfort RLM et al (2003) Oxidation state of the active-site cysteine in protein tyrosine phosphatase 1B. Nature 423(6941):773–777
Bourdeau A, Dubé N, Tremblay ML (2005) Cytoplasmic protein tyrosine phosphatases, regulation and function: the roles of PTP1B and TC-PTP. Curr Opin Cell Biol 17(2):203–209
Cortesio CL et al (2008) Calpain 2 and PTP1B function in a novel pathway with Src to regulate invadopodia dynamics and breast cancer cell invasion. J Cell Biol 180(5):957–971
Hsu M-F et al (2016) S-nitrosylation of endogenous protein tyrosine phosphatases in endothelial insulin signaling. Free Radical Biol Med 99:199–213
Taghibiglou C et al (2002) Hepatic very low density lipoprotein-ApoB overproduction is associated with attenuated hepatic insulin signaling and overexpression of protein-tyrosine phosphatase 1B in a fructose-fed hamster model of insulin resistance. J Biol Chem 277(1):793–803
Dubé N, Tremblay ML (2005) Involvement of the small protein tyrosine phosphatases TC-PTP and PTP1B in signal transduction and diseases: from diabetes, obesity to cell cycle, and cancer. Biochim Biophys Acta (BBA) Protein Proteomics 1754(1):108–117
Ahmad F, Goldstein BJ (1995) Increased abundance of specific skeletal muscle protein-tyrosine phosphatases in a genetic model of insulin-resistant obesity and diabetes mellitus. Metabolism 44(9):1175–1184
Stull AJ et al (2012) Skeletal muscle protein tyrosine phosphatase 1B regulates insulin sensitivity in African Americans. Diabetes 61(6):1415–1422
Yamakage H et al (2021) Association of protein tyrosine phosphatase 1B (PTPN1) gene polymorphism with the effects of weight reduction therapy on body weight and glycolipid profiles in obese patients. J Diabetes Investig 12(8):1462–1470
Cheyssac C et al (2006) Analysis of common PTPN1 gene variants in type 2 diabetes, obesity and associated phenotypes in the French population. BMC Med Genet 7(1):1
Palmer ND et al (2004) Association of protein tyrosine phosphatase 1B gene polymorphisms with measures of glucose homeostasis in Hispanic Americans. Diabetes 53(11):3013–3019
Meshkani R et al (2007) 1484insG polymorphism of the PTPN1 gene is associated with insulin resistance in an Iranian population. Arch Med Res 38(5):556–562
Cicirelli MF et al (1990) Microinjection of a protein-tyrosine-phosphatase inhibits insulin action in Xenopus oocytes. Proc Natl Acad Sci 87(14):5514–5518
Ahmad F et al (1995) Osmotic loading of neutralizing antibodies demonstrates a role for protein-tyrosine phosphatase 1B in negative regulation of the insulin action pathway. J Biol Chem 270(35):20503–20508
Chen H et al (1997) Protein-tyrosine phosphatases PTP1B and syp are modulators of insulin-stimulated translocation of GLUT4 in transfected rat adipose cells. J Biol Chem 272(12):8026–8031
Goldstein BJ et al (2000) Tyrosine dephosphorylation and deactivation of insulin receptor substrate-1 by protein-tyrosine phosphatase 1B Possible facilitation by the formation of a ternary complex with the Grb2 adaptor protein. J Biol Chem 275(6):4283–4289
Dadke S, Kusari J, Chernoff J (2000) Down-regulation of insulin signaling by protein-tyrosine phosphatase 1B is mediated by an N-terminal binding region. J Biol Chem 275(31):23642–23647
Salmeen A et al (2000) Molecular basis for the dephosphorylation of the activation segment of the insulin receptor by protein tyrosine phosphatase 1B. Mol Cell 6(6):1401–1412
Bandyopadhyay D et al (1997) Protein-tyrosine phosphatase 1B complexes with the insulin receptor in vivo and is tyrosine-phosphorylated in the presence of insulin. J Biol Chem 272(3):1639–1645
Seely BL et al (1996) Protein tyrosine phosphatase 1B interacts with the activated insulin receptor. Diabetes 45(10):1379–1385
Bence KK (2010) Hepatic PTP1B deficiency: the promise of a treatment for metabolic syndrome? J Clin Metabol Diabetes 1(1):27
Bence KK et al (2006) Neuronal PTP1B regulates body weight, adiposity and leptin action. Nat Med 12(8):917–924
Cheng A et al (2002) Attenuation of leptin action and regulation of obesity by protein tyrosine phosphatase 1B. Dev Cell 2(4):497–503
Klaman LD et al (2000) Increased energy expenditure, decreased adiposity, and tissue-specific insulin sensitivity in protein-tyrosine phosphatase 1B-deficient mice. Mol Cell Biol 20(15):5479–5489
Zabolotny JM et al (2002) PTP1B regulates leptin signal transduction in vivo. Dev Cell 2(4):489–495
Elchebly M et al (1999) Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 283(5407):1544–1548
Cho H (2013) Protein tyrosine phosphatase 1B (PTP1B) and obesity. Vitam Horm 91:405–424
Tiganis T (2013) PTP1B and TCPTP–nonredundant phosphatases in insulin signaling and glucose homeostasis. FEBS J 280(2):445–458
Kennedy BP, Ramachandran C (2000) Protein tyrosine phosphatase-1B in diabetes. Biochem Pharmacol 60(7):877–883
Kushner JA et al (2004) Islet-sparing effects of protein tyrosine phosphatase-1b deficiency delays onset of diabetes in IRS2 knockout mice. Diabetes 53(1):61–66
Gonzalez-Rodriguez A et al (2018) Dual role of protein tyrosine phosphatase 1B in the progression and reversion of non-alcoholic steatohepatitis. Mol Metab 7:132–146
Haj FG et al (2003) Regulation of receptor tyrosine kinase signaling by protein tyrosine phosphatase-1B. J Biol Chem 278(2):739–744
Dubé N, Cheng A, Tremblay ML (2004) The role of protein tyrosine phosphatase 1B in Ras signaling. Proc Natl Acad Sci 101(7):1834–1839
Sangwan V et al (2006) Protein-tyrosine phosphatase 1B deficiency protects against Fas-induced hepatic failure. J Biol Chem 281(1):221–228
Heinonen KM et al (2006) Protein tyrosine phosphatase 1B negatively regulates macrophage development through CSF-1 signaling. Proc Natl Acad Sci USA 103(8):2776–2781
Tsou RC, Bence KK (2012) The genetics of PTPN1 and obesity: insights from mouse models of tissue-specific PTP1B deficiency. J Obes 2012:926857
Delibegovic M et al (2009) Liver-specific deletion of protein-tyrosine phosphatase 1B (PTP1B) improves metabolic syndrome and attenuates diet-induced endoplasmic reticulum stress. Diabetes 58(3):590–599
Le Sommer S et al (2015) Protein tyrosine phosphatase 1B (PTP1B) in the immune system. Inflamm Cell Signal
Fernandez-Ruiz R et al (2014) Protein tyrosine phosphatase-1B (PTP1B) modulates pancreatic β-cell mass. PloS one 9(2):e90344
Liu S et al (2014) Disruption of protein-tyrosine phosphatase 1B expression in the pancreas affects β-cell function. Endocrinology 155(9):3329–3338
Delibegovic M et al (2007) Improved glucose homeostasis in mice with muscle-specific deletion of protein-tyrosine phosphatase 1B. Mol Cell Biol 27(21):7727–7734
Haj FG et al (2005) Liver-specific protein-tyrosine phosphatase 1B (PTP1B) re-expression alters glucose homeostasis of PTP1B–/–mice. J Biol Chem 280(15):15038–15046
Nieto-Vazquez I et al (2007) Protein–tyrosine phosphatase 1B–deficient myocytes show increased insulin sensitivity and protection against tumor necrosis factor-α–induced insulin resistance. Diabetes 56(2):404–413
Lorenzo M et al (2008) Insulin resistance induced by tumor necrosis factor-α in myocytes and brown adipocytes. J Anim Sci 86(14_suppl):E94–E104
Taheripak G et al (2013) Protein tyrosine phosphatase 1B inhibition ameliorates palmitate-induced mitochondrial dysfunction and apoptosis in skeletal muscle cells. Free Radical Biol Med 65:1435–1446
Owen C et al (2012) The physiological and molecular consequences of adipocyte-specific protein tyrosine phosphatase 1B deletion in C57BL/6 mice. Physiol Soc 69(8):156–182
Owen C et al (2012) Adipocyte-specific protein tyrosine phosphatase 1B deletion increases lipogenesis, adipocyte cell size and is a minor regulator of glucose homeostasis. PloS one 7(2):e32700
Picardi PK et al (2008) Reduction of hypothalamic protein tyrosine phosphatase improves insulin and leptin resistance in diet-induced obese rats. Endocrinology 149(8):3870–3880
Aberdein N et al (2018) Role of PTP1B in POMC neurons during chronic high-fat diet: sex differences in regulation of liver lipids and glucose tolerance. Am J Physiol Regul Integr Comp Physiol 314(3):R478-r488
Tsou RC et al (2014) Improved metabolic phenotype of hypothalamic PTP1B-deficiency is dependent upon the leptin receptor. Mol Metab 3(3):301–312
Xue B et al (2009) Neuronal protein tyrosine phosphatase 1B deficiency results in inhibition of hypothalamic AMPK and isoform-specific activation of AMPK in peripheral tissues. Mol Cell Biol 29(16):4563–4573
Mendes NF et al (2017) Knocking down amygdalar PTP1B in diet-induced obese rats improves insulin signaling/action, decreases adiposity and may alter anxiety behavior. Metab 70:1–11
Bruder-Nascimento T et al (2015) Deletion of protein tyrosine phosphatase 1b in proopiomelanocortin neurons reduces neurogenic control of blood pressure and protects mice from leptin-and sympatho-mediated hypertension. Pharmacol Res 102:235–244
Medgyesi D et al (2014) The protein tyrosine phosphatase PTP1B is a negative regulator of CD40 and BAFF-R signaling and controls B cell autoimmunity. J Exp Med 211(3):427–440
Grant L et al (2013) Myeloid-cell protein tyrosine phosphatase-1B deficiency in mice protects against high-fat diet and lipopolysaccharide induced inflammation, hyperinsulinemia and endotoxemia through an IL10 STAT3 dependent mechanism. Diabetes 63(2):456-70
Thompson D et al (2017) Myeloid protein tyrosine phosphatase 1B (PTP1B) deficiency protects against atherosclerotic plaque formation in the ApoE(-/-) mouse model of atherosclerosis with alterations in IL10/AMPKalpha pathway. Mol Metab 6(8):845–853
Panzhinskiy E, Ren J, Nair S (2013) Pharmacological inhibition of protein tyrosine phosphatase 1B: a promising strategy for the treatment of obesity and type 2 diabetes mellitus. Curr Med Chem 20(21):2609–2625
Krishnan N et al (2018) A potent, selective, and orally bioavailable inhibitor of the protein-tyrosine phosphatase PTP1B improves insulin and leptin signaling in animal models. J Biol Chem 293(5):1517–1525
Tang Y et al (2018) Novel benzamido derivatives as PTP1B inhibitors with anti-hyperglycemic and lipid-lowering efficacy. Acta Pharm Sin B 8(6):919–932
Yang Z et al (2018) A novel PTP1B inhibitor extracted from Ganoderma lucidum ameliorates insulin resistance by regulating IRS1-GLUT4 cascades in the insulin signaling pathway. Food Funct 9(1):397–406
Lantz KA et al (2010) Inhibition of PTP1B by trodusquemine (MSI-1436) causes fat-specific weight loss in diet-induced obese mice. Obesity 18(8):1516–1523
Forbes JM, Cooper ME (2013) Mechanisms of diabetic complications. Physiol Rev 93(1):137–188
Potenza MA, Addabbo F, Montagnani M (2009) Vascular actions of insulin with implications for endothelial dysfunction. Am J Physiol Endocrinol Metab 297(3):E568–E577
Mukai Y et al (2007) Phosphatidylinositol 3-kinase/protein kinase Akt negatively regulates plasminogen activator inhibitor type 1 expression in vascular endothelial cells. Am J Physiol Heart Circ Physiol 292(4):H1937–H1942
Montagnani M et al (2002) Inhibition of phosphatidylinositol 3-kinase enhances mitogenic actions of insulin in endothelial cells. J Biol Chem 277(3):1794–1799
Potenza MA et al (2005) Insulin resistance in spontaneously hypertensive rats is associated with endothelial dysfunction characterized by imbalance between NO and ET-1 production. Am J Physiol Heart Circ Physiol 289(2):H813–H822
Eringa EC et al (2007) Selective resistance to vasoactive effects of insulin in muscle resistance arteries of obese Zucker (fa/fa) rats. Am J Physiol Endocrinol Metab 293(5):E1134–E1139
Jiang ZY et al (1999) Characterization of selective resistance to insulin signaling in the vasculature of obese Zucker (fa/fa) rats. J Clin Investig 104(4):447–457
Kuboki K et al (2000) Regulation of endothelial constitutive nitric oxide synthase gene expression in endothelial cells and in vivo a specific vascular action of insulin. Circulation 101(6):676–681
Zecchin HG et al (2007) Defective insulin and acetylcholine induction of endothelial cell–nitric oxide synthase through insulin receptor substrate/Akt signaling pathway in aorta of obese rats. Diabetes 56(4):1014–1024
Ketsawatsomkron P, Stepp DW, de Chantemèle EJB (2013) PTP1B in obesity-related cardiovascular function, in Protein Tyrosine Phosphatase Control of Metabolism. Springer: New York, NY, USA, pp 129–145
Herre DJ et al (2015) Deletion of protein tyrosine phosphatase 1B (PTP1B) enhances endothelial cyclooxygenase 2 expression and protects mice from type 1 diabetes-induced endothelial dysfunction. PloS one 10(5):e0126866
Ali MI et al (2009) Deletion of protein tyrosine phosphatase 1b improves peripheral insulin resistance and vascular function in obese, leptin-resistant mice via reduced oxidant tone. Circ Res 105(10):1013–1022
Agouni A et al (2014) Hepatic protein tyrosine phosphatase 1B (PTP1B) deficiency protects against obesity-induced endothelial dysfunction. Biochem Pharmacol 92(4):607–617
Legeay S et al (2020) Selective deficiency in endothelial PTP1B protects from diabetes and endoplasmic reticulum stress-associated endothelial dysfunction via preventing endothelial cell apoptosis. Biomed Pharmacother 127:110200
Fernandez-Borja M, van Buul JD, Hordijk PL (2010) The regulation of leukocyte transendothelial migration by endothelial signaling events. Cardiovasc Res 1;86(2):202–210
Maupoint J et al (2016) Selective vascular endothelial protection reduces cardiac dysfunction in chronic heart failure. Circ Heart Fail 9(4):e002895
Gogiraju R et al (2016) Endothelial deletion of protein tyrosine phosphatase-1B protects against pressure overload-induced heart failure in mice. Cardiovasc Res 111(3):204–216
Gomez E et al (2012) Reduction of heart failure by pharmacological inhibition or gene deletion of protein tyrosine phosphatase 1B. J Mol Cell Cardiol 52(6):1257–1264
Coquerel D et al (2014) Gene deletion of protein tyrosine phosphatase 1B protects against sepsis-induced cardiovascular dysfunction and mortality. Arterioscler Thromb Vasc Biol 34(5):1032–1044
Kandadi MR et al (2012) Protein tyrosine phosphatase 1B knockout attenuates obesity-induced cardiomyopathy. Circulation 126(Suppl 21):A15595–A15595
Besnier M et al (2014) Enhanced angiogenesis and increased cardiac perfusion after myocardial infarction in protein tyrosine phosphatase 1B-deficient mice. FASEB J 28(8):3351–3361
DeFronzo RA (2010) Insulin resistance, lipotoxicity, type 2 diabetes and atherosclerosis: the missing links. The Claude Bernard Lecture 2009. Diabetologia 53(7):1270–1287
Pu Q et al (2009) Chronic insulin treatment suppresses PTP1B function, induces increased PDGF signaling, and amplifies neointima formation in the balloon-injured rat artery. Am J Physiol Heart Circ Physiol 296(1):H132–H139
Chang Y (2011) A central role of PTP1B in hyperinsulinemia-enhanced IL-6 signaling in dedifferentiated vascular smooth muscle cells. J Diabetes Metab 2:1–7
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet 10;376(9735):124–136
Wan T-T et al (2015) Recent advances in understanding the biochemical and molecular mechanism of diabetic retinopathy. Biomed Pharmacother 74:145–147
Robinson R et al (2012) Update on animal models of diabetic retinopathy: from molecular approaches to mice and higher mammals. Dis Model Mech 5(4):444–456
Rajala RVS et al (2009) Diabetes reduces autophosphorylation of retinal insulin receptor and increases protein-tyrosine phosphatase-1B activity. Invest Ophthalmol Vis Sci 50(3):1033–1040
Basavarajappa DK, Gupta VK, Rajala RVS (2012) Protein tyrosine phosphatase 1B: a novel molecular target for retinal degenerative diseases, in Retinal Degenerative Diseases. Adv Exp Med Biol. 723:829–834
Rajala A et al (2008) Loss of neuroprotective survival signal in mice lacking insulin receptor gene in rod photoreceptor cells. J Biol Chem 283(28):19781–19792
Song J et al (2003) Axons guided by insulin receptor in Drosophila visual system. Science 300(5618):502–505
Barber AJ et al (2001) Insulin rescues retinal neurons from apoptosis by a phosphatidylinositol 3-kinase/Akt-mediated mechanism that reduces the activation of caspase-3. J Biol Chem 276(35):32814–32821
Díaz B et al (1999) Apoptotic cell death of proliferating neuroepithelial cells in the embryonic retina is prevented by insulin. Eur J Neurosci 11(5):1624–1632
Reiter CEN et al (2006) Diabetes reduces basal retinal insulin receptor signaling reversal with systemic and local insulin. Diabetes 55(4):1148–1156
Barber AJ et al (1998) Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Investig 102(4):783
Rajala RVS et al (2013) Spatial and temporal aspects and the interplay of Grb14 and protein tyrosine phosphatase-1B on the insulin receptor phosphorylation. Cell Commun Signal 11(1):1
Dilly AK, Rajala RV (2008) Insulin growth factor 1 receptor/PI3K/AKT survival pathway in outer segment membranes of rod photoreceptors. Invest Ophthalmol Vis Sci 49(11):4765–4773
Arroba AI et al (2013) Loss of protein tyrosine phosphatase 1B increases IGF-I receptor tyrosine phosphorylation but does not rescue retinal defects in IRS2-deficient mice increased IGF-IR phosphorylation in PTP1B−/− retina. Invest Ophthalmol Vis Sci 54(6):4215–4225
Arora MK, Singh UK (2013) Molecular mechanisms in the pathogenesis of diabetic nephropathy: an update. Vascul Pharmacol 58(4):259–271
Sun Y-M et al (2013) Recent advances in understanding the biochemical and molecular mechanism of diabetic nephropathy. Biochem Biophys Res Commun 433(4):359–361
Lay AC, Coward RJ (2018) The evolving importance of insulin signaling in podocyte health and disease. Front Endocrinol 9:693
Kim EY, Anderson M, Dryer SE (2012) Insulin increases surface expression of TRPC6 channels in podocytes: role of NADPH oxidases and reactive oxygen species. Am J Physiol Renal Physiol 302(3):F298–F307
Kim EY, Dryer SE (2011) Effects of insulin and high glucose on mobilization of slo1 BKCa channels in podocytes. J Cell Physiol 226(9):2307–2315
Musso C et al (2006) Spectrum of renal diseases associated with extreme forms of insulin resistance. Clin J Am Soc Nephrol 1(4):616–622
Guzman J et al (2014) Podocyte-specific GLUT4-deficient mice have fewer and larger podocytes and are protected from diabetic nephropathy. Diabetes 63(2):701–714
Meran S, Steadman R (2011) Fibroblasts and myofibroblasts in renal fibrosis. Int J Exp Pathol 92(3):158–167
Ponnusamy M, Ma L, Zhuang S (2013) Necrotic renal epithelial cell inhibits renal interstitial fibroblast activation: role of protein tyrosine phosphatase 1B. Am J Physiol Renal Physiol 304(6):F698–F709
Kumagai T et al (2014) Protein tyrosine phosphatase 1B inhibition protects against podocyte injury and proteinuria. Am J Pathol 184(8):2211–2224
Tiganis T, Bennett AM (2007) Protein tyrosine phosphatase function: the substrate perspective. Biochem J 402(1):1–15
Verma R et al (2006) Nephrin ectodomain engagement results in Src kinase activation, nephrin phosphorylation, Nck recruitment, and actin polymerization. J Clin Investig 116(5):1346–1359
Li H et al (2006) Rat nephrin modulates cell morphology via the adaptor protein Nck. Biochem Biophys Res Commun 349(1):310–316
Jones N et al (2006) Nck adaptor proteins link nephrin to the actin cytoskeleton of kidney podocytes. Nature 440(7085):818–823
Aoudjit L et al (2011) Podocyte protein, nephrin, is a substrate of protein tyrosine phosphatase 1B. J Signal Transduct 2011:376543
Ito Y et al (2017) Protein tyrosine phosphatase 1B deficiency in podocytes mitigates hyperglycemia-induced renal injury. Metabolism 76:56–69
Garner KL et al (2018) Enhanced insulin receptor, but not PI3K, signalling protects podocytes from ER stress. Sci Rep 8(1):3902
Figueiredo A, Leal EC, Carvalho E (2020) Protein tyrosine phosphatase 1B inhibition as a potential therapeutic target for chronic wounds in diabetes. Pharmacol Res 159:104977
Zhang J et al (2015) Protein tyrosine phosphatase 1B impairs diabetic wound healing through vascular endothelial growth factor receptor 2 dephosphorylation. Arterioscler Thromb Vasc Biol 35(1):163–174
Dinh T et al (2012) Mechanisms involved in the development and healing of diabetic foot ulceration. Diabetes 61(11):2937–2947
Kakazu A, Sharma G, Bazan HE (2008) Association of protein tyrosine phosphatases (PTPs)-1B with c-Met receptor and modulation of corneal epithelial wound healing. Invest Ophthalmol Vis Sci 49(7):2927–2935
Zabolotny J et al (2011) Protein tyrosine phophatase 1B (PTP1B) deficiency promotes wound healing. in Diabetologia. SPRINGER 233 SPRING ST, New York
Sharma B et al (2020) Recent advance on PTP1B inhibitors and their biomedical applications. Eur J Med Chem 1(199):112376
Thareja S et al (2012) Protein tyrosine phosphatase 1B inhibitors: a molecular level legitimate approach for the management of diabetes mellitus. Med Res Rev 32(3):459–517
Tamrakar AK, Maurya CK, Rai AK (2014) PTP1B inhibitors for type 2 diabetes treatment: a patent review (2011–2014). Expert Opin Ther Pat 24(10):1101–1115
Ottanà R et al (2009) 5-Arylidene-2-phenylimino-4-thiazolidinones as PTP1B and LMW-PTP inhibitors. Bioorg Med Chem 17(5):1928–1937
Kumar R et al (2010) Probing interaction requirements in PTP1B inhibitors: a comparative molecular dynamics study. J Chem Inf Model 50(6):1147–1158
Liu J-Z et al (2013) Discovery of novel PTP1B inhibitors via pharmacophore-oriented scaffold hopping from Ertiprotafib. Bioorg Med Chem Lett 23(23):6217–6222
Kumar GS, Page R, Peti W (2020) The mode of action of the Protein tyrosine phosphatase 1B inhibitor Ertiprotafib. Plos one 15(10):e0240044
Qin Z et al (2015) Functional properties of Claramine: a novel PTP1B inhibitor and insulin-mimetic compound. Biochem Biophys Res Commun 458(1):21–27
Grijalvo S, Aviñó A, Eritja R (2014) Oligonucleotide delivery: a patent review (2010–2013). Expert Opin Ther Pat 24(7):801–819
González-Rodríguez Á et al (2010) Inhibition of PTP1B restores IRS1-mediated hepatic insulin signaling in IRS2-deficient mice. Diabetes 59(3):588–599
Revuelta-Cervantes J et al (2011) Protein tyrosine phosphatase 1B (PTP1B) deficiency accelerates hepatic regeneration in mice. Am J Pathol 178(4):1591–1604
Author information
Authors and Affiliations
Contributions
RM designed the study and revised the manuscript. MT wrote the paper. HH and ZA made the literature selection. RB participates in drafting. SG-F revised the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Research involving human participants and/or animals
NA.
Informed consent
NA.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key points
• PTP1B serves as a negative regulator of the insulin signaling pathway.
• PTP1B upregulation plays critical role in the T2DM and its complications.
• PTP1B inhibition is a potential therapeutic approach for T2DM treatment.
• It remains a challenge to identify appropriate inhibitors for PTP1B.
Rights and permissions
About this article
Cite this article
Teimouri, M., Hosseini, H., ArabSadeghabadi, Z. et al. The role of protein tyrosine phosphatase 1B (PTP1B) in the pathogenesis of type 2 diabetes mellitus and its complications. J Physiol Biochem 78, 307–322 (2022). https://doi.org/10.1007/s13105-021-00860-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-021-00860-7