Abstract
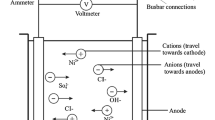
Ni-Co alloys with different compositions were electrodeposited from additive-free Watt’s type electrolytes utilizing mechanical-assisted electrodeposition. SEM, XRD, microhardness, and potentiodynamic polarization tests were used to investigate the effects of mechanical attrition speed and electrolyte composition on electrodeposits. Increased attrition speed resulted in reduced Co content in films with fcc crystal structure and raised Co content in hcp crystal structures. The microhardness values raised significantly, while the corrosion resistance was reduced by applying mechanical attrition. In order to choose the best electrodeposit, the C factor—a ratio of hardness to corrosion current density—is introduced. Based on the C factor, the deposits with 4 at% (Co/Co + Ni) in electrolyte and attrition speed of 600 rpm were selected as the best condition, which resulted in an alloy with a smooth and reflective surface containing 32 at% (Co/Co + Ni) with a hardness of 480 HV and corrosion current density close to that of pure nickel electrodeposits.






Similar content being viewed by others
References
Karimzadeh A, Aliofkhazraei M, and Walsh F C, Surf Coat Technol 372 (2019) 463. https://doi.org/10.1016/j.surfcoat.2019.04.079
Mishra R, and Balasubramaniam R, Corros Sci 46 (2004) 3019. https://doi.org/10.1016/j.corsci.2004.04.007
Godon A, Creus J, Cohendoz S, Conforto E, and Savall C, Scr Mater 62 (2010) 403. https://doi.org/10.1016/j.scriptamat.2009.11.038
Ebrahimi F, Bourne G R, Kelly M S, and Matthews T E, Nanostructured Mater 11 (1999) 343. https://doi.org/10.1016/S0965-9773(99)00050-1
Dalla Torre F, Swygenhoven H V, and Victoria M, Acta Mater 50 (2002) 3957. https://doi.org/10.1016/S1359-6454(02)00198-2
Wasekar N P, Haridoss P, Seshadri S K, and Sundararajan G, Wear 296 (2012) 536. https://doi.org/10.1016/j.wear.2012.08.003
Rashidi A, and Amadeh A, Surf Coat Technol 204 (2009) 353. https://doi.org/10.1016/j.surfcoat.2009.07.036
Pavlatou E A, Raptakis M, and Spyrellis N, Surf Coat Technol 201 (2007) 4571. https://doi.org/10.1016/j.surfcoat.2006.09.113
El-Sherik A, and Erb U, J Mater Sci 30 (1995) 5743. https://doi.org/10.1007/BF00356715
Das P, Samantaray B, Dolai S, Seshu K S, and Gollapudi S, Metall Mater Trans A 52 (2021) 1913. https://doi.org/10.1007/s11661-021-06202-y
Lokhande A C, and Bagi J S, Surf Coat Technol 258 (2014) 225. https://doi.org/10.1016/j.surfcoat.2014.09.023
Wang Y, Cheng S, Wei Q M, Ma E, and Hamza A, Scr Mater 51 (2004) 1023. https://doi.org/10.1016/j.scriptamat.2004.08.015
Zhang X, Fujita T, Pan D, Yu J S, and Chen M W, Mater Sci Eng A 527 (2010) 2297. https://doi.org/10.1016/j.msea.2009.12.005
Hu Y, Deb S, Li D, and Huang Q, Electrochim Acta 368 (2021) 137594. https://doi.org/10.1016/j.electacta.2020.137594
Wu B, Xu B, Zhang B, and Dong S, Surf Coat Technol 201 (2007) 5758. https://doi.org/10.1016/j.surfcoat.2006.10.013
Lv B, Hu Z, Wang X, and Xu B, Surf Coat Technol 270 (2015) 123. https://doi.org/10.1016/j.surfcoat.2015.03.012
Ping Z, Ping Z, He Y, Gu C, and Zhang T, Surf Coat Technol 202 (2008) 6023. https://doi.org/10.1016/j.surfcoat.2008.06.183
Ping Z, He Y, Gu C, and Zhang T, J Appl Electrochem 39 (2009) 6879. https://doi.org/10.1007/s10800-008-9734-9
Idris J, Christian C, and Gaius E, J Nanomater 2013 (2013) 12. https://doi.org/10.1155/2013/841260
Qiao G, Jing T, Wang N, Gao Y, and Wang W, Electrochim Acta 51 (2005) 85. https://doi.org/10.1016/j.electacta.2005.03.050
Liu C, Su F, and Liang J, Surf Coat Technol 292 (2016) 37. https://doi.org/10.1016/j.surfcoat.2016.03.027
He Y, Fu H F, Li X G, and Gao W, Scr Mater 58 (2008) 504. https://doi.org/10.1016/j.scriptamat.2007.10.051
Eisner S, Trans IMF 51 (1973) 13. https://doi.org/10.1080/00202967.1973.11870257
Ning Z, He Y, and Gao W, Surf Coat Technol 202 (2008) 2139. https://doi.org/10.1016/j.surfcoat.2007.08.062
Zhaoxia P, YUAN J, HE Y, and LI X, Acta Metall Sin Engl Lett 22 (2009) 225. https://doi.org/10.1016/S1006-7191(08)60093-9
Oriňáková R, Turoňová A, Kladeková D, Gálová M, and Smith R M, J Appl Electrochem 36 (2006) 957. https://doi.org/10.1007/s10800-006-9162-7
Brenner A, Electrodeposition of Alloys: Practical and specific information, Vol 2, Elsevier (1963), ISBN: 978–1–4831–9807–1
Li Y, Shan L, Sui Y, Qi J, and Liu J, J Mater Sci: Mater Electron 30 (2019) 13360. https://doi.org/10.1007/s10854-019-01703-4
Zhang H, Lv Y, Wu X, Guo J, and Jia D, Chem Eng J 431 (2022) 133233. https://doi.org/10.1016/j.cej.2021.133233
Matlosz M, J Electrochem Soc 140 (1993) 2272. https://doi.org/10.1149/1.2220807
Zech N, Podlaha E, and Landolt D, J Electrochem Soc 146 (1999) 2886. https://doi.org/10.1149/1.1392024
Grande W C, and Talbot J B, J Electrochem Soc 140 (1993) 675. https://doi.org/10.1149/1.2056141
Chen Y, Yang H, Feng H, Yang P, and Shu B, Today Commun 35 (2023) 106058. https://doi.org/10.1016/j.mtcomm.2023.106058
You Y H, Gu C D, Wang X L, and Tu J P, Surf Coat Technol 206 (2012) 3632. https://doi.org/10.1016/j.surfcoat.2012.03.001
Bakhit B, and Akbari A, J Coat Technol Res 10 (2013) 285. https://doi.org/10.1007/s11998-012-9437-3
Hu C, and Bai A, J Electrochem Soc 149 (2002) 615. https://doi.org/10.1149/1.1511753
Golodnitsky D, Gudin N, and Volyanuk G, J Electrochem Soc 147 (2000) 4156. https://doi.org/10.1149/1.1394034
Bai A, and Hu C, Electrochim Acta 47 (2002) 3447. https://doi.org/10.1016/S0013-4686(02)00281-5
Go´ mez E, Ramirez J, and Valle´s E, J Appl Electrochem 28 (1998) 71. https://doi.org/10.1023/A:1003201919054
Liu P, Chen D, Wang Q, Xu P, Long M, and Duan H, J Phys Chem Solids 137 (2020) 1. https://doi.org/10.1016/j.jpcs.2019.109194
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hajizadeh, K., Chianeh, V.A. Influence of Mechanical Attrition on Protective Properties of Anomalous Electrodeposited Ni-Co Alloys. Trans Indian Inst Met 77, 607–614 (2024). https://doi.org/10.1007/s12666-023-03148-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-023-03148-0