Abstract
Background
In the framework of cluster expansion—cluster variation methods (CE - CVM), the equilibrium values of the microscopic state variables called correlation functions (CFs) can be obtained by minimization of the Helmholtz energy. This involves solving the nonlinear equilibrium equations using numerical techniques. As an exception, Guggenheim obtained an analytical solution for the pair CF in the pair approximation of quasi-chemical theory for a binary alloy.
Results
In this communication, analytical solutions for the CFs for the case of exclusive second neighbour pair interactions in binary A2 & B32 phases and A1 & L11 phases respectively under tetrahedron and tetrahedron–octahedron approximations of CVM are obtained as functions of the corresponding energy coefficient, temperature, and composition.
Results obtained by results in the previous section and a few new results
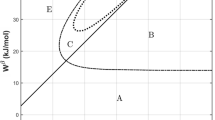
Further, analytical expressions for the phase boundaries in phase separating and ordering systems have been obtained. Apart from these, thermodynamic quantities such as heat capacities have also been evaluated. In addition, the effect of composition and temperature dependence of the energy coefficient on the phase boundary is also discussed.
Application
For the selected composition, the solution of CF reduces to a simple rational function. Taking this as a guide, the coefficients of the polynomials used to approximate the CFs are expressed as rational functions, which was discussed elsewhere.


















Similar content being viewed by others
References
Kikuchi R, Phys Rev 81 (1951) 988. https://doi.org/10.1103/PhysRev.81.988.
Sanchez J M, Ducastelle F, Gratias D, Phys A Stat Mech Appl 128 (1984) 334. https://doi.org/10.1016/0378-4371(84)90096-7.
Inden G, in Phase Transformations in Materials, Ch. 8, (ed) Kostorz G, Wiley-Blackwell, 2005, p. 519. https://doi.org/10.1002/352760264X.ch8.
Kikuchi R, J Chem Phys 60 (1974) 1071. https://doi.org/10.1063/1.1681115.
Xingjun L, Shiming H, Rongwen Y, Calphad 19 (1995) 81. https://doi.org/10.1016/0364-5916(95)00009-4.
Pretti M, J Stat Phys 119 (2005) 659. https://doi.org/10.1007/s10955-005-4426-x.
Kiyokane N, Mohri T, Mater Trans 52 (2011) 428. https://doi.org/10.2320/matertrans.MBW201011.
Anoune M, Aouachria Z, J Phase Equil Diffus 32 (2011) 17. https://doi.org/10.1007/s11669-010-9821-6.
Sanchez J M, De Fontaine D, Phys Rev B 17 (1978) 2926. https://doi.org/10.1103/PhysRevB.17.2926.
Yuille A L, Neural Comput 14 (2002) 1691. https://doi.org/10.1162/08997660260028674.
Lele S, Sarma B N, J Mater Sci 44 (2009) 2334. https://doi.org/10.1007/s10853-008-3197-6.
Harvey J P, Eriksson G, Orban D, Chartrand P, Am J Sci 313 (2013) 199. https://doi.org/10.2475/03.2013.02.
Guggenheim E A, Mixtures: The Theory of the Equilibrium Properties of Some Simple Classes of Mixtures Solutions and Alloys, Clarendon Press (1952).
Sarma B N, Shah S N, Kumar M, Lele S, Int J Mater Res 103 (2012) 1188. https://doi.org/10.3139/146.110755.
Gorrey R P, Jindal V, Sarma B N, Lele S, Calphad 71 (2020) 101773. https://doi.org/10.1016/j.calphad.2020.101773.
Laughlin D E, Soffa W A, Acta Mater 145 (2018) 49. https://doi.org/10.1016/J.ACTAMAT.2017.11.037.
Sluiter M, Kawazoe Y, Phys Rev B 59 (1999) 3280. https://doi.org/10.1103/PhysRevB.59.3280.
Kusoffsky A, Sundman B, J Phys Chem Solids 59 (1998) 1549. https://doi.org/10.1016/S0022-3697(98)00066-3.
Schön C, Inden G, Acta Mater 46 (1998) 4219. https://doi.org/10.1016/S1359-6454(98)00096-2.
Lupis C H P, Chemical Thermodynamics of Materials, North-Holland (1983).
Balogh Z, Schmitz G, in Physical Metallurgy, 5th edition, (ed) Laughlin D E, Hono K, Elsevier, 2014, Ch. 5, p. 387. https://doi.org/10.1016/B978-0-444-53770-6.00005-8.
Oates W A, Zhang F, Chen S L, Chang Y A, Phys Rev B 59 (1999) 11221–11225. https://doi.org/10.1103/PhysRevB.59.11221.
Ferreira L G, Mbaye A A, Zunger A, Phys Rev B 35 (1987) 6475. https://doi.org/10.1103/PhysRevB.35.6475.
Gajdics B, Tomán J J, Erdélyi Z, Calphad 67 (2019) 101665. https://doi.org/10.1016/J.CALPHAD.2019.101665.
Abe T, Ogawa K, Hashimoto K, Calphad 38 (2012) 161. https://doi.org/10.1016/j.calphad.2012.06.006.
Hillert M, J Phase Equil 15 (1994) 35. https://doi.org/10.1007/BF02667679.
van de Walle A, Ceder G, Rev Modern Phys 74 (2002) 11. https://doi.org/10.1103/RevModPhys.74.11.
Asta M, McCormack R, de Fontaine D, Phys Rev B 48 (1993) 748. https://doi.org/10.1103/PhysRevB.48.748.
Kaptay G, Calphad 56 (2017) 169. https://doi.org/10.1016/j.calphad.2017.01.002.
Kaptay G, Calphad 44 (2014) 81. https://doi.org/10.1016/J.CALPHAD.2013.08.007.
Gorrey R P, Jindal V, Sarma B N, Lele S, Comput Materi Sci 186 (2021) 109746. https://doi.org/10.1016/j.commatsci.2020.109746.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
At the phase boundary,
At the phase boundary, the differential terms in the numerator as well as the denominator in Eq. (58) vanish, making their ratio indeterminate. It can be evaluated by considering
The derivatives in Eq. (44) are evaluated from Eq. (17) and substituted in Eq. (44) which after simplification becomes
where
The terms which are independent of \(\xi ^2\) in the denominator of RHS in Eq. (60) together are expanded around \(u_0\) corresponding to its value at the phase boundary, namely \(u_0 = u_{0b}\) (given in Eq. (28)) which yields
X\(_b\) in the above expression corresponds to the value of X at \(u_0\) = \(u_{0b}\). Expanding the terms which are independent of (\(u_0 - u_{0b}\)) in the above expression together around \(\xi\) = 0 and with substitution of boundary condition corresponding to \(u_0\) from Eq. (28) yields
Substituting the above expression in Eq. (60) and cancelling \(\xi ^2\) from the numerator and denominator
In the limit of \(u_0 \rightarrow u_{0b}\) and \(\xi \rightarrow 0\) corresponding to the phase boundary,
Substituting from Eq. (63) in Eq. (62), we obtain
The above equation can be solved for \({{d}}\xi ^2/{{d}}x_\text{B}\) in terms of \(u_{0b}\) to yield
Substituting from Eq. (65) in Eq. (43) and utilizing the boundary equation, Eq. (28), we get
Rights and permissions
About this article
Cite this article
Gorrey, R.P., Jindal, V., Sarma, B.N. et al. Thermodynamics of Binary bcc and fcc Phases for Exclusive Second-Neighbour Pair Interactions Using Cluster Variation Method: Analytical Solutions. Trans Indian Inst Met 75, 1365–1381 (2022). https://doi.org/10.1007/s12666-021-02469-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-021-02469-2