Abstract
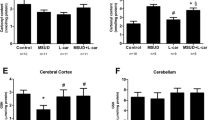
Sulfite oxidase, molybdenum cofactor, and ETHE1 deficiencies are autosomal recessive disorders that affect the metabolism of sulfur-containing amino acids. Patients with these disorders present severe neurological dysfunction and basal ganglia abnormalities, accompanied by high levels of thiosulfate in biological fluids and tissues. Aiming to better elucidate the pathophysiology of basal ganglia damage in these disorders, we evaluated the in vivo effects of thiosulfate administration on bioenergetics, oxidative stress, and neural damage in rat striatum. The in vitro effect of thiosulfate on creatine kinase (CK) activity was also studied. In vivo findings showed that thiosulfate administration decreased the activities of CK and citrate synthase, and increased the activity of catalase 30 min after administration. Activities of other antioxidant enzymes, citric acid cycle, and respiratory chain complex enzymes, as well as glutathione concentrations and markers of neural damage, were not altered by thiosulfate 30 min or 7 days after its administration. Thiosulfate also decreased the activity of CK in vitro in striatum of rats, which was prevented by the thiol reducing agents dithiothreitol (DTT), the antioxidants glutathione (GSH), melatonin, trolox (hydrosoluble analogue of vitamin E), and lipoic acid. DTT and GSH further prevented thiosulfate-induced decrease of the activity of a purified CK in a medium devoid of biological samples. These data suggest that thiosulfate inhibits CK activity by altering critical sulfhydryl groups of this enzyme. It may be also presumed that bioenergetics impairment and ROS generation induced by thiosulfate are mechanisms underlying the neuropathophysiology of disorders in which this metabolite accumulates.









Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Barth M, Ottolenghi C, Hubert L, Chretien D, Serre V, Gobin S, Romano S, Vassault A, Sefiani A, Ricquier D, Boddaert N, Brivet M, de Keyzer Y, Munnich A, Duran M, Rabier D, Valayannopoulos V, de Lonlay P (2010) Multiple sources of metabolic disturbance in ETHE1-related ethylmalonic encephalopathy. J Inherit Metab Dis 33(Suppl 3):S443–S453. https://doi.org/10.1007/s10545-010-9227-y
Bayram E, Topcu Y, Karakaya P, Yis U, Cakmakci H, Ichida K, Kurul SH (2013) Molybdenum cofactor deficiency: review of 12 cases (MoCD and review). Eur J Paediatr Neurol 17:1–6. https://doi.org/10.1016/j.ejpn.2012.10.003
Bhat MD, Prasad C, Tiwari S, Chandra SR, Christopher R (2016) Diffusion restriction in ethylmalonic encephalopathy - an imaging evidence of the pathophysiology of the disease. Brain Dev 38:768–771. https://doi.org/10.1016/j.braindev.2016.02.014
Brizzolari A, Campisi GM, Santaniello E, Razzaghi-Asl N, Saso L, Foti MC (2017) Effect of organic co-solvents in the evaluation of the hydroxyl radical scavenging activity by the 2-deoxyribose degradation assay: the paradigmatic case of alpha-lipoic acid. Biophys Chem 220:1–6. https://doi.org/10.1016/j.bpc.2016.10.005
Browne RW, Armstrong D (1998) Reduced glutathione and glutathione disulfide. Methods Mol Biol 108:347–352. https://doi.org/10.1385/0-89603-472-0:347
Burlina A, Zacchello F, Dionisi-Vici C, Bertini E, Sabetta G, Bennet MJ, Hale DE, Schmidt-Sommerfeld E, Rinaldo P (1991) New clinical phenotype of branched-chain acyl-CoA oxidation defect. Lancet 338:1522–1523. https://doi.org/10.1016/0140-6736(91)92338-3
Busanello EN, Moura AP, Viegas CM, Zanatta A, da Costa Ferreira G, Schuck PF, Wajner M (2010) Neurochemical evidence that glycine induces bioenergetical dysfunction. Neurochem Int 56:948–954. https://doi.org/10.1016/j.neuint.2010.04.002
Butterfield DA, Castegna A, Lauderback CM, Drake J (2002) Evidence that amyloid beta-peptide-induced lipid peroxidation and its sequelae in Alzheimer’s disease brain contribute to neuronal death. Neurobiol Aging 23:655–664. https://doi.org/10.1016/S0197-4580(01)00340-2
Cardoso GMF, Pletsch JT, Parmeggiani B, Grings M, Glanzel NM, Bobermin LD, Amaral AU, Wajner M, Leipnitz G (2017) Bioenergetics dysfunction, mitochondrial permeability transition pore opening and lipid peroxidation induced by hydrogen sulfide as relevant pathomechanisms underlying the neurological dysfunction characteristic of ethylmalonic encephalopathy. Biochim Biophys Acta 1863:2192–2201. https://doi.org/10.1016/j.bbadis.2017.06.007
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490. https://doi.org/10.1016/S0076-6879(85)13062-4
Carmi-Nawi N, Malinger G, Mandel H, Ichida K, Lerman-Sagie T, Lev D (2011) Prenatal brain disruption in molybdenum cofactor deficiency. J Child Neurol 26:460–464. https://doi.org/10.1177/0883073810383017
Chan KY, Li CK, Lai CK, Ng SF, Chan AY (2002) Infantile isolated sulphite oxidase deficiency in a Chinese family: a rare neurodegenerative disorder. Hong Kong Med J 8:279–282
Chang JY (1997) A two-stage mechanism for the reductive unfolding of disulfide-containing proteins. J Biol Chem 272:69–75. https://doi.org/10.1074/jbc.272.1.69
Chen LW, Tsai YS, Huang CC (2014) Prenatal multicystic encephalopathy in isolated sulfite oxidase deficiency with a novel mutaion. Pediatr Neurol 51:181–182. https://doi.org/10.1016/j.pediatrneurol.2014.03.010
Chiarani F, Bavaresco CS, Dutra-Filho CS, Netto CA, Wyse AT (2008) Sulfite increases lipoperoxidation and decreases the activity of catalase in brain of rats. Metab Brain Dis 23:123–132. https://doi.org/10.1007/s11011-007-9073-2
Colon AD, Plaitakis A, Perakis A, Berl S, Clarke DD (1986) Purification and characterization of a soluble and a particulate glutamate dehydrogenase from rat brain. J Neurochem 46:1811–1819. https://doi.org/10.1111/j.1471-4159.1986.tb08500.x
de Moura Alvorcem L, da Rosa MS, Glanzel NM, Parmeggiani B, Grings M, Schmitz F, Wyse ATS, Wajner M, Leipnitz G (2017) Disruption of energy transfer and redox status by sulfite in hippocampus, striatum, and cerebellum of developing rats. Neurotox Res 32:264–275. https://doi.org/10.1007/s12640-017-9732-y
Di Meo I, Fagiolari G, Prelle A, Viscomi C, Zeviani M, Tiranti V (2011) Chronic exposure to sulfide causes accelerated degradation of cytochrome c oxidase in ethylmalonic encephalopathy. Antioxid Redox Signal 15:353–362. https://doi.org/10.1089/ars.2010.3520
Di Meo I, Lamperti C, Tiranti V (2015) Mitochondrial diseases caused by toxic compound accumulation: from etiopathology to therapeutic approaches. EMBO Mol Med 7:1257–1266. https://doi.org/10.15252/emmm.201505040
Drousiotou A, DiMeo I, Mineri R, Georgiou T, Stylianidou G, Tiranti V (2011) Ethylmalonic encephalopathy: application of improved biochemical and molecular diagnostic approaches. Clin Genet 79:385–390. https://doi.org/10.1111/j.1399-0004.2010.01457.x
Fischer JC, Ruitenbeek W, Berden JA, Trijbels JMF, Veerkamp JH, Stadhouders AM, Sengers RCA, Janssen AJM (1985) Differential investigation of the capacity of succinate oxidation in human skeletal muscle. Clin Chim Acta 153:23–36. https://doi.org/10.1016/0009-8981(85)90135-4
Galano A (2011) On the direct scavenging activity of melatonin towards hydroxyl and a series of peroxyl radicals. Physical Chemistry Chemical Physics: PCCP 13:7178–7188. https://doi.org/10.1039/c0cp02801k
Grings M, Moura AP, Parmeggiani B, Marcowich GF, Amaral AU, de Souza Wyse AT, Wajner M, Leipnitz G (2013) Disturbance of brain energy and redox homeostasis provoked by sulfite and thiosulfate: potential pathomechanisms involved in the neuropathology of sulfite oxidase deficiency. Gene 531:191–198. https://doi.org/10.1016/j.gene.2013.09.018
Grings M, Moura AP, Amaral AU, Parmeggiani B, Gasparotto J, Moreira JC, Gelain DP, Wyse AT, Wajner M, Leipnitz G (2014) Sulfite disrupts brain mitochondrial energy homeostasis and induces mitochondrial permeability transition pore opening via thiol group modification. Biochim Biophys Acta 1842:1413–1422. https://doi.org/10.1016/j.bbadis.2014.04.022
Grings M, Moura AP, Parmeggiani B, Motta MM, Boldrini RM, August PM, Matte C, Wyse AT, Wajner M, Leipnitz G (2016) Higher susceptibility of cerebral cortex and striatum to sulfite neurotoxicity in sulfite oxidase-deficient rats. Biochim Biophys Acta 1862:2063–2074. https://doi.org/10.1016/j.bbadis.2016.08.007
Grings M, Moura AP, Parmeggiani B, Pletsch JT, Cardoso GMF, August PM, Matte C, Wyse ATS, Wajner M, Leipnitz G (2017) Bezafibrate prevents mitochondrial dysfunction, antioxidant system disturbance, glial reactivity and neuronal damage induced by sulfite administration in striatum of rats: implications for a possible therapeutic strategy for sulfite oxidase deficiency. Biochim Biophys Acta 1863:2135–2148. https://doi.org/10.1016/j.bbadis.2017.05.019
Grosso S, Mostardini R, Farnetani MA, Molinelli M, Berardi R, Dionisi-Vici C, Rizzo C, Morgese G, Balestri P (2002) Ethylmalonic encephalopathy: further clinical and neuroradiological characterization. J Neurol 249:1446–1450. https://doi.org/10.1007/s00415-002-0880-4
Holder JL Jr, Agadi S, Reese W, Rehder C, Quach MM (2014) Infantile spasms and hyperekplexia associated with isolated sulfite oxidase deficiency. JAMA Neurol 71:782–784. https://doi.org/10.1001/jamaneurol.2013.5083
Hughes BP (1962) A method for the estimation of serum creatine kinase and its use in comparing creatine kinase and aldolase activity in normal and pathological sera. Clin Chim Acta 7:597–603. https://doi.org/10.1016/0009-8981(62)90137-7
Jackson MR, Melideo SL, Jorns MS (2012) Human sulfide:quinone oxidoreductase catalyzes the first step in hydrogen sulfide metabolism and produces a sulfane sulfur metabolite. Biochemistry 51:6804–6815. https://doi.org/10.1021/bi300778t
Johnson JL, Duran M (2001) Molybdenum cofactor deficiency and isolated sulfite oxidase deficiency. In: Scriver CR, Beaudet AL, Valle D, Sly WS (eds) The metabolic and molecular bases of inherited disease. McGraw-Hill, New York, pp 3181–3217
Kitto GB (1969) Intra- and extramitochondrial malate dehydrogenases from chicken and tuna heart: [EC 1.1.1.37 l-malate: NAD oxidoreductase]. Methods Enzymol 13:106–116. https://doi.org/10.1016/0076-6879(69)13023-2
Kumar A, Dejanovic B, Hetsch F, Semtner M, Fusca D, Arjune S, Santamaria-Araujo JA, Winkelmann A, Ayton S, Bush AI, Kloppenburg P, Meier JC, Schwarz G, Belaidi AA (2017) S-sulfocysteine/NMDA receptor-dependent signaling underlies neurodegeneration in molybdenum cofactor deficiency. J Clin Invest 127:4365–4378. https://doi.org/10.1172/JCI89885
Lee HF, Chi CS, Tsai CR, Chen HC, Lee IC (2017) Prenatal brain disruption in isolated sulfite oxidase deficiency. Orphanet J Rare Dis 12:115. https://doi.org/10.1186/s13023-017-0668-3
Leong SF, Clark JB (1984) Regional development of glutamate dehydrogenase in the rat brain. J Neurochem 43:106–111. https://doi.org/10.1111/j.1471-4159.1984.tb06684.x
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mannervik B, Guthenberg C (1981) Glutathione transferase (human placenta). Methods Enzymol 77:231–235. https://doi.org/10.1016/S0076-6879(81)77030-7
Marklund S (1985) Pyrogallol autoxidation. In: Greenwald RA (ed) Handbook of methods for oxygen radical research. CRC Press, Boca Raton, pp 243–247
Mineri R, Rimoldi M, Burlina AB, Koskull S, Perletti C, Heese B, von Dobeln U, Mereghetti P, Di Meo I, Invernizzi F, Zeviani M, Uziel G, Tiranti V (2008) Identification of new mutations in the ETHE1 gene in a cohort of 14 patients presenting with ethylmalonic encephalopathy. J Med Genet 45:473–478. https://doi.org/10.1136/jmg.2008.058271
Mirandola SR, Melo DR, Schuck PF, Ferreira GC, Wajner M, Castilho RF (2008) Methylmalonate inhibits succinate-supported oxygen consumption by interfering with mitochondrial succinate uptake. J Inherit Metab Dis 31:44–54. https://doi.org/10.1007/s10545-007-0798-1
Moura AP, Grings M, Dos Santos Parmeggiani B, Marcowich GF, Tonin AM, Viegas CM, Zanatta A, Ribeiro CA, Wajner M, Leipnitz G (2013) Glycine intracerebroventricular administration disrupts mitochondrial energy homeostasis in cerebral cortex and striatum of young rats. Neurotox Res 24:502–511. https://doi.org/10.1007/s12640-013-9396-1
Moura AP, Parmeggiani B, Grings M, Alvorcem LM, Boldrini RM, Bumbel AP, Motta MM, Seminotti B, Wajner M, Leipnitz G (2016) Intracerebral glycine administration impairs energy and redox homeostasis and induces glial reactivity in cerebral cortex of newborn rats. Mol Neurobiol 53:5864–5875. https://doi.org/10.1007/s12035-015-9493-7
Olivera-Bravo S, Fernandez A, Sarlabos MN, Rosillo JC, Casanova G, Jimenez M, Barbeito L (2011) Neonatal astrocyte damage is sufficient to trigger progressive striatal degeneration in a rat model of glutaric acidemia-I. PloS one 6:e20831. https://doi.org/10.1371/journal.pone.0020831
Parmeggiani B, Moura AP, Grings M, Bumbel AP, de Moura Alvorcem L, Tauana Pletsch J, Fernandes CG, Wyse AT, Wajner M, Leipnitz G (2015) In vitro evidence that sulfite impairs glutamatergic neurotransmission and inhibits glutathione metabolism-related enzymes in rat cerebral cortex. Int J Dev Neurosci 42:68–75. https://doi.org/10.1016/j.ijdevneu.2015.03.005
Paxinos G, Watson C (1986) The rat brain in stereotaxic coordinates, 2nd edn. Academic Press, San Diego
Plaut GWE (1969) Isocitrate dehydrogenase (DPN-specific) from bovine heart. Methods Enzymol 13:34–42. https://doi.org/10.1016/0076-6879(69)13012-8
Reiss J, Hahnewald R (2011) Molybdenum cofactor deficiency: mutations in GPHN, MOCS1, and MOCS2. Hum Mutat 32:10–18. https://doi.org/10.1002/humu.21390
Reiter RJ, Tan DX, Manchester LC, Qi W (2001) Biochemical reactivity of melatonin with reactive oxygen and nitrogen species: a review of the evidence. Cell Biochem Biophys 34:237–256. https://doi.org/10.1385/CBB:34:2:237
Rosenthal RE, Hamud F, Fiskum G, Varghese PJ, Sharpe S (1987) Cerebral ischemia and reperfusion - prevention of brain mitochondrial injury by lidoflazine. J Cereb Blood Flow Metab 7:752–758. https://doi.org/10.1038/jcbfm.1987.130
Rustin P, Chretien D, Bourgeron T, Gerard B, Rotig A, Saudubray JM, Munnich A (1994) Biochemical and molecular investigations in respiratory chain deficiencies. Clin Chim Acta 228:35–51. https://doi.org/10.1016/0009-8981(94)90055-8
Schlattner U, Klaus A, Ramirez Rios S, Guzun R, Kay L, Tokarska-Schlattner M (2016) Cellular compartmentation of energy metabolism: creatine kinase microcompartments and recruitment of B-type creatine kinase to specific subcellular sites. Amino acids 48:1751–1774. https://doi.org/10.1007/s00726-016-2267-3
Schuck PF, Rosa RB, Pettenuzzo LF, Sitta A, Wannmacher CM, Wyse AT, Wajner M (2004) Inhibition of mitochondrial creatine kinase activity from rat cerebral cortex by methylmalonic acid. Neurochem Int 45:661–667. https://doi.org/10.1016/j.neuint.2004.03.006
Schwarz G, Mendel RR, Ribbe MW (2009) Molybdenum cofactors, enzymes and pathways. Nature 460:839–847. https://doi.org/10.1038/nature08302
Seminotti B, Amaral AU, Ribeiro RT, Rodrigues MD, Colin-Gonzalez AL, Leipnitz G, Santamaria A, Wajner M (2015) Oxidative stress, disrupted energy metabolism, and altered signaling pathways in glutaryl-CoA dehydrogenase knockout mice: potential implications of quinolinic acid toxicity in the neuropathology of glutaric acidemia type I. Mol Neurobiol 53:6459–6475. https://doi.org/10.1007/s12035-015-9548-9
Shepherd D, Garland PB (1969) Citrate synthase from rat liver. Methods Enzymol 13:11–13. https://doi.org/10.1016/0076-6879(69)13006-2
Tan WH, Eichler FS, Hoda S, Lee MS, Baris H, Hanley CA, Grant PE, Krishnamoorthy KS, Shih VE (2005) Isolated sulfite oxidase deficiency: a case report with a novel mutation and review of the literature. Pediatrics 116:757–766. https://doi.org/10.1542/peds.2004-1897
Tavasoli AR, Rostami P, Ashrafi MR, Karimzadeh P (2017) Neurological and Vascular Manifestations of Ethylmalonic Encephalopathy. Iran J Child Neurol 11:57–60
Tiranti V, D'Adamo P, Briem E, Ferrari G, Mineri R, Lamantea E, Mandel H, Balestri P, Garcia-Silva MT, Vollmer B, Rinaldo P, Hahn SH, Leonard J, Rahman S, Dionisi-Vici C, Garavaglia B, Gasparini P, Zeviani M (2004) Ethylmalonic encephalopathy is caused by mutations in ETHE1, a gene encoding a mitochondrial matrix protein. Am J Hum Genet 74:239–252. https://doi.org/10.1086/381653
Tiranti V, Briem E, Lamantea E, Mineri R, Papaleo E, De Gioia L, Forlani F, Rinaldo P, Dickson P, Abu-Libdeh B, Cindro-Heberle L, Owaidha M, Jack RM, Christensen E, Burlina A, Zeviani M (2006) ETHE1 mutations are specific to ethylmalonic encephalopathy. J Med Genet 43:340–346. https://doi.org/10.1136/jmg.2005.036210
Tiranti V, Viscomi C, Hildebrandt T, Di Meo I, Mineri R, Tiveron C, Levitt MD, Prelle A, Fagiolari G, Rimoldi M, Zeviani M (2009) Loss of ETHE1, a mitochondrial dioxygenase, causes fatal sulfide toxicity in ethylmalonic encephalopathy. Nat Med 15:200–205. https://doi.org/10.1038/nm.1907
Vijayakumar K, Gunny R, Grunewald S, Carr L, Chong KW, DeVile C, Robinson R, McSweeney N, Prabhakar P (2011) Clinical neuroimaging features and outcome in molybdenum cofactor deficiency. Pediatr Neurol 45:246–252. https://doi.org/10.1016/j.pediatrneurol.2011.06.006
Viscomi C, Burlina AB, Dweikat I, Savoiardo M, Lamperti C, Hildebrandt T, Tiranti V, Zeviani M (2010) Combined treatment with oral metronidazole and N-acetylcysteine is effective in ethylmalonic encephalopathy. Nat Med 16:869–871. https://doi.org/10.1038/nm.2188
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–333
Zafeiriou DI, Augoustides-Savvopoulou P, Haas D, Smet J, Triantafyllou P, Vargiami E, Tamiolaki M, Gombakis N, van Coster R, Sewell AC, Vianey-Saban C, Gregersen N (2007) Ethylmalonic encephalopathy: clinical and biochemical observations. Neuropediatrics 38:78–82. https://doi.org/10.1055/s-2007-984447
Zaki MS, Selim L, El-Bassyouni HT, Issa MY, Mahmoud I, Ismail S, Girgis M, Sadek AA, Gleeson JG, Abdel Hamid MS (2016) Molybdenum cofactor and isolated sulphite oxidase deficiencies: clinical and molecular spectrum among Egyptian patients. Eur J Paediatr Neurol 20:714–722. https://doi.org/10.1016/j.ejpn.2016.05.011
Zhang X, Vincent AS, Halliwell B, Wong KP (2004) A mechanism of sulfite neurotoxicity: direct inhibition of glutamate dehydrogenase. J Biol Chem 279:43035–43045. https://doi.org/10.1074/jbc.M402759200
Funding
This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Programa de Apoio a Núcleos de Excelência (PRONEX II)—Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), Pró-Reitoria de Pesquisa/Universidade Federal do Rio Grande do Sul (PROPESQ/UFRGS), Rede Instituto Brasileiro de Neurociência (IBN-Net), and Instituto Nacional de Ciência e Tecnologia em Excitotoxicidade e Neuroproteção (INCT-EN). We thank the Centro de Microscopia e Microanálise, UFRGS, for the support with the immunofluorescence analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
The experimental protocol was approved by the Ethics Committee for Animal Research of the Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil, and followed the NIH Guide for the Care and Use of Laboratory Animals (NIH publication 85–23, revised 1996).
Conflict of Interests
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Grings, M., Parmeggiani, B., Moura, A.P. et al. Evidence that Thiosulfate Inhibits Creatine Kinase Activity in Rat Striatum via Thiol Group Oxidation. Neurotox Res 34, 693–705 (2018). https://doi.org/10.1007/s12640-018-9934-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-018-9934-y