Abstract
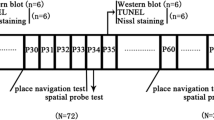
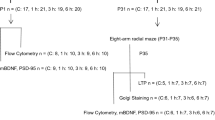
The increasing usage of general anesthetics on young children and infants has drawn extensive attention to the effects of these drugs on cognitive function later in life. Recent animal studies have revealed improvement in hippocampus-dependent performance after lower concentrations of sevoflurane exposure. However, the long-term effects of low-dose sevoflurane on the developing brain remain elusive. On postnatal day (P) 7, rats were treated with 1.2% sevoflurane (1.2% sevo group), 2.4% sevoflurane (2.4% sevo group), and air control (C group) for 6 h. On P35–40, rats’ hippocampus-dependent learning and memory was tested using the Morris water maze. Cognition-related and synapse-related proteins in the hippocampus were measured using Western blotting on P35. On the same day, neurogenesis and synapse ultrastructure were evaluated using immunofluorescence and transmission electron microscopy (TEM). On P35, the rats neonatally exposed to 1.2% sevoflurane showed better behavioral results than control rats, but not in the 2.4% sevo group. Exposure to 1.2% sevoflurane increased the number of 5′-bromo-2-deoxyuridine (BrdU)-positive cells in the dentate gyrus and improved both synaptic number and ultrastructure in the hippocampus. The expression levels of BDNF, TrkB, postsynaptic density (PSD)-95, and synaptophysin in the hippocampus were also increased in the 1.2% sevo group. In contrast, no significant changes in neurogenesis or synaptic plasticity were observed between the C group and the 2.4% sevo group on P35. These results showed that exposure of the developing brain to a low concentration of sevoflurane for 6 h could promote spatial learning and memory function, along with increased hippocampal neurogenesis and synaptic plasticity, in later life.




Similar content being viewed by others
References
Amrock LG, Starner ML, Murphy KL, Baxter MG (2015) Long-term effects of single or multiple neonatal sevoflurane exposures on rat hippocampal ultrastructure. Anesthesiology 122(1):87–95. https://doi.org/10.1097/ALN.0000000000000477
Callaway JK, Jones NC, Royse AG, Royse CF (2012) Sevoflurane anesthesia does not impair acquisition learning or memory in the Morris water maze in young adult and aged rats. Anesthesiology 117(5):1091–1101. https://doi.org/10.1097/ALN.0b013e31826cb228
Chen C, Shen FY, Zhao X, Zhou T, Xu DJ, Wang ZR, Wang YW (2015) Low-dose sevoflurane promotes hippocampal neurogenesis and facilitates the development of dentate gyrus-dependent learning in neonatal rats. ASN neuro 7(2):175909141557584. https://doi.org/10.1177/1759091415575845
Fang F, Song R, Ling X, Peng M, Xue Z, Cang J (2017) Multiple sevoflurane anesthesia in pregnant mice inhibits neurogenesis of fetal hippocampus via repressing transcription factor Pax6. Life Sci 175:16–22. https://doi.org/10.1016/j.lfs.2017.03.003
Feng X, Liu JJ, Zhou X, Song FH, Yang XY, Chen XS, Huang WQ, Zhou LH, Ye JH (2012) Single sevoflurane exposure decreases neuronal nitric oxide synthase levels in the hippocampus of developing rats. Br J Anaesth 109(2):225–233. https://doi.org/10.1093/bja/aes121
Goa KL, Noble S, Spencer CM (1999) Sevoflurane in paediatric anaesthesia: a review. Paediatr Drugs 1:127–153
Gray JD, Milner TA, McEwen BS (2013) Dynamic plasticity: the role of glucocorticoids, brain-derived neurotrophic factor and other trophic factors. Neuroscience 239:214–227. https://doi.org/10.1016/j.neuroscience.2012.08.034
Hariri A, Goldberg T, Mattay V, Kolachana B, Callicott J, Egan M, Weinberger D (2003) Brain-derived neurotrophic factor val(66)met polymorphism affects human memory-related hippocampal activity and predicts memory performance. J Neurosci 23:6690–6694
Haseneder R, Kratzer S, von Meyer L, Eder M, Kochs E, Rammes G (2009) Isoflurane and sevoflurane dose-dependently impair hippocampal long-term potentiation. Eur J Pharmacol 623(1-3):47–51. https://doi.org/10.1016/j.ejphar.2009.09.022
Head E, Corrada MM, Kahle-Wrobleski K, Kim RC, Sarsoza F, Goodus M, Kawas CH (2009) Synaptic proteins, neuropathology and cognitive status in the oldest-old. Neurobiol Aging 30(7):1125–1134. https://doi.org/10.1016/j.neurobiolaging.2007.10.001
Ishizeki J, Nishikawa K, Kubo K, Saito S, Goto F (2008) Amnestic concentrations of sevoflurane inhibit synaptic plasticity of hippocampal CA1 neurons through gamma-aminobutyric acid-mediated mechanisms. Anesthesiology 108:447–456
Jevtovic-Todorovic V, Hartman RE, Izumi Y, Benshoff ND, Dikranian K, Zorumski CF, Olney JW, Wozniak DF (2003) Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J Neurosci : Off J Soc Neurosci 23(3):876–882
Kee N, Sivalingam S, Boonstra R, Wojtowicz JM (2002) The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods 115(1):97–105. https://doi.org/10.1016/S0165-0270(02)00007-9
Lee J, Duan W, Mattson MP (2002) Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J Neurochem 82:1367–1375
Lee JL, Everitt BJ, Thomas KL (2004) Independent cellular processes for hippocampal memory consolidation and reconsolidation. Science (New York, NY) 304:839–843
Lee S, Chung W, Park H, Park H, Yoon S, Park S, Park J, Heo JY, Ju X, Yoon SH, Kim YH, Ko Y (2017) Single and multiple sevoflurane exposures during pregnancy and offspring behavior in mice. Paediatr Anaesth 27(7):742–751. https://doi.org/10.1111/pan.13139
Liang G, Ward C, Peng J, Zhao Y, Huang B, Wei H (2010) Isoflurane causes greater neurodegeneration than an equivalent exposure of sevoflurane in the developing brain of neonatal mice. Anesthesiology 112(6):1325–1334. https://doi.org/10.1097/ALN.0b013e3181d94da5
Loepke AW, Soriano SG (2008) An assessment of the effects of general anesthetics on developing brain structure and neurocognitive function. Anesth Analg 106(6):1681–1707. https://doi.org/10.1213/ane.0b013e318167ad77
Lu B, Nagappan G, Guan X, Nathan PJ, Wren P (2013) BDNF-based synaptic repair as a disease-modifying strategy for neurodegenerative diseases. Nat Rev Neurosci 14(6):401–416. https://doi.org/10.1038/nrn3505
Martin SJ, Grimwood PD, Morris RG (2000) Synaptic plasticity and memory: an evaluation of the hypothesis. Annu Rev Neurosci 23(1):649–711. https://doi.org/10.1146/annurev.neuro.23.1.649
Mellon RD, Simone AF, Rappaport BA (2007) Use of anesthetic agents in neonates and young children. Anesth Analg 104(3):509–520. https://doi.org/10.1213/01.ane.0000255729.96438.b0
Nie H, Peng Z, Lao N, Dong H, Xiong L (2013) Effects of sevoflurane on self-renewal capacity and differentiation of cultured neural stem cells. Neurochem Res 38:1758–1767
Payne RS, Akca O, Roewer N, Schurr A, Kehl F (2005) Sevoflurane-induced preconditioning protects against cerebral ischemic neuronal damage in rats. Brain Res
Sairanen M, Lucas G, Ernfors P, Castren M, Castren E (2005) Brain-derived neurotrophic factor and antidepressant drugs have different but coordinated effects on neuronal turnover, proliferation, and survival in the adult dentate gyrus. J Neurosci : Off J Soc Neurosci 25:1089–1094
Tao G, Luo Y, Xue Q, Li G, Tan Y, Xiao J, Yu B (2016) Docosahexaenoic acid rescues synaptogenesis impairment and long-term memory deficits caused by postnatal multiple sevoflurane exposures. Biomed Res Int 2016:4062579
Tozuka Y, Fukuda S, Namba T, Seki T, Hisatsune T (2005) GABAergic excitation promotes neuronal differentiation in adult hippocampal progenitor cells. Neuron 47:803–815
Tyler WJ, Alonso M, Bramham CR, Pozzo-Miller LD (2002) From acquisition to consolidation: on the role of brain-derived neurotrophic factor signaling in hippocampal-dependent learning. Learning Mem (Cold Spring Harbor, NY) 9:224–237
van Praag H, Schinder AF, Christie BR, Toni N, Palmer TD, Gage FH (2002) Functional neurogenesis in the adult hippocampus. Nature 415(6875):1030–1034. https://doi.org/10.1038/4151030a
Wang SQ, Fang F, Xue ZG, Cang J, Zhang XG (2013) Neonatal sevoflurane anesthesia induces long-term memory impairment and decreases hippocampal PSD-95 expression without neuronal loss. Eur Rev Med Pharmacol Sci 17:941–950
Yang Z, Lv J, Li X, Meng Q, Yang Q, Ma W, Li Y, Ke ZJ (2017) Sevoflurane decreases self-renewal capacity and causes c-Jun N-terminal kinase-mediated damage of rat fetal neural stem cells. Sci Rep 7:46304. https://doi.org/10.1038/srep46304
Zheng H, Dong Y, Xu Z, Crosby G, Culley DJ, Zhang Y, Xie Z (2013) Sevoflurane anesthesia in pregnant mice induces neurotoxicity in fetal and offspring mice. Anesthesiology 118(3):516–526. https://doi.org/10.1097/ALN.0b013e3182834d5d
Funding
This work was supported by the grants from the National Natural Science Foundation of China (No. 81571032 to Xia Feng and No. 81701047 to Xue Zhou).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The use of rats in this study was approved by the Institutional Animal Care and Use Committee at Sun Yat-sen University (Guangzhou, China). All experiments were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and ARRIVE guidelines.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chen, X., Zhou, X., Yang, L. et al. Neonatal Exposure to Low-Dose (1.2%) Sevoflurane Increases Rats’ Hippocampal Neurogenesis and Synaptic Plasticity in Later Life. Neurotox Res 34, 188–197 (2018). https://doi.org/10.1007/s12640-018-9877-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-018-9877-3