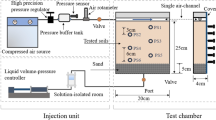
Abstract
Leakage of wastewater from sewer lines may result in contamination of soil and groundwater. The widespread use of detergents is causing relatively high concentrations of surfactants in wastewater. We studied the effects of surfactants on the infiltration process of wastewater through soil. To that aim, in a laboratory experiment three micro-pore glass filters were installed. A laboratory wastewater substitute was created by adding a commercially available detergent to degassed tap water producing surfactant concentrations of 100, 200 and 400 mg l−1. Rapid changes in permeability after using the surfactant solution were detected. These were further examined during a process of washing the glass filters with water. The experimental results indicated that changes in permeability were induced by significant adsorption of surfactant molecules on the solids surfaces and thus reducing the size of the pores. Higher levels of permeability changes were detected for higher surfactant concentrations. Additionally, the efficiency of washing process of an adsorbed surfactant molecular layer at the pore surfaces was greater that before the critical micelle concentration.







Similar content being viewed by others
References
Abu-Zreig M, Rudra R, Dickinson W (2003) Effect of application of surfactants on hydraulic properties of soils. Biosyst Eng 84(3):363–372
Allaire SE, Roulier S, Cessna AJ (2009) Quantifying preferential flow in soils: a review of different techniques. J Hydrol 378(1):179–204
Azam MR, Tan IM, Ismail L, Mushtaq M, Nadeem M, Sagir M (2013) Static adsorption of anionic surfactant onto crushed Berea sandstone. J Petroleum Explor Prod Technol 3(3):195–201
Birdi KS (2002) Self-assembly monolayer structures of lipids and macromolecules at interfaces. Kluwer Academic, New York. doi:10.1007/b114152
Barnes H (1995) A review of the slip (wall depletion) of polymer solutions, emulsions and particle suspensions in viscometers: its cause, character, and cure. J NonNewton Fluid Mech 56(3):221–251
Blackwood D, Ellis J, Revitt D, Gilmour D (2005) Factors influencing exfiltration processes in sewers. Water Sci Technol 51(2):147–154
Bond R (1964) The influence of the microflora on the physical properties of soils II. Field studies on water repellent sands. Soil Res 2(1):123–131
Cirelli A, Ojeda C, Castro M, Salgot M (2008) Surfactants in sludge-amended agricultural soils: a review. Environ Chem Lett 6(3):135–148
Davies J, Clarke B, Whiter J, Cunningham R (2001) Factors influencing the structural deterioration and collapse of rigid sewer pipes. Urban Water 3(1–2):73–89
Doerr S, Shakesby R, Walsh R (2000) Soil water repellency: its causes, characteristics and hydro-geomorphological significance. Earth Sci Rev 51(1):33–65
Duan C, Majumdar A (2010) Anomalous ion transport in 2-nm hydrophilic nanochannels. Nat Nanotechnol 5(12):848–852
Du Y, Shen C, Zhang H, Huang Y (2013) Effects of flow velocity and nonionic surfactant on colloid straining in saturated porous media under unfavorable conditions. Transp Porous Media 98(1):193–208
Ellis JB, Revitt DM, Vollertsen J, Blackwood DJ (2009) Sewer exfiltration and the colmation layer. Water Sci Technol 59(11):2273–2280
Giles CH, Macewan T, Nakhwa S, Smith D (1960) 786. Studies in adsorption. Part XI. A system of classification of solution adsorption isotherms, and its use in diagnosis of adsorption mechanisms and in measurement of specific surface areas of solids. J Chem Soc (Resumed), pp 3973–3993
Held I, Wolf L, Eiswirth M et al (2006) Impacts of sewer leakage on urban groundwater. Springer, pp 189–204. doi: 10.1007/1-4020-5175-1_15
Hillel D (2004) Introduction to environmental soil physics. Elsevier, Amsterdam
Jódar-Reyes A, Ortega-Vinuesa J, Martín-Rodríguez A (2006) Electrokinetic behavior and colloidal stability of polystyrene latex coated with ionic surfactants. J Colloid Interface Sci 297(1):170–181
Karpf C, Hoeft S, Scheffer C, Fuchs L, Krebs P (2011) Groundwater infiltration, surface water inflow and sewerage exfiltration considering hydrodynamic conditions in sewer systems. Water Sci Technol 63(9):1841–1848
Li H, Chen J, Jiang L (2014a) Elevated critical micelle concentration in soil–water system and its implication on PAH removal and surfactant selecting. Environ Earth Sci 71(9):3991–3998
Li J, Zhan Y, Lin J, Jiang A, Xi W (2014b) Removal of bisphenol A from aqueous solution using cetylpyridinium bromide (CPB)-modified natural zeolites as adsorbents. Environ Earth Sci 72(10):3969–3980
Mitchell R, Nevo Z (1964) Effect of bacterial polysaccharide accumulation on infiltration of water through sand. Appl Microbiol 12(3):219–223
Myers D (2006) Surfactant science and technology, 3rd edn. Wiley Inc., New Jersey
Nikpay M, Lazik D, Krebs P (2014) Water displacement by surfactant solution: an experimental study to represent wastewater loss from sewers to saturated soil. Int J Environ Sci Technol. doi:10.1007/s13762-014-0681-1
Nissen HH, Moldrup P, Henriksen K (1998) High-resolution time domain reflectometry coil probe for measuring soil water content. Soil Sci Soc Am J 62(5):1203–1211
Paria S, Khilar K (2004) A review on experimental studies of surfactant adsorption at the hydrophilic solid–water interface. Adv Colloid Interface Sci 110(3):75–95
Renshaw CE, Zynda GD, Fountain JC (1997) Permeability reductions induced by sorption of surfactant. Water Resour Res 33(3):371–378
Schwarzenbach R, Escher B, Fenner K, Hofstetter T, Johnson C, Von Gunten U, Wehrli B (2006) The challenge of micropollutants in aquatic systems. Science 313(5790):1072–1077
Somasundaran P, Huang L (2000) Adsorption/aggregation of surfactants and their mixtures at solid–liquid interfaces. Adv Colloid Interface Sci 88(1):179–208
Song B, Wang Z, Zhang X (2006) Stabilizing interfacial micellar aggregates by enhanced supramolecular interaction or surface polymerization. Pure Appl Chem 78(5):1015–1023
Stein D, Kruithof M, Dekker C (2004) Surface-charge-governed ion transport in nanofluidic channels. Phys Rev Lett 93(3):035901
Tiberg F, Brinck J, Grant L (1999) Adsorption and surface-induced self-assembly of surfactants at the solid–aqueous interface. Curr Opin Colloid Interface Sci 4(6):411–419
Troitsky V, Berzina T, Shchukin D, Sukhorukov G, Erokhin V, Fontana M (2004) Simple method of hydrophilic/hydrophobic patterning of solid surfaces and its application to self-assembling of nano engineered polymeric capsules. Colloids Surf, A 245(1):163–168
Wang L, Ye M, Fernando Rios J, Fernandes R, Lee P, Hicks R (2013) Estimation of nitrate load from septic systems to surface water bodies using an ArcGIS-based software. Environ Earth Sci 70(4):1911–1926
Yao C, Alvarado JL, Marsh CP, Jones BG, Collins MK (2014) Wetting behavior on hybrid surfaces with hydrophobic and hydrophilic properties. Appl Surf Sci 290:59–65
Zhang R, Somasundaran P (2006) Advances in adsorption of surfactants and their mixtures at solid/solution interfaces. Adv Colloid Interface Sci 123:213–229
Acknowledgments
The work was kindly supported by Helmholtz Interdisciplinary Graduate School for Environmental Research (HIGRADE).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nikpay, M., Lazik, D. & Krebs, P. Permeability changes by surfactant solution: an experimental study to represent wastewater loss from sewers to saturated soil. Environ Earth Sci 73, 8443–8450 (2015). https://doi.org/10.1007/s12665-014-4003-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-014-4003-1