Abstract
Purpose
Patients undergoing mitral valve surgery are at risk for right ventricular (RV) dysfunction resulting from increased left atrial pressure and increased pulmonary artery impedance. Measures of longitudinal measures of RV function, such as displacement, are commonly performed but have been shown to be depressed after cardiac surgery despite good patient recovery. The aim of this observational study was to assess the early perioperative time course of longitudinal transthoracic echocardiographic (TTE) markers of RV function in a patient population undergoing mitral valve surgery.
Methods
Twenty patients undergoing mitral valve surgery were enrolled in this observational study. Right ventricular longitudinal measurements (tricuspid annular plane systolic excursion [TAPSE], strain, annular velocity [S′], and isovolumic acceleration [IVA]) were performed using TTE and colour Doppler imaging preoperatively (day 1) and postoperatively (days 2 and 6). Comparisons were made between the preoperative and postoperative measurements.
Results
Adequate echocardiographic imaging was obtained for all 20 patients. The TAPSE, strain, and S′ measures remained depressed for up to one week (i.e., day 6) after surgery compared to preoperative values. The IVA was depressed on the first postoperative day (P > 0.001), but by day 6 it was no different from the preoperative value (P = 0.37). The median [interquartile range] time to discharge from hospital was 7 [6-9] days.
Conclusion
Persistent, significant depression of longitudinal markers of RV function despite functional improvement (discharge from hospital) make it difficult to assess recovery during the early perioperative period. Isovolumic acceleration, a load-independent measure of contractility, might be a more reliable measure of early recovery in RV function in this patient population.
Résumé
Objectif
Les patients subissant une chirurgie de valve mitrale courent le risque de dysfonction du ventricule droit (VD) résultant d’une augmentation de la pression auriculaire gauche et de l’impédance artérielle pulmonaire. Des mesures des mesures longitudinales de la fonction du VD, telles que le déplacement, sont fréquentes, mais il a été démontré qu’elles étaient diminuées après une chirurgie cardiaque malgré une bonne récupération du patient. L’objectif de cette étude observationnelle était d’évaluer le décours temporel périopératoire précoce des marqueurs longitudinaux de l’échocardiographie transthoracique (ETT) de la fonction VD dans une population de patients subissant une chirurgie de valve mitrale.
Méthode
Vingt patients subissant une chirurgie de valve mitrale ont été recrutés pour cette étude observationnelle. Des mesures longitudinales du VD (TAPSE—excursion systolique du plan de l’anneau tricuspide, déformation (strain), vélocité annulaire [S′] et accélération isovolumétrique [AIV]) ont été réalisées par ETT et imagerie Doppler couleur en période préopératoire (jour 1) et postopératoire (jours 2 et 6). Nous avons ensuite comparé les mesures préopératoires et postopératoires.
Résultats
Des images échocardiographiques adéquates ont été obtenues pour les 20 patients. Les mesures de TAPSE, de déformation et S′ se sont maintenues en-dessous des valeurs préopératoires pour une durée allant jusqu’à une semaine (c.-à-d. jour 6) après la chirurgie. L’AIV était inférieure le premier jour postopératoire (P > 0,001), mais au jour 6 elle était semblable à la valeur préopératoire (P = 0,37). Le temps médian [écart interquartile] jusqu’à obtention du congé de l’hôpital était de 7 [6-9] jours.
Conclusion
Une dépression significative et persistante des marqueurs longitudinaux de la fonction du VD en dépit d’une amélioration fonctionnelle (congé de l’hôpital) rend difficile l’évaluation de la récupération en période périopératoire précoce. L’accélération isovolumétrique, une mesure de la contractilité indépendante de la charge, pourrait constituer une mesure plus fiable de récupération précoce de la fonction VD chez cette population de patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The assessment of right ventricular (RV) function during the early postoperative period remains an important element when devising treatment strategies and assessing progress when RV dysfunction is suspected. Studies have shown that measures of RV longitudinal function (base to apex displacement) are depressed after cardiac surgery, a condition that may persist for up to a month or more despite good functional recovery.1-5 Finding a measure of longitudinal RV function that accurately reflects ventricular recovery would be helpful during the postoperative assessment.
When using transesophageal echocardiography (TEE), the RV free wall (RV FW) motion frequently does not align with the optimal Doppler imaging plane. Transthoracic echocardiography (TTE) with proper Doppler, however, does allow alignment of the RV FW. During the immediate postoperative period, however, the window is often limited because of the presence of dressings and chest drains. In addition, the image quality in ventilated patients is frequently poor. As a result of their large amplitude and low velocity signal, Doppler-derived longitudinal measures of RV function may be obtained using TTE despite the modest image quality. Longitudinal measurements such as tricuspid annular systolic plane excursion (TAPSE), systolic annular velocity (S′), and systolic strain are load-dependent measures of RV function that may be derived from colour Doppler imaging of tissues. Isovolumic acceleration (IVA), a load-independent measure of RV function may also be measured using colour Doppler imaging.6 Importantly, RV strain and IVA have not been extensively studied during the early perioperative period.
The aim of this study was to examine longitudinal displacement and deformation measures of RV function using colour Doppler imaging during the early postoperative period in a population at risk for RV dysfunction (e.g., patients undergoing mitral valve surgery). We hypothesized that markers of RV longitudinal function would be depressed during the early postoperative period but that load-independent parameters would have an early recovery.
Methods
Study population
Our institutional review board approved this study (August 2013) prior to patient enrolment. Written informed consent was obtained from all patients before surgery. Cardiac patients in a tertiary care university hospital scheduled to undergo elective mitral valve replacement with or without coronary artery bypass grafting were prospectively recruited from August 2013 to March 2014. They were excluded from the study if they were hemodynamically unstable, had more than moderate tricuspid regurgitation, or were not in sinus rhythm. Finally, 20 patients were selected for the study.
Measurement intervals
Echocardiographic studies were performed by the authors at three time points: day 1, on the day of surgery prior to entering the operating room; day 2, on the day after surgery in the intensive care unit; and day 6, before discharge, five days after surgery.
Echocardiographic measurements and data analysis
All echocardiographic measurements were performed with a transthoracic probe (M3S; GE Medical, Milwaukee, WI, USA) using standard harmonic imaging on a GE Vivid Seven ultrasound system. In an apical four-chamber view optimized for the right ventricle, colour Doppler imaging was applied to a sector of the lateral wall, where a recording of at least eight heartbeats was obtained. The colour Doppler was then applied to the septum and another eight heartbeats were recorded. For each recording, the two-dimensional sector was adjusted to obtain a colour Doppler frame rate of at least 200 fps. In the parasternal short axis view, we obtained pulsed-wave Doppler scans using a 4-mm sample volume from the RV outflow tract at the pulmonary valve. Each recording included at least five heartbeats.
All echocardiographic images were analyzed offline. The Doppler analyses were later performed at a reading station by one of the authors (who was blinded to the hemodynamic data) using GE Echopac quantitative analysis software (GE, Milwaukee, WI, USA).
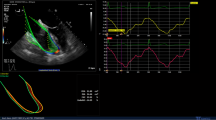
For tricuspid annular velocity (S′) and displacement (TAPSE), a 6 × 6 mm sample volume was placed in the colour Doppler sector at the RV annulus (Fig. E) to maximize the velocity trace (Fig. 1A). The peak systolic velocity (S′) was averaged over five heartbeats (Fig. 1A). The IVA was measured from the first systolic upslope (pre-ejection) of the velocity trace using a slope caliper and averaged over five beats (Fig. 1A). Smoothing was employed for 30 msec to the velocity-derived measurements to minimize noise. Because of the poor image quality of the RV FW during the perioperative period, the velocity trace (for an average of five heartbeats) was integrated over time to yield the TAPSE (Fig. 1B). Linear drift compensation was employed.
Colour Doppler velocity-based measurements of longitudinal right ventricular (RV) function using quantitative analysis software. A tricuspid annular velocity (y axis, in cm/s) trace over time (x axis). The peak systolic velocity (S′) and isovolumic velocity (IVV) are demonstrated. The isovolumic acceleration (IVA) is measured as the slope (between arrows, red line) of the IVV. B Integration of systolic velocity over time (x axis) yields the tricuspid annular plane systolic excursion (TAPSE), measured as peak displacement (arrow) in millimeters (y axis). C Trace of systolic strain (strain, % on y axis) over time (x axis). Values are negative because of myocardial shortening during systole. D, E Four-chamber two-dimensional (2D) views of the right ventricle (bottom) and with colour Doppler imaging (top). The 2D sectors are minimized to obtain a color Doppler frame rate in excess of 200 fps. A 6 × 12 mm sample volume is demonstrated in the mid wall of the right ventricle for strain measurements (D). A 6 × 6 mm sample volume is demonstrated at the base of the RV free wall for velocity measurements (E)
For natural strain measurements, a sample volume measuring 6 × 12 mm and a strain length of 12 mm with 30-msec Gaussian spatial smoothing was used. Linear drift compensation was employed. The sample volume was placed in the colour Doppler sector on each of six segments of the RV FW and septum: basal, mid, apex (Fig. 1D). Care was taken to ensure that the sample volume was rotated to align with and remain within the myocardium throughout systole. Peak systolic strain was measured as the largest strain value during systole and was averaged over five heartbeats (Fig. 1C).
Pulmonary flow velocity acceleration time (AT)—the time from onset to peak flow velocity—was measured from the RV outflow tract on a Doppler trace (Fig. 2). We used an average of five heartbeats.
Statistical methods
Continuous variables were tested for normality (Kolmogorov-Smirnov/Lilliefor). Demographic and heart rate (HR) data were expressed as means (standard deviation [SD]). Doppler data were expressed as means (95% confidence interval [CI]). Intensive care unit (ICU) and hospital lengths of stay were expressed as medians [interquartile range (IQR)]. A paired Student’s t-test was used to compare the day 1 Doppler data and the HR with the corresponding data that had been obtained on days 2 and 6. An unpaired Student’s t-test was used to compare overall day 1 septal strain with the overall RV FW strain. Statistical significance after Bonferroni correction was assumed at P < 0.025 (two-sided). Statistical analyses were performed using XLSTAT software (version 2016.1; Addinsoft, New York, NY, USA).
Results
Twenty-one patients undergoing mitral valve surgery (replacement or repair) with or without coronary artery bypass grafts were recruited. One of these patients was excluded because the images were too poor to evaluate. There were 13 men and seven women. The mean (SD) for age, height, weight, and body surface area were as follows: 63 (9) yr, 170 (11) cm, 82 (17) kg, and 1.96 (0.25) m−2, respectively. Fifteen patients underwent isolated mitral valve replacement, and 5 underwent combined mitral valve replacement and coronary artery bypass graft. Of the 20 patients, 18 had mitral regurgitation, and two had mitral stenosis. Two patients had moderate tricuspid regurgitation. The remainder had mild or no regurgitation.
In all, 16 patients completed the study. The other four patients did not undergo echocardiographic examination on day 6 as they were discharged prior to the sixth day of the study period. The mean (SD) cardiopulmonary bypass time was 109 (44) min, and the mean (SD) cross-clamp time was 95 (30) min. The median [IQR] duration of ICU stay was 23 [19-30] hr. The median [IQR] time to discharge was 7 [6-9] days. There were no deaths. One patient remained in the ICU during the entire 6-day study period and was the only patient who required vasoactive agents during the postoperative period and the only patient who was still intubated on days 2 and 6. All patients were discharged from the hospital. There was no difference in mean (SD) HR between days 1 and 2 [3 (11) beats·min−1 vs 73 (17) beats·min−1, respectively (P = 0.76)]. The mean (SD) HR was significantly higher on day 6 than on day 1 [84 (15) beats·min−1 vs 70 (9) beats·min−1, respectively (P < 0.001)].
Echocardiographic measurements
The changes in echocardiographic variables (excluding strain) are shown in Table 1. There was no significant change in pulmonary AT over the course of the study. Both the TAPSE and S′ were significantly decreased on day 2 compared to day 1 (P < 0.001). This reduction persisted until day 6 (P < 0.001). The IVA was significantly reduced on day 2 compared to day 1 (P < 0.001), but on day 6 it was not significantly different from the preoperative value (day 1) (P = 0.37).
Strain analysis
The overall mean (SD) septal strain was lower than the overall RV FW strain on day 1 [−23.0 (8.6) % vs −17.6 (7.2) %; P < 0.001] (Table 2). There was a significant reduction in global free wall and septal strain on day 2 compared to the preoperative (day 1) value (P < 0.001 for the free wall and P = 0.01 for the septum). The FW strain remained depressed on day 6 compared to that on day 1 (P < 0.001), but there was no significant difference in septal strain on day 6 compared to that on day 1 (Table 2).
When examined by segment on day 2, there was a significant reduction (compared to day 1) in all FW segments and the mid and apical portions of the septum (Table 2). In the RV FW, the base and mid portions remained depressed on day 6 compared to those on day 1. There were no significant differences in any of the septal segments on day 6 compared to those on day 1.
Discussion
In patients undergoing mitral valve surgery, longitudinal measures of FW RV function—TAPSE, S′, strain—were depressed during the immediate postoperative period and up to five postoperative days (i.e., day 6). Although depressed on the first postoperative day, the IVA was not significantly different from its preoperative measurement on day 6.
Perioperative longitudinal markers of RV function
Right ventricular FW measures—TAPSE, S′, strain—are simple and easy to perform, and they correlate well with other measures of RV function.7,8 Longitudinal markers of RV function have been shown to be depressed during the early postoperative period and up to several years after cardiac surgery.1-4,9-12 One study suggested that these changes might be permanent.4 Causes might include intraoperative ischemia, poor myocardial protection, and pericardial tethering or disruption. The prolonged reduction in longitudinal markers remains perplexing as exercise capacity improves significantly after three months and there is no associated reduction in left ventricular function.1,4,5 We previously demonstrated that induction of anesthesia does not alter TAPSE or S′.13 It has also been suggested that reductions in markers of RV function begin during surgery, at the moment of pericardial opening.14 The opening of the pericardial sac might induce changes in RV geometry, resulting in alterations in longitudinal parameters.
In patients with mitral regurgitation, it was demonstrated that left ventricular deformation and size were related.15 Increases in cavity size were associated with decreases in deformation. Preservation or improvements in stroke volume are achieved through a combination of inotropic modulation and chamber size. By extension, it could be inferred that, in the RV after cardiac surgery, adequate stroke volume may be maintained at lower TAPSE, S′, and strain values when associated with increased RV size and changes in geometry. We previously found a loss of association between TAPSE and stroke volume in critically ill patients after cardiac surgery whose stroke volumes were normal at significantly reduced TAPSE.16 This finding suggests that TAPSE alone might markedly underestimate RV output performance in this postoperative patient population, possibly as a result of a change in geometry and dimension.
Global and regional changes in strain
In a study of patients undergoing mitral valve replacements for severe mitral regurgitation, a six-month follow-up revealed significant depression of the global RV FW and septal longitudinal strain compared to preoperative values.1 The authors postulated that a reduction in the radius of the curvature in the longitudinal axis resulting from the loss of pericardial support (bulging of the RV FW) was associated with the reduced global longitudinal strain. When examined by segment, however, we found that all FW segments were significantly depressed on day 2. Although still lower, the apical strain on day six was not significantly different from that on day 1. The apparent apical recovery might have simply been the result of an inability to discriminate subtler changes on day 6 possibly due to the small number of patients or that changes in apical geometry were more favourable.
Although intrinsic myocardial depression would certainly play a role in the reductions in longitudinal measures or RV function (TAPSE, S′, and strain), one must take into consideration that geometric changes might contribute significantly or may mask intrinsic myocardial recovery.
Pulmonary vascular constraints
Pulmonary flow AT is a surrogate marker for the sum of the factors responsible for increased impedance to RV ejection, such as pulmonary vascular resistance, compliance, and wave reflections.17-19 The AT value in our patient population was low (89-100 msec), indicating increased impedance to RV ejection. However, its value remained unchanged throughout the study period, making it less likely that it contributed to the significant alterations seen in RV longitudinal function.
Postoperative IVA recovery
Isovolumic acceleration is a load-independent measure of contractility that indicates the inotropic state of the myocardium.6,20 In a study examining IVA in patients undergoing isolated valve surgery (mitral, aortic, or combined), the authors found that in the mitral surgical subgroup (as in the others) the IVA was depressed for up to one month during the postoperative period compared to preoperative values.21 By six months, the IVA recovered to preoperative values along with functional recovery. We found similar preoperative IVA values and found a marked reduction in IVA on day 2. Contrary to what the previous authors reported, however, we found a prompter recovery at one week. The differences in time course for IVA recovery may have been the result of different patient populations. For instance, in our study there were fewer patients with mitral stenosis. Alternatively, differences in surgical technique, preoperative condition, co-morbidities, and patient dispositions during the early postoperative period and at one week may have been different.
We previously demonstrated that IVA is reduced in cardiac patients undergoing surgery compared to that in controls.13 In a study examining elite endurance athletes, a higher RV IVA was found in the presence of reduced global longitudinal strain than that in the controls.22 The reduced strain was thought to be the result of geometric changes in the RV in this athletic population as the RV wall and cavity sizes were increased when compared to those in the controls. In our study, the IVA was depressed on day 2, but by day 6 the IVA was not significantly different from values prior to surgery despite persistent reductions in the other longitudinal parameters. Although sensitive to HR changes, the small increase in HR at day 6 was likely not large enough to account for the significant difference from day 1.
Depression of RV longitudinal deformation (strain), displacement (TAPSE), and peak displacement velocity (S′), however, may reflect reduced RV systolic function. These reductions may also be the result of changes in geometry, whether induced by changes in volume, loss of pericardial support, or decreased systolic function. Also, because it is independent of loading conditions, the IVA might be a more robust measure of RV recovery and response to therapy during the early postoperative period.
Limitations
This study was an observational study with a relatively small number of patients. Despite this limitation, we found significant changes in all of the longitudinal parameters. We did not examine radial deformation indices (strain). Improvements in deformation orthogonal to the longitudinal axis examined in this study may have accounted for the maintained RV function overall. The presence of a control group, such as patients undergoing uncomplicated coronary artery bypass surgery, might be helpful in determining whether a difference exists in the magnitude of the changes in patients undergoing valvular surgery. We did not correlate our results with exercise capacity or New York Heart Association functional status. However, all patients were successfully discharged from the hospital. We also did not assess geometric changes in RV morphology because the image quality did not permit it.
Conclusion
Right ventricular TAPSE, S′, and free wall strain remained depressed following mitral valve surgery in the presence of patients’ functional recovery and discharge from the hospital. The tricuspid annular IVA, however, may present a more robust marker of RV function for assessing recovery or response to therapy during the early postoperative period in this patient population.
References
Maffessanti F, Gripari P, Tamborini G, et al. Evaluation of right ventricular systolic function after mitral valve repair: a two dimensional Doppler, speckle tracking and three dimensional echocardiographic study. J Am Soc Echocardiogr 2012; 25: 701-8.
Alam M, Hedman A, Nordlander R, Samad B. Right ventricular function before and after uncomplicated coronary artery bypass graft as assessed by pulsed wave Doppler tissue imaging of the tricuspid annulus. Am Heart J 2003; 146: 520-6.
Tamborini G, Muratori M, Brusoni D, et al. Is right ventricular systolic function reduced after cardiac surgery? A two- and three-dimensional echocardiographic study. Eur J echocardiogr 2009; 10: 630-4.
Yadav H, Unsworth B, Fontana M, et al. Selective right ventricular impairment following coronary artery bypass surgery. Eur J Cardiothorac Surg 2010; 37: 393-8.
Hedman A, Alam M, Zuber E, Nordlander R, Abdel Samad B. Decreased right ventricular function after coronary artery bypass grafting and its relation to exercise capacity: a tricuspid annular motion-based study. J Am Soc Echocardiogr 2004; 17: 126-31.
Vogel M, Schmidt MR, Kristiansen SB, et al. Validation of myocardial acceleration during isovolumic contraction as a novel noninvasive index or right ventricular contractility. Circulation 2002; 105: 1693-9.
Miller D, Farah MG, Liner A, et al. The relationship between quantitative ejection fraction and indices of tricuspid annular motion and myocardial performance. J am Soc Echocardiogr 2004; 17: 443-7.
Ueti OM, Camargo EE, de A Ueti A, de Lima-Filho EC, Nogueira EA. Assessment of right ventricular function with Doppler echocardiographic indices derived from tricuspid annular motion: comparison with radionuclide angiography. Heart 2002; 88: 244-8.
Chrustowicz A, Gackowski A, El-Massri N, Sadowski J, Piwowarska W. Preoperative right ventricular function in patients with organic mitral regurgitation. Echocardiography 2010; 27: 282-5.
Desai RR, Vargas Abello LM, Klein AL, et al. Tricuspid regurgitation and right ventricular function after mitral valve surgery with or without concomitant tricuspid valve procedure. J Thorac Cardiovas Surg 2013; 146: 1126-32.
Rafferty T, Durkin M, Harris S, et al. Transesophageal two dimensional echocardiographic analysis of right ventricular systolic performance indices during coronary artery bypass grafting. J Cardiothorac Vasc Anesth 1993; 7: 160-6.
Carr-White GS, Kon M, Glennan S, et al. Right ventricular function after pulmonary autograft replacement of the aortic valve. Circulation 1999; 100: II36-41.
Tousignant C, Bowry R, Cruz JD, Mazer CD. Induction of anesthesia does not alter tricuspid annular velocities: a tissue Doppler assessment. Can J Anesth 2009; 56: 757-62.
Unsworth B, Casula RP, Kyriacou AA, et al. The right ventricular annular velocity reduction caused by coronary artery bypass graft surgery occurs at the moment of pericardial incision. Am Heart J 2010; 159: 314-22.
Marciniak A, Sutherland GR, Marciniak M, et al. Prediction of postoperative left ventricular systolic function in patients with chronic mitral regurgitation undergoing valve surgery—the role of deformation imaging. Eur J Cardiothorac Surg 2011; 40: 1131-7.
Hu R, Mazer CD, Tousignant C. Relationship between tricuspid annular excursion and velocity in cardiac surgical patients. J Cardiothorac Vasc Anesth 2014; 28: 1198-202.
Yared K, Noseworthy P, Weyman AE, et al. Pulmonary acceleration time provides an accurate assessment of systolic pulmonary arterial pressure during transthoracic echocardiography. J Am Soc Echocardiogr 2011; 24: 687-92.
Dabestani A, Mahan G, Gardin JM, et al. Evaluation of pulmonary artery pressure and resistance by pulsed Doppler echocardiography. Am J Cardiol 1987; 59: 662-8.
Granstam SO, Björklund E, Wikström G, et al. Use of echocardiographic pulmonary acceleration time and estimated vascular resistance for the evaluation of possible pulmonary hypertension. Cardiovasc Untrasound 2013; 11: 7.
Tousignant CP, Miller AL, Denault A, Zhu L, Bowry R. The effect of heart rate on tricuspid annular velocities in cardiac surgical patients with atrial fibrillation. J Cardiothorac Vasc Anesth 2008; 4: 565-9.
Tigen K, Karaahmet T, Zencirkiran H, et al. Usefulness of isovolumic acceleration and tissue Doppler echocardiographic parameters for predicting post operative function recovery after heart valve surgery. Rev Esp Cardiol 2010; 63: 430-8.
King G, Almuntaser I, Murphy RT, et al. Reduced right ventricular myocardial strain in the elite athlete may not be a consequence of myocardial damage. “Cream masquerades as skim milk”. Echocardiography 2013; 30: 929-35.
Funding
Educational funding was provided to Dr. Albinmousa by the Prince Sultan Cardiac Centre, Riyadh, Saudi Arabia.
Conflicts of interest
None declared.
Author contributions
Jordan Van Orman and Claude Tousignant contributed substantially to all aspects of this manuscript, including conception and design; acquisition, analysis, and interpretation of data; and drafting the article. Zakariya Albinmousa contributed substantially to the acquisition of data. Kim Connelly contributed substantially to the conception and design of the manuscript.
Editorial responsibility
This submission was handled by Dr. Hilary P. Grocott, Editor-in-Chief, Canadian Journal of Anesthesia.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study (REB 13-168) was approved by the St Michael’s hospital IRB.
Contact SMH research ethics office: Dharmista Patel: pateld@smh.ca.
Rights and permissions
About this article
Cite this article
Van Orman, J.R., Connelly, K., Albinmousa, Z. et al. Early recovery of tricuspid annular isovolumic acceleration after mitral valve surgery – an observational study. Can J Anesth/J Can Anesth 63, 920–927 (2016). https://doi.org/10.1007/s12630-016-0651-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-016-0651-9