Abstract
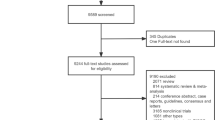
Despite the growing disease burden of non-alcoholic fatty liver disease (NAFLD), approved medical treatments to improve or prevent liver fibrosis are effective only in a small number of patients. Recent studies have found the new use of antiplatelet agents for antifibrotic benefits in NAFLD, but human studies are still limited. The goal of this meta-analysis was to combine the findings of existing relevant studies to investigate the effects of antiplatelet therapy in reducing or preventing advanced liver fibrosis in patients with NAFLD. We conducted a systematic literature search in PubMed, EMBASE, and Web of Science databases from inception to January 2021 to identify all original studies that investigated the use of antiplatelet agents in patients with NAFLD. We used the National Institutes of Health’s quality assessment tool for observational cohort and cross-sectional studies to assess study quality and risk of bias. The primary outcome was the prevalence of advanced liver fibrosis stage 3–4. Data from each study was combined using the random-effects, generic inverse variance method of DerSimonian and Laird to calculate pooled odds ratio (OR) and 95% confidence intervals (CIs). Of the 2,498 studies identified, 4 studies involving 2,593 patients with NAFLD were included in this study (949 antiplatelet agent users and 1,644 non-antiplatelet agent users). The use of aspirin and/or P2Y12 receptor inhibitors was associated with a lower pooled OR of advanced liver fibrosis in patients with NAFLD (pooled OR = 0.66; 95% CI: 0.53–0.81, I2 = 0.0%; p < 0.001). This study focuses on the outcome of advanced liver fibrosis in patients with NAFLD. Our study is limited by the small number of studies that were included. Preliminary evidence from this meta-analysis suggests a protective association between antiplatelet therapy and the prevalence of advanced liver fibrosis in patients with NAFLD. Our findings support future research into repositioning an antiplatelet agent as a novel NAFLD treatment.



Similar content being viewed by others
Data availability
Template data collection forms, data extracted from included studies, data used for all analyses, analytic code, and any other materials used in the review will be provided upon request to corresponding author.
References
Younossi ZM, Stepanova M, Afendy M, et al. Changes in the prevalence of the most common causes of chronic liver diseases in the United States from 1988 to 2008. Clin Gastroenterol Hepatol. 2011;9:524-30.e1.
Rinella ME. Nonalcoholic fatty liver disease: a systematic review. JAMA. 2015;313:2263–73.
Golabi P, Bush H, Stepanova M, et al. Liver transplantation (LT) for cryptogenic cirrhosis (CC) and nonalcoholic steatohepatitis (NASH) cirrhosis: data from the Scientific Registry of Transplant Recipients (SRTR): 1994 to 2016. Medicine (Baltimore). 2018;97:e11518.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–57.
Calzadilla Bertot L, Adams LA. The natural course of non-alcoholic fatty liver disease. Int J Mol Sci. 2016;17:774.
Dulai PS, Singh S, Patel J, et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology. 2017;65:1557–65.
Tana C, Ballestri S, Ricci F, et al. Cardiovascular risk in non-alcoholic fatty liver disease: mechanisms and therapeutic implications. Int J Environ Res Public Health. 2019;16:3104.
Lombardi R, Onali S, Thorburn D, Davidson BR, Gurusamy KS, Tsochatzis E. Pharmacological interventions for non-alcohol related fatty liver disease (NAFLD): an attempted network meta-analysis. Cochrane Database Syst Rev. 2017;3:CD011640.
Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med. 2018;24:908–22.
Estes C, Razavi H, Loomba R, Younossi Z, Sanyal AJ. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology. 2018;67:123–33.
Liu Y, Nong L, Jia Y, et al. Aspirin alleviates hepatic fibrosis by suppressing hepatic stellate cells activation via the TLR4/NF-ΚB pathway. Aging (Albany NY). 2020;12:6058–66.
Li C-J, Yang Z-H, Shi X-L, Liu D-L. Effects of aspirin and enoxaparin in a rat model of liver fibrosis. World J Gastroenterol. 2017;23:6412–9.
Shen H, Shahzad G, Jawairia M, Bostick RM, Mustacchia P. Association between aspirin use and the prevalence of nonalcoholic fatty liver disease: a cross-sectional study from the Third National Health and Nutrition Examination Survey. Aliment Pharmacol Ther. 2014;40:1066–73.
Yoshida S, Ikenaga N, Liu SB, et al. Extrahepatic platelet-derived growth factor-beta, delivered by platelets, promotes activation of hepatic stellate cells and biliary fibrosis in mice. Gastroenterology. 2014;147:1378–92.
Jiang ZG, Feldbrugge L, Tapper EB, et al. Aspirin use is associated with lower indices of liver fibrosis among adults in the United States. Aliment Pharmacol Ther. 2016;43:734–43.
Schwarzkopf K, Bojunga J, Rüschenbaum S, et al. Use of antiplatelet agents is inversely associated with liver fibrosis in patients with cardiovascular disease. Hepatol Commun. 2018;2:1601–9.
Singh A, Gosai F, Lopez R, Vigni A. Regular aspirin use is not protective against advanced fibrosis in type-2 diabetics with biopsy-proven nonalcoholic fatty liver disease. J Hepatol. 2018;68:S582–3.
Simon TG, Henson J, Osganian S, et al. Daily aspirin use associated with reduced risk for fibrosis progression in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2019;17:2776–2784.e4.
Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
National Institutes of Health. Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies. In: Study Quality Assessment Tools. 2013. https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools. Accessed 2 Feb 2021.
Parthasarathy G, Revelo X, Malhi H. Pathogenesis of nonalcoholic steatohepatitis: an overview. Hepatol Commun. 2020;4:478–92.
Yeomans ND. Aspirin: old drug, new uses and challenges. J Gastroenterol Hepatol. 2011;26:426–31.
Katsarou A, Moustakas II, Pyrina I, Lembessis P, Koutsilieris M, Chatzigeorgiou A. Metabolic inflammation as an instigator of fibrosis during non-alcoholic fatty liver disease. World J Gastroenterol. 2020;26:1993–2011.
Ying H-Z, Chen Q, Zhang W-Y, et al. PDGF signaling pathway in hepatic fibrosis pathogenesis and therapeutics (Review). Mol Med Rep. 2017;16:7879–89.
Borkham-Kamphorst E, Meurer SK, Van de Leur E, Haas U, Tihaa L, Weiskirchen R. PDGF-D signaling in portal myofibroblasts and hepatic stellate cells proves identical to PDGF-B via both PDGF receptor type α and β. Cell Signal. 2015;27:1305–14.
Yoshida S, Ikenaga N, Liu SB, et al. Extrahepatic platelet-derived growth factor-β, delivered by platelets, promotes activation of hepatic stellate cells and biliary fibrosis in mice. Gastroenterology. 2014;147:1378–92.
Fujita K, Nozaki Y, Wada K, et al. Effectiveness of antiplatelet drugs against experimental non-alcoholic fatty liver disease. Gut. 2008;57:1583–91.
Kanai S, Ishihara K, Kawashita E, et al. ASB14780, an orally active inhibitor of Group IVA Phospholipase A2, is a pharmacotherapeutic candidate for nonalcoholic fatty liver disease. J Pharmacol Exp Ther. 2016;356:604–14.
Madan SA, John F, Pitchumoni CS. Nonalcoholic fatty liver disease and mean platelet volume: a systemic review and meta-analysis. J Clin Gastroenterol. 2016;50:69–74.
Alkhouri N, Kistangari G, Campbell C, Lopez R, Zein NN, Feldstein AE. Mean platelet volume as a marker of increased cardiovascular risk in patients with nonalcoholic steatohepatitis. Hepatology. 2012;55:331.
Hu X, Cifarelli V, Sun S, Kuda O, Abumrad NA, Su X. Major role of adipocyte prostaglandin E2 in lipolysis-induced macrophage recruitment. J Lipid Res. 2016;57:663–73.
Han Y-M, Lee Y-J, Jang Y-N, et al. Aspirin improves nonalcoholic fatty liver disease and atherosclerosis through regulation of the PPARδ-AMPK-PGC-1α pathway in dyslipidemic conditions. Biomed Res Int. 2020;2020:7806860.
Iqbal U, Dennis BB, Li AA, et al. Use of anti-platelet agents in the prevention of hepatic fibrosis in patients at risk for chronic liver disease: a systematic review and meta-analysis. Hepatol Int. 2019;13:84–90.
Acknowledgements
We would like to thank Dr. Poemlarp Mekraksakit for his intellectual assistance with the eligibility criteria.
Author information
Authors and Affiliations
Contributions
T.T. conception, design, data interpretation, manuscript drafting, and corresponding. A.D. manuscript drafting, data acquisition, and quality assessment. W.V. conception, data acquisition, and statistical analysis. P.L. and T. M. data interpretation. E.I. and S.I. manuscript revision.
Corresponding author
Ethics declarations
Conflict of interest
TT, AD, LV, PL, TM, SI, and EI declare no competing interests.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Society of Gastroenterology or the printer/publishers are responsible for the results/findings and content of this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 148 KB)
Rights and permissions
About this article
Cite this article
Thongtan, T., Deb, A., Vutthikraivit, W. et al. Antiplatelet therapy associated with lower prevalence of advanced liver fibrosis in non-alcoholic fatty liver disease: A systematic review and meta-analysis. Indian J Gastroenterol 41, 119–126 (2022). https://doi.org/10.1007/s12664-021-01230-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-021-01230-3