Abstract
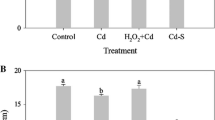
We investigated changes in reactive oxygen species (ROS) and antioxidant levels in rice (Oryza sativa L. cv ‘Dongjin’) seedlings treated with toxic cadmium (Cd) and/or sulfur (S). Exposure of rice seedlings to 30 μM Cd inhibited plant growth and resulted in increased levels of superoxide, hydrogen peroxide, and malondialdehyde (MDA), and induced Cd uptake by the roots, stems, and leaves. Application of S to Cd-stressed seedlings ameliorated Cd-induced oxidative stress by increasing the capacity of the glutathione (GSH)-ascorbate (AsA) cycle, promoting S assimilation by increasing cysteine, GSH, and AsA content in treated plants, and decreasing Cd transfer from the roots to the stems and leaves. Therefore, these results indicate that S application represents a viable strategy of alleviating Cd-induced growth inhibition and oxidative damage by restricting Cd translocation from the roots to the stems and leaves, thereby maintaining sufficient levels of GSH and AsA by sustaining homeostasis of the GSH-AsA cycle.
Similar content being viewed by others
Abbreviations
- AsA:
-
ascorbate
- Cys:
-
cysteine
- GSH:
-
glutathione
- MDA:
-
malondialdehyde
- ROS:
-
reactive oxygen species
References
Anjum NA, Umar S, Ahmad A, Iqbal M, Khan NA. 2008. Sulphur protects mustard (Brassica campestris L.) from cadmium toxicity by improving leaf ascorbate and glutathione. Plant Growth Regul. 54: 271–279
Astolfi S, Zuchi S, Neumann G, Cesco S, di Toppi LS, Pinton R. 2012. Response of barley plants to Fe deficiency and Cd contamination as affected by S starvation. J. Exp. Bot. 63: 1241–1250
Barceló J, Vázquez MD, Poschenrieder C. 1988. Structural and ultrastructural disorders in cadmium-treated bush bean plants (Phaseolus vulgaris L.). New Phytol. 108: 37–49
Buege JA, Aust SD. 1978. Microsomal lipid peroxidation. Methods Enzymol. 52: 302–310
Cai Y, Lin L, Cheng W, Zhang G, Wu F. 2010. Genotypic dependent effect of exogenous glutathione on Cd-induced changes in cadmium and mineral uptake and accumulation in rice seedlings (Oryza sativa). Plant Soil Environ. 56: 516–525
Elstner EF, Heupel A. 1976. Inhibition of nitrite formation from hydroxylammonium-chloride: a simple assay for superoxide dismutase. Anal. Biochem. 70: 616–620
Gaitonde MK. 1967. A spectrophotometric method for the direct determination of cysteine in the presence of other naturally occurring amino acids. Biochem. J. 104: 627–633
Gill SS, Tuteja N. 2010. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 48: 909–930
Gill SS, Tuteja N. 2011. Cadmium stress tolerance in crop plants probing the role of sulfur. Plant Signal. Behav. 6: 215–222
Greger M, Löfstedt M. 2004. Comparison of uptake and distribution of cadmium in different cultivars of bread and durum wheat. Crop Sci. 44: 501–507
Harada E, Yamaguchi Y, Koizumi N, Hiroshi S. 2002. Cadmium stress induces production of thiol compounds and transcripts for enzymes involved in sulfur assimilation pathways in Arabidopsis. J. Plant Physiol. 159: 445–448
Herschbach C, Rennenberg H. 1994. Influence of glutathione (GSH) on net uptake of sulfate and sulfate transport in tobacco plants. J. Exp. Bot. 45: 1069–1076
Jan AT, Azam M, Siddiqui K, Ali A, Choi I, Haq QM. 2015. Heavy metals and human health: mechanistic insight into toxicity and counter defense system of antioxidants. Int. J. Mol. Sci. 16: 29592–29630
Jana S, Choudhuri MA. 1982. Glycolate metabolism of three submerged aquatic angiosperms during aging. Aquat. Bot. 12: 345–354
Jung HI, Chae MJ, Kim SJ, Kong MS, Kang SS, Lee DB, Ju HJ, Kim YH. 2015. Effects of cadmium and arsenic on physiological responses and copper and zinc homeostasis of rice. Kor. J. Soil Sci. Fert. 48: 397–403
Kamachi K, Yamaya T, Mae T, Ojima K. 1991. A role for glutamine synthetase in the remobilization of leaf nitrogen during natural senescence in rice leaves. Plant Physiol. 96: 411–417
Kerk NM, Feldman LJ. 1995. A biochemical model for the initiation and maintenance of the quiescent center: implications for organization of root meristems. Development 121: 2825–2833
Kirkham MB. 2006. Cadmium in plants on polluted soils: effects of soil factors, hyperaccumulation and amendments. Geoderma 37: 19–32
Lagriffoul A, Mocquot B, Mench M, Vangronsveld J. 1998. Cadmium toxicity effects on growth, mineral and chlorophyll contents, and activities of stress related enzymes in young maize plants (Zea mays L.). Plant Soil 200: 241–250
Lappartient AG, Touraine B. 1996. Demand-driven control of root ATP sulfurylase activity and sulfate uptake in intact canola. Plant Physiol. 111: 147–157
Law MY, Charles SA, Halliwell B. 1983. Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts: the effect of hydrogen peroxide and of paraquat. Biochem. J. 210: 899–903
Liang T, Ding H, Wang G, Kang J, Pang H, Lv J. 2016. Sulfur decreases cadmium translocation and enhances cadmium tolerance by promoting sulfur assimilation and glutathione metabolism in Brassica chinensis L. Ecotoxicol. Environ. Saf. 124: 129–137
Luwe M. 1996. Antioxidants in the apoplast and symplast of beech (Fagus sylvatica L.) leaves: seasonal variations and response to changing ozone concentrations in air. Plant Cell Environ. 19: 321–328
Meister A, Anderson ME. 1983. Glutathione. Annu. Rev. Biochem. 52: 711–760
Meuwly P, Rauser WE. 1992. Alteration of thiol pools in roots and shoots of maize seedlings exposed to cadmium: adaptation and developmental cost. Plant Physiol. 99: 8–15
Nagajyoti PC, Lee KD, Sreekanth TVM. 2010. Heavy metals, occurrence and toxicity for plants: a review. Environ. Chem. Lett. 8: 199–216
Ranieri A, D’ Urso G, Nali C, Lorenzini G, Soldatini GG. 1996. Ozone stimulates apoplastic antioxidant systems in pumpkin leaves. Physiol. Plant. 97: 381–387
Salt DE, Blaylock M, Kumar NPBA, Dushenkov V, Ensley BD, Chet I, Raskin I. 1995. Phytoremediation: a novel strategy for the removal of toxic metals from the environment using plants. Biotechnology 13: 468–474
Singh RP, Agrawal M. 2010. Biochemical and physiological responses of rice (Oryza sativa L.) grown on different sewage sludge amendments rates. Bull. Environ. Contam. Toxicol. 84: 606–612
Singh S, Eapen S, D’ Souza SF. 2006. Cadmium accumulation and its influence on lipid peroxidation and antioxidative system in an aquatic plant, Bacopa monnieri L. Chemosphere 62: 233–246
Talukdar D. 2012. Exogenous calcium alleviates the impact of cadmium-induced oxidative stress in Lens culinaris medic. seedlings through modulation of antioxidant enzyme activities. J. Crop Sci. Biotechnol. 15: 325–334
Vassilev A, Yordanov I, Tsonev T. 1997. Effects of Cd2+ on the physiological state and photosynthetic activity of young barley plants. Photosynthetica 34: 293–302
Xu X, Liu C, Zhao X, Li R, Deng W. 2014. Involvement of an antioxidant defense system in the adaptive response to cadmium in maize seedlings (Zea mays L.). Bull. Environ. Contam. Toxicol. 93: 618–624
Yan YF, Choi DH, Kim DS, Lee BW. 2010a. Genotypic variation of cadmium accumulation and distribution in rice. J. Crop Sci. Biotechnol. 13: 69–73
Yan YF, Choi DH, Kim DS, Lee BW. 2010b. Absorption, translocation, and remobilization of cadmium supplied at different growth stages of rice. J. Crop Sci. Biotechnol. 13: 113–119
Zhang B, Shang S, Zhang H, Jabeen Z, Zhang G. 2013. Sodium chloride enhances cadmium tolerance through reducing cadmium accumulation and increasing anti-oxidative enzyme activity in tobacco. Environ. Toxicol. Chem. 32: 1420–1425
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jung, Hi., Lee, BR., Chae, MJ. et al. Sulfur alleviates cadmium toxicity in rice (Oryza sativa L.) seedlings by altering antioxidant levels. J. Crop Sci. Biotechnol. 20, 213–220 (2017). https://doi.org/10.1007/s12892-017-0072-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-017-0072-0