Abstract
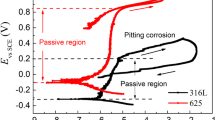
The effect of H2S on the corrosion behavior of 316L stainless steel was investigated using electrochemical methods by changing the gas condition from CO2 to H2S and then back to CO2. The presence of H2S showed an acceleration effect on the corrosion of 316L stainless steel in comparison with CO2. The acceleration effect remained even after the complete removal of H2S by CO2, indicating that the passive film was irreversibly damaged. X-ray photoelectron spectroscopy (XPS) analysis indicated that the passive film was composed of Cr2O3, Fe2O3, and FeS2 after being immersed in H2S-containing solutions. The semiconducting property of the passive film was then investigated by using the Mott–Schottky approach. The presence of sulfides resulted in higher acceptor and donor densities and thus was responsible for the deterioration of passive films.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J.H. Ding, L. Zhang, M.X. Lu, J. Wang, Z.B. Wen, and W.H. Hao, The electrochemical behaviour of 316L austenitic stainless steel in Cl- containing environment under different H2S partial pressures, Appl. Surf. Sci., 289(2014), p. 33.
X.H. Zhao, Y. Han, Z.Q. Bai, and B. Wei, The experiment research of corrosion behaviour about Ni-based alloys in simulant solution containing H2S/CO2, Electrochim. Acta, 56(2011), No. 22, p. 7725.
O.E. Albores-Silva, E.A. Charles, and C. Padovani, Effect of chloride deposition on stress corrosion cracking of 316L stainless steel used for intermediate level radioactive waste containers, Corros. Eng. Sci. Technol., 46(2011), No. 2, p. 124.
M. Ghahari, D. Krouse, N. Laycock, T. Rayment, C. Padovani, M. Stampanoni, F. Marone, R. Mokso, and A.J. Davenport, Synchrotron X-ray radiography studies of pitting corrosion of stainless steel: Extraction of pit propagation parameters, Corros. Sci., 100(2015), p. 23.
L.K. Zhu, Y. Yan, L.J. Qiao, and A.A. Volinsky, Stainless steel pitting and early-stage stress corrosion cracking under ultra-low elastic load, Corros. Sci., 77(2013), p. 360.
L. Gardner, A. Insausti, K.T. Ng, and M. Ashraf, Elevated temperature material properties of stainless steel alloys, J. Constr. Steel Res., 66(2010), No. 5, p. 634.
A. Abbasi Aghuy, M. Zakeri, M.H. Moayed, and M. Mazinani, Effect of grain size on pitting corrosion of 304L austenitic stainless steel, Corros. Sci., 94(2015), p. 368.
I. Taji, M.H. Moayed, and M. Mirjalili, Correlation between sensitisation and pitting corrosion of AISI 403 martensitic stainless steel, Corros. Sci., 92(2015), p. 301.
S.F. Yang and D.D. Macdonald, Theoretical and experimental studies of the pitting of type 316L stainless steel in borate buffer solution containing nitrate ion, Electrochim. Acta, 52(2007), No. 5, p. 1871.
I. Olefjord and L. Wegrelius, Surface analysis of passive state, Corros. Sci, 31(1990), p. 89.
R. Kirchheim, B. Heine, H. Fischmeister, S. Hofmann, H. Knote, and U. Stolz, The passivity of iron-chromium alloys, Corros. Sci., 29(1989), No. 7, p. 899.
C.O.A. Olsson and D. Landolt, Passive films on stainless steels—chemistry, structure and growth, Electrochim. Acta, 48(2003), No. 9, p. 1093.
H. Luo, C.F. Dong, K. Xiao, and X.G. Li, The passive behaviour of ferritic stainless steel containing alloyed tin in acidic media, RSC Adv., 6(2016), No. 12, p. 9940.
J. Banas, U. Lelek-Borkowska, B. Mazurkiewicz, and W. Solarski, Effect of CO2 and H2S on the composition and stability of passive film on iron alloys in geothermal water, Electrochim. Acta, 52(2007), No. 18, p. 5704.
C.H. Liang, W. Hua, and N.B. Huang, Effects of sulphate-reducing bacteria on corrosion behaviour of 2205 duplex stainless steel, J. Iron Steel Res. Int., 21(2014), No. 4, p. 444.
Y.S. Choi, S. Nesic, and S. Ling, Effect of H2S on the CO2 corrosion of carbon steel in acidic solutions, Electrochim. Acta, 56(2011), No. 4, p. 1752.
H.H. Ge, Z. G.D. Zhou, and W.Q. Wu, Passivation model of 316 stainless steel in simulated cooling water and the effect of sulfide on the passive film, Appl. Surf. Sci., 211(2003), No. 1-4, p. 321.
W. He, O.O. Knudsen, and S. Diplas, Corrosion of stainless steel 316L in simulated formation water environment with CO2-H2S-Cl-, Corros. Sci., 51(2009), No. 12, p. 2811.
S. Nešic, Key issues related to modelling of internal corrosion of oil and gas pipelines?A review, Corros. Sci., 49(2007), No. 12, p. 4308.
L. Jun and D.D. Macdonald, The passivity of iron in the presence of ethylenediaminetetraacetic acid. II. The defect and electronic structures of the barrier layer, J. Electrochem. Soc., 148(2001), No. 11, p. B425.
G.S. Lauvstad, R. Johnsen, O. Borck, E.F. da Silva, and J.C. Walmsley, Breakdown in passivity of austenitic stainless steels in Cl- and H2S—modelling and characterization of the pit initiation process, [in] Corrosion 2007, Nashville, NACE-07660.
M. Ben Salah, R. Sabot, P. Refait, I. Liascukiene, C. Methivier, J. Landoulsi, L. Dhouibi, and M. Jeannin, Passivation behaviour of stainless steel (UNS N-08028) in industrial or simplified phosphoric acid solutions at different temperatures, Corros. Sci., 99(2015), p. 320.
E. Abelev, J. Sellberg, T.A. Ramanarayanan, and S.L. Bernasek, Effect of H2S on Fe corrosion in CO2-saturated brine, J. Mater. Sci., 44(2009), No. 22, p. 6167.
L. Liu, M.Y. Wang, Z. Wang, and Y. Zhang, Corrosion behavior of 316L stainless steel anode in alkaline sulfide solutions and the consequent influence on Ga electrowinning, Hydrometallurgy, 157(2015), p. 285.
S. Ningshen, U.K. Mudali, V.K. Mittal, and H.S. Khatak, Semiconducting and passive film properties of nitrogen- containing type 316LN stainless steels. Corros. Sci., 49(2007), No. 2, p. 481.
E.E. Oguzie, J.B. Li, Y.Q. Liu, D.M. Chen, Y. Li, K. Yang, and F.H. Wang, The effect of Cu addition on the electrochemical corrosion and passivation behavior of stainless steels, Electrochim. Acta, 55(2010), No. 17, p. 5028.
K.N. Ohs, S. Ahn, K.S. Eom, and H.S. Kwon, A study on the localized corrosion and repassivation kinetics of Fe-20Cr-xNi (x = 0-20wt%) stainless steels via electrochemical analysis, Corros. Sci., 100(2015), p. 158.
A.M.P. Simoes, M.G.S. Ferreira, B. Rondot, and M. de Cunha Belo, Study of passive films formed on AISI 304 stainless steel by impedance measurements and photoelectrochemistry, J. Electrochem. Soc., 137(1990), No. 1, p. 82.
E. Sikora and D.D. Macdonald, Nature of the passive film on nickel, Electrochim. Acta, 48(2002), No. 1, p. 69.
J.C. Liu, G. Zhang, S. Nagao, J.T. Jiu, M. Nogi, T. Sugahara, J.S. Ma, and K. Suganuma, Metastable pitting and its correlation with electronic properties of passive films on Sn-xZn solder alloys, Corros. Sci., 99(2015), p. 154.
M. BenSalah, R. Sabot, E. Triki, L. Dhouibi, P. Refait, and M. Jeannin, Passivity of Sanicro28 (UNS N-08028) stainless steel in polluted phosphoric acid at different temperatures studied by electrochemical impedance spectroscopy and Mott-Schottky analysis, Corros. Sci., 86(2014), p. 61.
L.V. Taveira, M.F. Montemor, M.D.C. Belo, M.G. Ferreira, and L.F.P. Dick, Influence of incorporated Mo and Nb on the Mott-Schottky behaviour of anodic films formed on AISI 304L, Corros. Sci., 52(2010), No. 9, p. 2813.
J. Williamson and O.B. Isgor, The effect of simulated concrete pore solution composition and chlorides on the electronic properties of passive films on carbon steel rebar, Corros. Sci., 106(2016), p. 82.
D.D. Macdonald, Some personal adventures in passivity?A review of the point defect model for film growth, Russ. J. Electrochem., 48(2012), No. 3, p. 235.
D.D. Macdonald, The point defect model for the passive state, J. Electrochem. Soc., 139(1992), No. 12, p. 3434.
Y.X. Qiao, Y.G. Zheng, W. Ke, and P.C. Okafor, Electrochemical behaviour of high nitrogen stainless steel in acidic solutions, Corros. Sci., 51(2009), No. 5, p. 979.
M.J. Carmezim, A.M. Simões, M.F. Montemor, and M.D.C. Belo, Capacitance behaviour of passive films on ferritic and austenitic stainless steel, Corros. Sci., 47(2005), No. 3, p. 581.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 51271025) and the Beijing Higher Education Young Elite Teacher Project.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Wang, Z., Zhang, L., Tang, X. et al. Investigation of the deterioration of passive films in H2S-containing solutions. Int J Miner Metall Mater 24, 943–953 (2017). https://doi.org/10.1007/s12613-017-1482-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-017-1482-6