Abstract
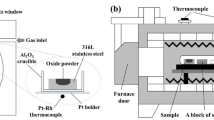
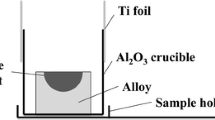
To clarify the influence of FeO and sulfur on solid state reaction between an Fe−Mn−Si alloy and MnO−SiO2−FeO oxides under the restricted oxygen diffusion flux, two diffusion couples with different sulfur contents in the oxides were produced and investigated after heat treatment at 1473 K. The experimental results were also compared with previous work in which the oxides contained higher FeO. It was found that although the FeO content in the oxides decreased from 3wt% to 1wt% which was lower than the content corresponding to the equilibrium with molten steel at 1873 K, excess oxygen still diffused from the oxides to solid steel during heat treatment at 1473 K and formed oxide particles. In addition, increasing the sulfur content in the oxides was observed to suppress the diffusion of oxygen between the alloy and the oxides.
Similar content being viewed by others
References
F.M. Yuan, X.H. Wang, and X.F. Yang, Influence of calcium content on solid ratio of inclusions in Ca-treated liquid steel, J. Univ. Sci. Technol. Beijing, 13(2006), No. 6, p. 486.
H. Ono, K. Nakajima, T. Ibuta, and T. Usui, Equilibrium relationship between the oxide compounds in MgO-Al2O3-Ti2O3 and molten iron at 1873 K, ISIJ Int., 50(2010), No. 12, p. 1955.
S.F. Yang, J.S. Li, Z.F. Wang, J. Li, and L. Lin, Modification of MgO·Al2O3 spinel inclusions in Al-killed steel by Ca-treatment, Int. J. Miner. Metall. Mater., 18(2011), No. 1, p. 18.
W.J. Choi, H. Matsuura, and F. Tsukihashi, Changing behavior of non-metallic inclusions in solid iron deoxidized by Al–Ti addition during heating at 1473 K, ISIJ Int., 51(2011), No. 12, p. 1951.
H. Shibata, K. Kimura, T. Tanaka, and S. Kitamura, Mechanism of change in chemical composition of oxide inclusions in Fe–Cr Alloys deoxidized with Mn and Si by heat treatment at 1473 K, ISIJ Int., 51(2011), No. 12, p. 1944.
H. Shibata, T. Tanaka, K. Kimura, and S.Y. Kitamura, Composition change in oxide inclusions of stainless steel by heat treatment, Ironmaking Steelmaking, 37(2010), No. 7, p. 522.
J.H. Park and D.S. Kim, Effect of CaO-Al2O3-MgO slags on the formation of MgO-Al2O3 inclusions in ferritic stainless steel, Metall. Mater. Trans. B, 36(2005), No. 4, p. 495.
C. Lee, S. Nambu, J. Inoue, and T. Koseki, Ferrite formation behaviors from B1 compounds in steels, ISIJ Int., 51(2011), No. 12, p. 2036.
K.H. Kim, H. Shibata, and S. Kitamura, Solid-state reaction between manganese silicate and Fe-Mn-Si alloy containing sulphur, [in] Proceedings of the 5th International Congress on the Science and Technology of Steelmaking, Dresden, 2012, p. 772.
K.H. Kim, H. Shibata, and S. Kitamura, Reaction between MnO–SiO2–FeO oxide and Fe–Mn–Si solid alloy during heat treatment, ISIJ Int., 54(2014), No. 10, p. 2144.
K.H. Kim, H. Shibata, and S. Kitamura, Influence of sulfur on the reaction between MnO-SiO2-FeO oxide and Fe-Mn-Si solid alloy by heat treatment, ISIJ Int., 54(2014), No. 12, p. 2678.
C.S. Liu, K.H. Kim, S.J. Kim, J.S. Li, S. Ueda, X. Gao, H. Shibata, and S.Y. Kitamura, Reaction between MnO–SiO2–FeO solid oxide and solid steel deoxidized by Si and Mn during heat treatment at 1473 K (1200°C), Metall. Mater. Trans. B, 2015, Online.
S. Ban-ya, Mathematical expression of slag-metal reactions in steelmaking process by quadratic formalism based on the regular solution model, ISIJ Int., 33(1993), No. 1, p. 2.
M. Hino and K. Ito, Thermodynamic Data for Steelmaking, Tohoku University Press, Sendai, 2011, p. 167.
S. Ban-ya, E. Tatsuhiko, and S. Mitsuo, Physical Chemistry of Metals, Maruzen Press, Tokyo, 1996, p. 198.
W.J. Yin, S.H. Wei, M.M. Al-Jassim, and Y.F. Yan, Prediction of the chemical trends of oxygen vacancy levels in binary metal oxides, Appl. Phys. Lett., 99(2011), No. 142109, p. 1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, Cs., Yang, Sf., Kim, Kh. et al. Influence of FeO and sulfur on solid state reaction between MnO−SiO2−FeO oxides and an Fe−Mn−Si solid alloy during heat treatment at 1473 K. Int J Miner Metall Mater 22, 811–819 (2015). https://doi.org/10.1007/s12613-015-1138-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-015-1138-3