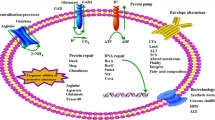
Abstract
Probiotics play a significant role in functional foods. Heat stress and dehydration are the two principal mechanisms leading to inactivation and loss of probiotics viability in its production. There is a need to develop an industrial organism to withstand higher temperatures during its processing and storage. This current study aims to develop thermotolerant strains of Lacticaseibacillus casei N (N) and Lactobacillus helveticus NRRL B-4526 (H) by acclimatizing the wild-type strains to the higher temperature of 45 °C by adaptive laboratory evolution. A two-fold increase in biomass was observed in both acclimatized strains up to the 200th generation, which subsequently remained stable after 500 generations. The morphological change of these acclimatized strains was observed under scanning electron microscopy. Also, there was an increase in probiotic attributes of these acclimatized strains compared to their wild-types. Among two acclimatized strains, L. casei N-45 had shown higher tolerance in the acidic pH 3.0 (89.31%), the bile of 0.3% (84.45%), simulated gastric juice (79.12%), and simulated intestinal juice (73.86%). There was also an increase in salt tolerance (NaCl), radical scavenging activity, autoaggregation, coaggregation, and hydrophobicity of these adapted strains. The total protein profiling using 2D gel electrophoresis reveals the differences in protein expressions between wild-type and acclimatized strains. Specific protein spots from acclimatized strains of H-45 and N-45 were further subjected to MALDI-TOF MS/MS. Some of the identified proteins were recognized to play a role in RNA chaperones and protein synthesis during stress conditions.




Similar content being viewed by others
Data Availability
Sequence data of an isolate Lacticaseibacillus casei N (N) is deposited with the primary accession number: CP077759 (https://www.ncbi.nlm.nih.gov/nuccore/CP077759). All data used or analyzed during this study are included in this article.
References
Food and Agriculture Organization of the United Nations World Health Organization FAO, WHO (2002) Joint FAO/WHO working group report on drafting guidelines for the evaluation of probiotics in food. London, Ontario, Canada. https://www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf
Helland MH, Wicklund T, Narvhus JA (2004) Growth and metabolism of selected strains of probiotic bacteria, in maize porridge with added malted barley. Int J Food Microbiol 91:305–313. https://doi.org/10.1016/j.ijfoodmicro.2003.07.007
Parvez S, Malik KA, Ah Kang S, Kim HY (2006) Probiotics and their fermented food products are beneficial for health. J Appl Microbiol 100:1171–1185. https://doi.org/10.1111/j.1365-2672.2006.02963.x
Hussain MA, Hosseini Nezhad M, Sheng Y, Amoafo O (2013) Proteomics and the stressful life of lactobacilli. FEMS Microbiol Lett 349:1–8. https://doi.org/10.1111/1574-6968.12274
Dragosits M, Mattanovich D (2013) Adaptive laboratory evolution - principles and applications for biotechnology. Microb Cell Fact 12:1–17. https://doi.org/10.1186/1475-2859-12-64
Kulkarni S, Haq SF, Samant S, Sukumaran S (2018) Adaptation of Lactobacillus acidophilus to thermal stress yields a thermotolerant variant which also exhibits improved survival at pH 2. Probiotics Antimicrob Proteins 10:717–727. https://doi.org/10.1007/s12602-017-9321-7
Mbye M, Baig MA, AbuQamar SF, El-Tarabily AK, Obaid RS, Osaili TM, Al-Nabulsi AA, Turner MS, Shah NP, Ayyash MM (2020) Updates on understanding of probiotic lactic acid bacteria responses to environmental stresses and highlights on proteomic analyses. Compr Rev Food Sci Food Saf 19:1110–1124. https://doi.org/10.1111/1541-4337.12554
Khalil AA (2006) Nutritional improvement of an Egyptian breed of mung bean by probiotic lactobacilli. African J Biotechnol 5:206–212
Erkus O (2007) Thesis: isolation, phenotypic and genotypic characterization of yoghurt starter bacteria. Msc Thesis 117
Kenneth T (2020) Growth of bacterial populations. Todar's online textbook of bacteriology 3. http://textbookofbacteriology.net/growth_3.html
Van Heerden JH, Kempe H, Doerr A, Maarleveld T, Nordholt N, Bruggeman FJ (2017) Statistics and simulation of growth of single bacterial cells: illustrations with B. subtilis and E. coli. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-15895-4
Wang J, Dong X, Shao Y, Guo H, Pan L, Hui W, Kwok LY, Zhang H, Zhang W (2017) Genome adaptive evolution of Lactobacillus casei under long-term antibiotic selection pressures. BMC Genomics 18:320. https://doi.org/10.1186/s12864-017-3710-x
Hippolyte MT, Augustin M, Hervé TM, Robert N, Somashekar D (2018) Application of response surface methodology to improve the production of antimicrobial biosurfactants by Lactobacillus paracasei subsp. tolerans N2 using sugar cane molasses as substrate. Bioresour Bioprocess 5:48. https://doi.org/10.1186/s40643-018-0234-4
Archer AC, Halami PM (2015) Probiotic attributes of Lactobacillus fermentum isolated from human feces and dairy products. Appl Microbiol Biotechnol 99:8113–8123. https://doi.org/10.1007/s00253-015-6679-x
Yadav R, Puniya AK, Shukla P (2016) Probiotic properties of Lactobacillus plantarum RYPR1 from an indigenous fermented beverage Raabadi. Front Microbiol 7:1683. https://doi.org/10.3389/fmicb.2016.01683
Zárate G, Chaia AP, González S, Oliver G (2000) Viability and β-galactosidase activity of dairy propionibacteria subjected to digestion by artificial gastric and intestinal fluids. J Food Prot 63:1214–1221. https://doi.org/10.4315/0362-028X-63.9.1214
Bao Y, Zhang Y, Zhang Y, Liu Y, Wang S, Dong X, Wang Y, ZhangH, (2010) Screening of potential probiotic properties of Lactobacillus fermentum isolated from traditional dairy products. Food Control 21:695–701. https://doi.org/10.1016/j.foodcont.2009.10.010
Collado MC, Meriluoto J, Salminen S (2008) Adhesion and aggregation properties of probiotic and pathogen strains. Eur Food Res Technol 226:1065–1073. https://doi.org/10.1007/s00217-007-0632-x
Kaushik JK, Kumar A, Duary RK, Mohanty AK, Grover S, Batish VK (2009) Functional and probiotic attributes of an indigenous isolate of Lactobacillus plantarum. PLoS One 4(12):e80999. https://doi.org/10.1371/journal.pone.0008099
Sharma K, Mahajan R, Attri S, Goel G (2017) Selection of indigenous Lactobacillus paracasei CD4 and Lactobacillus gastricus BTM 7 as probiotic: assessment of traits combined with principal component analysis. J Appl Microbiol 122:1310–1320. https://doi.org/10.1111/jam.13426
Reuben RC, Roy PC, Sarkar SL, Alam RU, Jahid IK (2019) Isolation, characterization, and assessment of lactic acid bacteria toward their selection as poultry probiotics. BMC Microbiol 19:253. https://doi.org/10.1186/s12866-019-1626-0
Du Toit M, Franz CMAP, Dicks LMT, Schillinger U, Haberer P, Warlies B, Ahrens F, Holzapfel WH (1998) Characterisation and selection of probiotic lactobacilli for a preliminary minipig feeding trial and their effect on serum cholesterol levels, faeces pH and faeces moisture content. Int J Food Microbiol 40:93–104. https://doi.org/10.1016/S0168-1605(98)00024-5
Perkins DN, Pappin DJC, Creasy DM, Cottrell JS (1999) Probability-based protein identification by searching sequence databases using mass spectrometry data. Electrophoresis 20:3551–3567. https://doi.org/10.1002/(sici)1522-2683(19991201)20:18%3C3551::aid-elps3551%3E3.0.co;2-2
Paim DRSF, Costa SDO, Walter EHM, Tonon RV (2016) Microencapsulation of probiotic jussara (Euterpe edulis M.) juice by spray drying. LWT - Food Sci Technol 74:21–25. https://doi.org/10.1016/j.lwt.2016.07.022
Haddaji N, Krifi B, Lagha R, Khouadja S, Bakhrouf A (2015) Effect of high temperature on viability of Lactobacillus casei and analysis of secreted and GroEL proteins profiles. African J Bacteriol Res 7:29–34
Brizuela MA, Serrano P, Ferez Y (2001) Studies on probiotics properties of two lactobacillus strains. Brazilian Arch Biol Technol 44:95–99. https://doi.org/10.1590/S1516-89132001000100013
Tajabadi N, Mardan M, Saari N, Mustafa S, Bahreini R, Manap MYA (2013) Identification of Lactobacillus plantarum, Lactobacillus pentosus and Lactobacillus fermentum from honey stomach of honeybee. Brazilian J Microbiol 44:717–722. https://doi.org/10.1590/S1517-83822013000300008
Capozzi V, Weidmann S, Fiocco D, Rieu A, Hols P, Guzzo J, Spano G (2011) Inactivation of a small heat shock protein affects cell morphology and membrane fluidity in Lactobacillus plantarum WCFS1. Res Microbiol 162:419–425. https://doi.org/10.1016/j.resmic.2011.02.010
De Angelis M, Di Cagno R, Huet C, Crecchio C, Fox PF, Gobbetti M (2004) Heat shock response in Lactobacillus plantarum. Appl Environ Microbiol 70:1336–1346. https://doi.org/10.1128/AEM.70.3.1336-1346.2004
Ferrando V, Quiberoni A, Reinheimer J, Suárez V (2016) Functional properties of Lactobacillus plantarum strains: a study in vitro of heat stress influence. Food Microbiol 54:154–161. https://doi.org/10.1016/j.fm.2015.10.003
Del Re B, Sgorbati B, Miglioli M, Palenzona D (2000) Adhesion, autoaggregation and hydrophobicity of 13 strains of Bifidobacterium longum. Lett Appl Microbiol 31:438–442. https://doi.org/10.1046/j.1365-2672.2000.00845.x
Moorman MA, Thelemann CA, Zhou S, Pestka JJ, Linz JE, Ryser ET (2008) Altered hydrophobicity and membrane composition in stress-adapted Listeria innocua. J Food Prot 71:182–185. https://doi.org/10.4315/0362-028X-71.1.182
de Souza BMS, Borgonovi TF, Casarotti SN, Todorov SD, Penna ALB (2019) Lactobacillus casei and Lactobacillus fermentum strains isolated from mozzarella Cheese: probiotic potential, safety, acidifying kinetic parameters and viability under gastrointestinal tract conditions. Probiotics Antimicrob Proteins 11:382–396. https://doi.org/10.1007/s12602-018-9406-y
Haddaji N, Mahdhi AK, Krifi B, Ismail MB, Bakhrouf A (2015) Change in cell surface properties of Lactobacillus casei under heat shock treatment. FEMS Microbiol Lett 362:1–7. https://doi.org/10.1093/femsle/fnv047
Leyer GJ, Johnson EA (1993) Acid adaptation induces cross-protection against environmental stresses in Salmonella typhimurium. Appl Environ Microbiol 59:1842–1847. https://doi.org/10.1128/aem.59.6.1842-1847.1993
Mafu AA, Roy D, Goulet J, Savoie L (1991) Characterization of physicochemical forces involved in adhesion of Listeria monocytogenes to surfaces. Appl Environ Microbiol 57:1969–1973. https://doi.org/10.1128/aem.57.7.1969-1973.1991
Assefa E, Beyene F, Santhanam A (2008) Effect of temperature and pH on the antimicrobial activity of inhibitory substances produced by lactic acid bacteria isolated from Ergo, an Ethiopian traditional fermented milk. African J Microbiol Res 2:229–234
Wang AN, Yi XW, Yu HF, Dong B, Sy Q (2009) Free radical scavenging activity of Lactobacillus fermentum in vitro and its antioxidative effect on growing-finishing pigs. J Appl Microbiol 107:1140–1148. https://doi.org/10.1111/j.1365-2672.2009.04294.x
Grujović M, Mladenović KG, Nikodijević DD, Čomić LR (2019) Autochthonous lactic acid bacteria—presentation of potential probiotics application. Biotechnol Lett 41:1319–1331. https://doi.org/10.1007/s10529-019-02729-8
Begley M, Gahan CGM, Hill C (2005) The interaction between bacteria and bile. FEMS Microbiol Rev 29:625–651. https://doi.org/10.1016/j.femsre.2004.09.003
Bove P, Russo P, Capozzi V, Gallone A, Fiocco D (2013) Lactobacillus plantarum passage through an oro-gastro-intestinal tract simulator: carrier matrix effect and transcriptional analysis of genes associated to stress and probiosis. Microbiol Res 168:351–359. https://doi.org/10.1016/j.micres.2013.01.004
Chen MJ, Tang HY, Chiang ML (2017) Effects of heat, cold, acid and bile salt adaptations on the stress tolerance and protein expression of kefir-isolated probiotic Lactobacillus kefiranofaciens M1. Food Microbiol 66:20–27. https://doi.org/10.1016/j.fm.2017.03.020
Wu R, Zhang W, Sun T, Wu J, Yue X, He M, Zhang H (2011) Proteomic analysis of responses of a new probiotic bacterium Lactobacillus casei Zhang to low acid stress. Int J Food Microbiol 147:181–187. https://doi.org/10.1016/j.ijfoodmicro.2011.04.003
Hernández-Alcántara AM, Wacher C, Llamas MG, López P, Chabela MLP (2018) Probiotic properties and stress response of thermotolerant lactic acid bacteria isolated from cooked meat products. LWT - Food Sci Technol 91:249–257. https://doi.org/10.1016/j.lwt.2017.12.063
Pan H, Agarwalla S, Moustakas DT, Moore JF, Stroud RM (2003) Structure of tRNA pseudouridine synthase TruB and its RNA complex: RNA recognition through a combination of rigid docking and induced fit. Proc Natl Acad Sci U S A 100:12648–12653. https://doi.org/10.1073/pnas.2135585100
Foster PG, Huang L, Santi DV, Stroud RM (2000) The structural basis for tRNA recognition and pseudouridine formation by pseudouridine synthase I. Nat Struct Biol 7:23–27. https://doi.org/10.1038/71219
Unciuleac MC, Goldgur Y, Shuman S (2015) Structure and two-metal mechanism of a eukaryal nick-sealing RNA ligase. Proc Natl Acad Sci USA 112:13868–13873. https://doi.org/10.1073/pnas.1516536112
Koistinen KM, Plumed-Ferrer C, Lehesranta SJ, Kärenlampi SO, Wright AV (2007) Comparison of growth-phase-dependent cytosolic proteomes of two Lactobacillus plantarum strains used in food and feed fermentations. FEMS Microbiol Lett 273:12–21. https://doi.org/10.1111/j.1574-6968.2007.00775.x
Feirer N, Fuqua C (2017) Pterin function in bacteria. Pteridines 28:23–36. https://doi.org/10.1515/pterid-2016-0012
Jiang M, Chen X, Guo ZF, Cao Y, Chen M, Guo Z (2008) Identification and characterization of (1R,6R)-2-succinyl-6-hydroxy-2,4- cyclohexadiene-1-carboxylate synthase in the menaquinone biosynthesis of Escherichia coli. Biochemistry 47:3426–3434. https://doi.org/10.1021/bi7023755
Acknowledgements
The authors gratefully thank the Director of CSIR-CFTRI, Mysore, for providing facilities to carry out this work. One of the authors, JB, gratefully acknowledges CSIR, New Delhi, for the award of Senior Research Fellowship.
Funding
This work was supported by the Council of Scientific Industrial Research-Senior research Fellowship (CSIR-SRF), New Delhi (File No: 31/5 (542)/2017-EMR-I).
Author information
Authors and Affiliations
Contributions
JB: Investigation, writing, editing, and reviewing, PK, SK: Investigation of 2D gel electrophoresis, DS: Conceptualization of protein-related work, SD: Supervision, Conceptualization, editing, and reviewing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bommasamudram, J., Kumar, P., Kapur, S. et al. Development of Thermotolerant Lactobacilli Cultures with Improved Probiotic Properties Using Adaptive Laboratory Evolution Method. Probiotics & Antimicro. Prot. 15, 832–843 (2023). https://doi.org/10.1007/s12602-021-09892-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-021-09892-3