Abstract
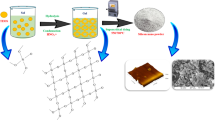
The nano-silicon tetrachloride was prepared by an effective and environmentally friendly ball milling technique. The protonation constants of 2-oxo-N-(pyridine-2-yl)-2-(2-(1-(pyridin-2-yl)ethylidene)hydrazinyl)acetamide (H2L) and the stability constants of its nano silicon complexes were estimated by the potentiometric technique at different temperatures 298, 308 & 318°K.
Similar content being viewed by others
References
Muthuramalingam T, Mohan B, Vignesh S (2018) Performance analysis of pulse generators on residual stress of machined silicon steel using the EDM process. Silicon 10:1785–1792
Yadav TP, Yadav RM, Singh DP (2012) Mechanical milling: a top down approach for the synthesis of nanomaterials and nanocomposites. Nanosci Nanotechnol 2:22–48
Lin PC, Lin S, Wang PC, Sridhar R (2014) Techniques for physicochemical characterization of nanomaterials. Biotechnol Adv 32:711–726
Franco JH, Silva BF, Oliveira RV, Meireles G, Oliveira DP, Castro AA, Ramalho TC, Zanoni MVB (2018) Biotransformation products of disperse dyes with rat liver microsomes by LC-MS/MS and theoretical studies with DNA: structure-mutagenicity relationship using Salmonella/microsome assay. Sci Total Environ 613:1093–1103
Lino JBR, Rocha EP, Ramalho TC (2017) Value of NMR parameters and DFT calculations for quantum information processing utilizing phosphorus heterocycles. J Phys Chem 121:4486–4495
Torres JA, Nogueira FGE, Silva MC, Lopes JH, Tavares TS, Ramalho TC, Corrêa AD (2017) Novel eco-friendly biocatalyst: soybean peroxidase immobilized onto activated carbon obtained from agricultural waste. RSC Adv 7:16460–16466
Carlton CE, Ferreira PJ (2012) In situ TEM nanoindentation of nanoparticles. Micron 43:1134–1139
Martins TLC, Ramalho TC, Figueroa-Villar JD, Flores AFC, Pereira CMP (2003) Theoretical and experimental 13C and 15N NMR investigation of guanylhydrazones in solution. Magn Reson Chem 41:983–988
Silva TC, Almeida KJ, Pires MS, Castro AA, Gonçalves MA, Cunha EFF, Ramalho TC (2017) Theoretical structural and electronic analyses with emphasis on the reactivity of iron oxide prototypes in methane C–H bond activation. Reac Kinet Mech Cat 120:195–208
Bjerrum J (1941) Metal ammine formation in aqueous solution, Haase P and Son, Copenhagen, Denmark
Calvin M, Wilson KW (1945) Stability of chelate compounds. J Am Chem Soc 67:2003–2007
Irving HM, Rossotti HS (1954) The calculation of formation curves of metal complexes from pH titration curves in mixed solvents. J Chem Soc:2904–2910
Uitert LGV, Hass CG (1953) Studies on Coördination compounds. I. a method for determining thermodynamic equilibrium constants in mixed solvents. J Am Chem Soc 75:451–455
Shukla JP, Tandon SG (1972) Corrections to pH measurements for titrations on dioxane–water mixtures. Electroanal Chem Interfacial Electrochem 35:423–427
Harned HS, Owen BB (1967) The physical chemistry of electrolytic solution, 3rd. Reinhold, New York
Izatt RM, Hass CG, Block BP, Fernelius WC (1954) Studies on Coördination compounds. XII. Calculation of thermodynamic formation constants at varying ionic strengths. J Phys Chem 58:1133–1136
Bell P, Rolin RO (1942) Studies in chemotherapy. VII. A theory of the relation of structure to activity of sulfanilamide type compounds. J Am Chem Soc 64:2905–2917
Marshal PB (1995) Some chemical and physical properties association with histamine antagonism. Br J Pharmacol 10:270–278
Martin A, Bustamanate P, Chum AHC (1993) Physical pharmacy, 4th. ed. Lea & Febiger, Philadelphia
Myer MC, Guttman DE (1968) Interactions of xanthine derivatives with bovine serum albumin. 3 Inhibition of binding J Pharm Sci 57:245–249
Brodie BB (1964) Physico-chemical factors in drug absorption. In: Binns TB (ed) Absorption and distribution of drugs. Williams & Wilikins, Baltimore, pp 16–48
Abd El-Wahed MG, Metwally SM, El-Gamel MM, Abd-el Haleem SM (2001) Thermodynamic and electrical properties of aminophenol and Anthranilic acid complexes with some transition metals. Bull Kor Chem Soc 22:663–668
Moeller T, Martin DF, Thompson LC, Ferrus R, Feistel GR, Randal WJ (1965) The coordination chemistry of yttrium and the rare earth metal ions. Chem Rev 65:1–50
Snavely FA, Krecker BD (1959) Metal derivatives of aryl azo Pyrazolone Dyes. III. Molarity quotients of Para-and meta-substituted Pyrazolone dyes. J Am Chem Soc 81:4199–4200
Gutmann K (1966) Conductimetric titration of charge transfer complexes in solution. Electrochim Acta 11:555–568
Singh P, Ojha AC (1980) Synthesis of N-isonicotinyl-3-methyl-4-(substituted hydrazono)-2-pyrazolin-5-one. J Indian Chem Soc 57:1172–1173
Abdel-latif SA, Mansour SE, Fatouh AA (2010) Potentiometric, spectrophotometric, conductimetric and thermodynamic studies on some transition metal complexes derived from 3-methyl-1-phenyl- and1, 3-diphenyl-4-arylazo-5-pyrazolones. Nat Sci 2:793–802
Haschke JM, Hodges AE, Smith CM, Oetting FL (1980) Equilibria and thermodynamic properties of the plutonium-hydrogen system. J Less Common Met 73:41–48
William WP (1993) Inorganic chemistry, 2nd ed. Academic Press Inc, Cambridge
Mcaulery A, Nacollas GH (1961) Thermodynamics of ion association. Part V1I.l A % x m transition-metal oxalates. J Chem Soc:2215–2221
Abd El-Rhim AH (1999) In Faculty of Science. Mansoura University, Egypt
Sari H, Can M, Macit M (2005) Scientific paper potentiometric and theoretical studies of stability constants of glyoxime derivatives and their nickel, copper, cobalt and zinc complexes. Acta Chim Slov 52:317–322
Shokrollahi A, Ghaedi M, Ghaedi H (2007) Potentiometric and spectrophotometric studies of copper(II) complexes of some ligands in aqueous and nonaqueous solution. J Chin Chem Soc 54:933–940
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zaky, R.R., Al-Ahmed, Z.A. Potentiometric Determination of the Stability Constants of Nano-Silicon Complexes in Mixed Solvent at Different Temperatures. Silicon 11, 2811–2818 (2019). https://doi.org/10.1007/s12633-019-0075-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-019-0075-6