Abstract
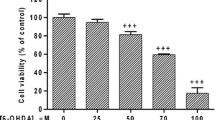
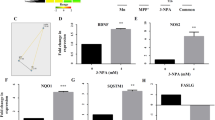
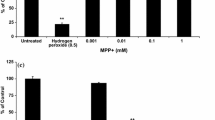
1-Trichloromethyl-1,2,3,4-tetrahydro-β-carboline, abbreviated as TaClo, is an endogenous neurotoxin capable of formation in the brain through the condensation of neuronal tryptamine with ingested exogenous toxins such as trichloroethylene or chloral hydrate. Due to its structural resemblance to 1-methyl-4-phenyl-1,2,5,6-tetrahydropyridine (MPTP), and similar ability to inhibit mitochondrial complex I, TaClo has been implicated in the etiology of Parkinson’s disease. Previous studies have shown the cytotoxicity of TaClo in various cell culture models. In this study, we were interested in identifying the early molecular events within the cell upon exposure to TaClo, a potent mitochondrial toxin. We found increased phosphorylation of 5′-adenosine monophosphate-activated protein kinase (AMPK), induction of autophagy, and a dependence on glycolysis as some of the downstream events to TaClo treatment. Furthermore, TaClo-treated cells undergo accelerated late proliferation but form daughter cells containing fewer neurites, leading to their eventual apoptosis. We also found that TaClo inhibits neuronal prostaglandin E2 synthesis which may play an important role in synaptic plasticity. These results show that TaClo-mediated inhibition of mitochondrial complex I have multiple effects on cellular physiology which are in line with other mitochondrial effectors of Parkinson’s disease.






Similar content being viewed by others
References
Aarts M, Sharpe R, Garcia-Murillas I, Gevensleben H, Hurd MS, Shumway SD, Toniatti C, Ashworth A, Turner NC (2012) Forced mitotic entry of S-phase cells as a therapeutic strategy induced by inhibition of WEE1. Cancer Discov 2(6):524–539
Abdulhamid I, Tremblay M, Stenger J, Tutag Lehr V (2016) Chloral hydrate for sedation of children with asthma during dental treatment. Eur J Paediatr Dent 17(2):141–146
Ahmed H, Al A, Gabr M, Negida A, Abdel-Daim MM (2017) Parkinson’s disease and pesticides: a meta-analysis of disease connection and genetic alterations. Biomed Pharmacother 90:638–649
Akundi RS, Hull M, Clement HW, Fiebich BL (2003) 1-Trichloromethyl-1,2,3,4-tetrahydro-beta-carboline (TaClo) induces apoptosis in human neuroblastoma cell lines. Ann N Y Acad Sci 1010:304–306
Akundi RS, Macho A, Munoz E, Lieb K, Bringmann G, Clement HW, Hull M, Fiebich BL (2004) 1-Trichloromethyl-1,2,3,4-tetrahydro-beta-carboline-induced apoptosis in the human neuroblastoma cell line SK-N-SH. J Neurochem 91(2):263–273
Akundi RS, Zhi L, Sullivan PG, Bueler H (2013) Shared and cell type-specific mitochondrial defects and metabolic adaptations in primary cells from PINK1-deficient mice. Neurodegener Dis 12(3):136–149
Biedler JL, Helson L, Spengler BA (1973) Morphology and growth, tumorigenicity, and cytogenetics of human neuroblastoma cells in continuous culture. Cancer Res 33(11):2643–2652
Boulton SJ, Keane PC, Morris CM, McNeil CJ, Manning P (2012) Real-time monitoring of superoxide generation and cytotoxicity in neuroblastoma mitochondria induced by 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline. Redox Rep 17(3):108–114
Bringmann G, Bruckner R, Mossner R, Feineis D, Heils A, Lesch KP (2000) Effect of 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline (TaClo) on human serotonergic cells. Neurochem Res 25(6):837–843
Bringmann G, Feineis D, Brückner R, God R, Grote C, Wesemann W (2006) Synthesis of radiolabelled 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline (TaClo), a neurotoxic chloral-derived mammalian alkaloid, and its biodistribution in rats. Eur J Pharm Sci 28(5):412–422
Bringmann G, God R, Fahr S, Feineis D, Fornadi K, Fornadi F (1999) Identification of the dopaminergic neurotoxin 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline in human blood after intake of the hypnotic chloral hydrate. Anal Biochem 270(1):167–175
Bringmann G, God R, Feineis D, Wesemann W, Riederer P, Rausch WD, Reichmann H, Sontag KH (1995) The TaClo concept: 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline (TaClo), a new toxin for dopaminergic neurons. J Neural Transm Suppl 46:235–244
Bringmann G, Hille A (1990) Endogenous alkaloids in man, VII: 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline—a potential chloral-derived indol alkaloid in man. Arch Pharm (Weinheim) 323:567–569
Cairns RA, Harris IS, Mak TW (2011) Regulation of cancer cell metabolism. Nat Rev Cancer 11:85–95
Camins A, Pizarro JG, Alvira D, Gutierrez-Cuesta J, de la Torre AV, Folch J, Sureda FX, Verdaguer E, Junyent F, Jordán J, Ferrer I, Pallàs M (2010) Activation of ataxia telangiectasia muted under experimental models and human Parkinson’s disease. Cell Mol Life Sci 67:3865–3882
Chen C, Bazan NG (2005) Endogenous PGE2 regulates membrane excitability and synaptic transmission in hippocampal CA1 pyramidal neurons. J Neurophysiol 93(2):929–941
Chen ML, Chen Q, Xu F, Zhang JX, Su XY, Tu XZ (2017) Safety and efficacy of chloral hydrate for conscious sedation of infants in the pediatric cardiovascular intensive care unit. Medicine (Baltimore) 96(1):e5842
Chiu WA, Jinot J, Scott CS, Makris SL, Cooper GS, Dzubow RC, Bale AS, Evans MV, Guyton KZ, Keshava N, Lipscomb JC, Barone S Jr, Fox JF, Gwinn MR, Schaum J, Caldwell JC (2013) Human health effects of trichloroethylene: key findings and scientific issues. Environ Health Perspect 121(3):303–311
Cole WJ, Mitchell RG, Salamonsen RF (1975) Isolation, characterization and quantitation of chloral hydrate as a transient metabolite of trichloroethylene in man using electron capture gas chromatography and mass fragmentography. J Pharm Pharmacol 27:167–171
Deheshi S, Dabiri B, Fan S, Tsang M, Rintoul GL (2015) Changes in mitochondrial morphology induced by calcium or rotenone in primary astrocytes occur predominantly through ros-mediated remodelling. J Neurochem 133(5):684–699
Demidenko ZN, Kalurupalle S, Hanko C, Lim CU, Broude E, Blagosklonny MV (2008) Mechanism of G1-like arrest by low concentrations of paclitaxel: next cell cycle p53-dependent arrest with sub G1 DNA content mediated by prolonged mitosis. Oncogene 27:4402–4410
Ellis GI, Zhi L, Akundi R, Bueler H, Marti F (2013) Mitochondrial and cytosolic roles of PINK1 shape induced regulatory T cell development and function. Eur J Immunol 43(12):3355–3360
Fiebich BL, Mueksch B, Boehringer M, Hull M (2000) Interleukin-1 beta induces cyclooxygenase 2 and prostaglandin E(2) synthesis in human neuroblastoma cells: involvement of p38 mitogen-activated protein kinase and nuclear factor kappaB. J Neurochem 75(5):2020–2028
Folch J, Junyent F, Verdaguer E, Auladell C, Pizarro JG, Beas-Zarate C, Pallas M, Camins A (2012) Role of cell cycle re-entry in neurons: a common apoptotic mechanism of neuronal cell death. Neurotox Res 22(3):195–207
Frade JM, Ovejero-Benito MC (2015) Neuronal cell cycle: the neuron itself and its circumstances. Cell Cycle 14(5):712–720
Gash DM, Rutland K, Hudson NL, Sullivan PG, Bing G, Cass WA, Pandya JD, Liu M, Choi DY, Hunter RL, Gerhardt GA, Smith CD, Slevin JT, Prince TS (2008) Trichloroethylene: parkinsonism and complex I mitochondrial neurotoxicity. Ann Neurol 63(2):184–192
Goldman SM, Quinlan PJ, Ross GW, Marras C, Meng C, Bhudhikanok GS, Comyns K, Korell M, Chade AR, Kasten M, Priestley B, Chou KL, Fernandez HH, Cambi F, Langston JW, Tanner CM (2012) Solvent exposures and Parkinson’s disease risk in twins. Ann Neurol 71(6):776–784
Guehl D, Bezard E, Dovero S, Boraud T, Bioulac B, Gross C (1999) Trichloroethylene and parkinsonism: a human and experimental observation. Eur J Neurol 6(5):609–611
Hardie DG (2004) The AMP-activated protein kinase pathway—new players upstream and downstream. J Cell Sci 117(Pt23):5479–5487
Höglinger GU, Breunig JJ, Depboylu C, Rouaux C, Michel PP, Alvarez-Fischer D, Boutillier AL, Degregori J, Oertel WH, Rakic P, Hirsch EC, Hunot S (2007) The pRb/E2F cell-cycle pathway mediates cell death in Parkinson’s disease. Proc Natl Acad Sci U S A 104:3585–3590
Hunter T (1995) Protein kinases and phosphatases: the yin and yang of protein phosphorylation and signalling. Cell 80(2):225–236
Istaphanous GK, Loepke AW (2009) General anesthetics and the developing brain. Curr Opin Anaesthesiol 22(3):368–373
Janetzky B, God R, Bringmann G, Reichmann H (1995) 1-Trichloromethyl-1,2,3,4-tetrahydro-beta-carboline, a new inhibitor of complex I. J Neural Transm Suppl 46:265–273
Jiang N, Bo H, Song C, Guo J, Zhao F, Feng H, Ding H, Ji L, Zhang Y (2014) Increased vulnerability with aging to MPTP: the mechanisms underlying mitochondrial dynamics. Neurol Res 36(8):722–732
Kochen W, Kohlmuller D, De Biasi P, Ramsay R (2003) The endogenous formation of highly chlorinated tetrahydro-beta-carbolines as a possible causative mechanism in idiopathic Parkinson’s disease. Adv Exp Med Biol 527:253–263
Kwon HJ, Heo JY, Shim JH, Park JH, Seo KS, Ryu MJ, Han JS, Shong M, Son JH, Kweon GR (2011) DJ-1 mediates paraquat-induced dopaminergic neuronal cell death. Toxicol Lett 202(2):85–92
Lash LH, Chiu WA, Guyton KZ, Rusyn I (2014) Trichloroethylene biotransformation and its role in mutagenicity, carcinogenicity and target organ toxicity. Mutat Res Rev Mutat Res 762:22–36
Lin TK, Cheng CH, Chen SD, Liou CW, Huang CR, Chuang YC (2012) Mitochondrial dysfunction and oxidative stress promote apoptotic cell death in the striatum via cytochrome c/caspase 3 signaling cascade following chronic rotenone intoxication in rats. Int J Mol Sci 13(7):8722–8739
Liu M, Choi DY, Hunter RL, Pandya JD, Cass WA, Sullivan PG, Kim HC, Gash DM, Bing G (2010) Trichloroethylene induces dopaminergic neurodegeneration in fisher 344 rats. J Neurochem 112(3):773–783
Martinez-Vicente M (2017) Neuronal mitophagy in neurodegenerative diseases. Front Mol Neurosci 10:64
Nair VD (2006) Activation of p53 signaling initiates apoptotic death in a cellular model of Parkinson’s disease. Apoptosis 11:955–966
Narendra DP, Jin SM, Tanaka A, Suen DF, Gautier CA, Shen J, Cookson MR, Youle RJ (2010) PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol 8:e1000298
Pietenpol JA, Stewart ZA (2002) Cell cycle checkpoint signalling: cell cycle arrest versus apoptosis. Toxicology 181-182:475–481
Przedborski S (2017) The two-century journey of Parkinson’s disease research. Nat Rev Neurosci 18(4):251–259
Riccardi C, Nicoletti I (2006) Analysis of apoptosis by propidium iodide staining and flow cytometry. Nat Protocols 1(3):1458–1461
Riederer P, Foley P, Bringmann G, Feineis D, Bruckner R, Gerlach M (2002) Biochemical and pharmacological characterization of 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline: a biologically relevant neurotoxin? Eur J Pharmacol 442(1–2):1–16
Rossignol R, Gilkerson R, Aggeler R, Yamagata K, Remington SJ, Capaldi RA (2004) Energy substrate modulates mitochondrial structure and oxidative capacity in cancer cells. Cancer Res 64(3):985–993
Sang N, Zhang J, Marcheselli V, Bazan NG, Chen C (2005) Postsynaptically synthesized prostaglandin E2 (PGE2) modulates hippocampal synaptic transmission via a presynaptic PGE2 EP2 receptor. J Neurosci 25(43):9858–9870
Shridas P, Zhi L, Akundi RS, Webb NR, Pearson KJ, Bueler H (2013) PTEN-induced kinase 1 regulates mitochondrial integrity and insulin secretion in mouse pancreatic β cells. J Endocrinol Diabetes Obes 1(1):1007
Sontag TA, Lange KW, Heim C, Kolasiewicz W, Tucha O, Sontag KH (2007) The long-term effects of the neurotoxin 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline (TaClo) on cognitive performance in rats. J Neural Transm Suppl 72:149–154
Sontag TA, Lange KW, Heim C, Kolasiewicz W, Tucha O, Sontag KH (2009) Alterations of nocturnal activity in rats following subchronic oral administration of the neurotoxin 1-trichloromethyl-1,2,3,4-tetrahydro-beta-carboline. J Neural Transm (Vienna) 116(10):1267–1271
Storch A, Hwang YI, Bringmann G, Feineis D, Ott S, Bruckner R, Schwarz J (2006) Cytotoxicity of chloral-derived beta-carbolines is not specific towards neuronal nor dopaminergic cells. J Neural Transm (Vienna) 113(12):1895–1901
Tanida I, Ueno T, Kominami E (2004) Human light chain 3/MAP1LC3B is cleaved at its carboxyl-terminal Met121 to expose Gly120 for lipidation and targeting to autophagosomal membranes. J Biol Chem 279(46):47704–47710
Toyama EQ, Herzig S, Courchet J, Lewis TL Jr, Losón OC, Hellberg K, Young NP, Chen H, Polleux F, Chan DC, Shaw RJ (2016) Metabolism. AMP-activated protein kinase mediates mitochondrial fission in response to energy stress. Science 351(6270):275–281
Tumilowicz JJ, Nichols WW, Cholon JJ, Greene AE (1970) Definition of a continuous human cell line derived from neuroblastoma. Cancer Res 30(8):2110–2118
Wang R, Cui Y, Xu Y, Irudayaraj J (2017) Basic studies on epigenetic carcinogenesis of low-dose exposure to 1-trichloromethyl-1,2,3,4-tetrahydro-β-carboline (TaClo) in vitro. PLoS One 12(2):e0172243
Zaheer F, Slevin JT (2011) Trichloroethylene and Parkinson’s disease. Neurol Clin 29(3):657–665
Zhang C, Lin M, Wu R, Wang X, Yang B, Levine AJ, Hu W, Feng Z (2011) Parkin, a p53 target gene, mediates the role of p53 in glucose metabolism and the Warburg effect. Proc Natl Acad Sci U S A 108:16259–16264
Zong ZP, Fujikawa-Yamamoto K, Li AL, Yamaguchi N, Chang YG, Murakami M, Odashima S, Ishikawa Y (1999) Both low and high concentrations of staurosporine induce G1 arrest through down-regulation of cyclin E and cdk2 expression. Cell Struct Funct 24:457–463
Acknowledgements
The technical assistance of Ulrike Gotzinger-Berger, Brigitte Gunter, and Raina Seupel is sincerely acknowledged. This work was supported in part by the EU grant QLK3-CT2000 00463 (to B.L.F.) and South Asian University Start Up fund (to R.S.A.). E.C.J was supported by the Alexander von Humboldt Foundation (Bonn, Germany).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sharma, R.K., Candelario-Jalil, E., Feineis, D. et al. 1-Trichloromethyl-1,2,3,4-tetrahydro-beta-carboline (TaClo) Alters Cell Cycle Progression in Human Neuroblastoma Cell Lines. Neurotox Res 32, 649–660 (2017). https://doi.org/10.1007/s12640-017-9782-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-017-9782-1