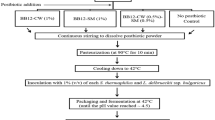
Abstract
Consumption of dairy products is one of the most natural ways to introduce probiotics. However, the beneficial effects of the probiotics might depend on the administration form. The aim of this study was to investigate the beneficial properties of two probiotic strains: Bifidobacterium animalis subsp. lactis (BB-12) and Lactobacillus acidophilus (LA-5) in different administration forms (capsules and yogurt). First, in vitro resistance to gastrointestinal condition, surface properties, and immunomodulation capacities were determined. Then, the anti-inflammatory properties of the probiotic strains administrated on yogurt or capsules were tested in a dinitrobenzene sulfonic acid (DNBS)-induced colitis mouse model. The survival rates of BB-12 and LA-5 strains to gastrointestinal conditions were slightly higher when yogurt was used as carrier. They showed most affinity to hexane (no-polar basic solvent) than ethyl-acetate (polar basic solvent). BB-12 showed the higher binding capacity to HT-29, Caco-2, and mucin. Both probiotic candidates suppress the secretion of IL-8 secretion by HT-29-TNF-α stimulated cells. Finally, administration of BB-12 and LA-5 strains improve colitis in mice. They protect against weight loss, inflammation, and hyperpermeability induced by DNBS. However, these anti-inflammatory effects were limited when mice were treated with the probiotic strain on a yogurt matrix. Overall results indicate that BB-12 and LA-5 positive properties are compromised depending on the matrix. Consequently, the selection of an appropriate matrix is an important criterion to conserve the positive benefits of these probiotic strains.





Similar content being viewed by others
Availability of Data and Material
All data and materials support their published claims and comply with field standards.
References
FAO/WHO (2002) Food and agriculture organization of the united nations/world health organization. Guidelines for the Evaluation of Probiotics in Food. Report of a Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food
Alves LL, Richards NSPS, Mattanna P, Andrade DF, Rezer APS, Milani LIG, Cruz AG, Faria JAF (2013) Cream cheese as a symbiotic food carrier using Bifidobacterium animalis Bb-12 and Lactobacillus acidophilus La-5 and inulin. Int J Dairy Technol 66(1):63–69. https://doi.org/10.1111/j.1471-0307.2012.00880.x
Magariños H, Selaive S, Costa M, Flores M, Pizarro O (2007) Viability of probiotic micro-organisms (Lactobacillus acidophilus La-5 and Bifidobacterium animalis subsp. lactis Bb-12) in ice cream. Int J Dairy Technol 60(2):128–134. https://doi.org/10.1111/j.1471-0307.2007.00307.x
Putta S, Yarla NS, Lakkappa DB, Imandi SB, Malla RR, Chaitanya AK, Chari BPV, Saka S, Vechalapu RR, Kamal MA, Tarasov VV, Chubarev VN, Siva Kumar K, Aliev G (2018) Chapter 2 - probiotics: supplements, food, pharmaceutical industry. In: Grumezescu AM, Holban AM (eds) Therapeutic, probiotic, and unconventional foods. Academic Press, pp 15-25. https://doi.org/10.1016/B978-0-12-814625-5.00002-9
Sheikhi A, Shakerian M, Giti H, Baghaeifar M, Jafarzadeh A, Ghaed V, Heibor MR, Baharifar N, Dadafarin Z, Bashirpour G (2016) Probiotic yogurt culture Bifidobacterium Animalis Subsp. Lactis BB-12 and Lactobacillus Acidophilus LA-5 modulate the cytokine secretion by peripheral blood mononuclear cells from patients with ulcerative colitis. Drug Res (Stuttg) 66(06):300–305. https://doi.org/10.1055/s-0035-1569414
Putt KK, Pei R, White HM, Bolling BW (2017) Yogurt inhibits intestinal barrier dysfunction in Caco-2 cells by increasing tight junctions. Food Funct 8(1):406–414. https://doi.org/10.1039/C6FO01592A
Bogovič Matijašić B, Obermajer T, Lipoglavšek L, Sernel T, Locatelli I, Kos M, Šmid A, Rogelj I (2016) Effects of synbiotic fermented milk containing Lactobacillus acidophilus La-5 and Bifidobacterium animalis subsp. lactis BB-12 on the fecal microbiota of adults with irritable bowel syndrome: a randomized double-blind, placebo-controlled trial. J Dairy Sci 99(7):5008–5021. https://doi.org/10.3168/jds.2015-10743
de Vrese M, Kristen H, Rautenberg P, Laue C, Schrezenmeir J (2011) Probiotic lactobacilli and bifidobacteria in a fermented milk product with added fruit preparation reduce antibiotic associated diarrhea and Helicobacter pylori activity. JJ Dairy Res 78(4):396–403. https://doi.org/10.1017/S002202991100063X
Sheu B-S, Cheng H-C, Kao A-W, Wang S-T, Yang Y-J, Yang H-B, Wu J-J (2006) Pretreatment with Lactobacillus- and Bifidobacterium-containing yogurt can improve the efficacy of quadruple therapy in eradicating residual Helicobacter pylori infection after failed triple therapy. Am J Clin Nutr 83(4):864–869. https://doi.org/10.1093/ajcn/83.4.864
Ruiz PA, Hoffmann M, Szcesny S, Blaut M, Haller D (2005) Innate mechanisms for Bifidobacterium lactis to activate transient pro-inflammatory host responses in intestinal epithelial cells after the colonization of germ-free rats. Immunology 115(4):441–450. https://doi.org/10.1111/j.1365-2567.2005.02176.x
Lee B, Yin X, Griffey SM, Marco ML (2015) Attenuation of colitis by Lactobacillus casei BL23 is dependent on the dairy delivery matrix. Appl Environ Microbiol 81(18):6425–6435. https://doi.org/10.1128/AEM.01360-15
Sharp JA, Digby M, Lefevre C, Mailer S, Khalil E, Topcic D, Auguste A, Kwek J, Brennan AJ, Familari M, Nicholas KR (2008) Chapter 2 - the comparative genomics of tammar wallaby and Cape fur seal lactation models to examine function of milk proteins. In: Thompson A, Boland M, Singh H (eds) Milk Proteins. Academic Press, San Diego, pp 55–79. https://doi.org/10.1016/B978-0-12-374039-7.00002-7
Roobab U, Batool Z, Manzoor MF, Shabbir MA, Khan MR, Aadil RM (2020) Sources, formulations, advanced delivery and health benefits of probiotics. Curr Opin Food Sci 32:17–28. https://doi.org/10.1016/j.cofs.2020.01.003
Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S, Calder PC, Sanders ME (2014) The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol 11(8):506–514. https://doi.org/10.1038/nrgastro.2014.66
Zucko J, Starcevic A, Diminic J, Oros D, Mortazavian AM, Putnik P (2020) Probiotic – friend or foe? Curr Opin Food Sci 32:45–49. https://doi.org/10.1016/j.cofs.2020.01.007
Paseephol T, Sherkat F (2009) Probiotic stability of yoghurts containing Jerusalem artichoke inulins during refrigerated storage. J Funct Foods 1(3):311–318. https://doi.org/10.1016/j.jff.2009.07.001
Zhai Z, Wang J, Huang B, Yin S (2019) Low-fat yogurt alleviates the pro-inflammatory cytokine IL-1β-induced intestinal epithelial barrier dysfunction. J Dairy Sci 102(2):976–984. https://doi.org/10.3168/jds.2018-15226
Cordeiro BF, Lemos L, Oliveira ER, Silva SH, Savassi B, Figueiroa A, Faria AMC, Ferreira E, Esmerino EA, Rocha RS, Freitas MQ, Silva MC, Cruz AG, do Carmo FLR, Azevedo V (2019) Prato cheese containing Lactobacillus casei 01 fails to prevent dextran sodium sulphate-induced colitis. Int Dairy J 99:104551. https://doi.org/10.1016/j.idairyj.2019.104551
Vinderola CG, Bailo N, Reinheimer JA (2000) Survival of probiotic microflora in Argentinian yoghurts during refrigerated storage. Food Res Int 33(2):97–102. https://doi.org/10.1016/s0963-9969(00)00011-9
FAO/WHO (2011) Fermented milks (CODEX STAN 243–2003). In Milk and Milk Products. World health organization Food and agriculture organization of the United Nations, Rome
Zendeboodi F, Khorshidian N, Mortazavian AM, da Cruz AG (2020) Probiotic: conceptualization from a new approach. Curr Opin Food Sci 32:103–123. https://doi.org/10.1016/j.cofs.2020.03.009
Bailey JR, Probert CSJ, Cogan TA (2011) Identification and characterisation of an iron-responsive candidate probiotic. PLoS One 6(10):e26507. https://doi.org/10.1371/journal.pone.0026507
Lee HM, Lee Y (2008) A differential medium for lactic acid-producing bacteria in a mixed culture. Lett Appl Microbiol 46(6):676–681. https://doi.org/10.1111/j.1472-765X.2008.02371.x
Vinderola CG, Reinheimer JA (2003) Lactic acid starter and probiotic bacteria: a comparative “in vitro” study of probiotic characteristics and biological barrier resistance. Food Res Int 36(9):895–904. https://doi.org/10.1016/S0963-9969(03)00098-X
Turpin W, Humblot C, Noordine ML, Thomas M, Guyot JP (2012) Lactobacillaceae and cell adhesion: genomic and functional screening. PLoS One 7(5):e38034-e38034. https://doi.org/10.1371/journal.pone.0038034
Kechaou N, Chain F, Gratadoux JJ, Blugeon S, Bertho N, Chevalier C, Le Goffic R, Courau S, Molimard P, Chatel JM, Langella P, Bermúdez-Humarán LG (2013) Identification of one novel candidate probiotic Lactobacillus plantarum strain active against influenza virus infection in mice by a large-scale screening. Appl Environ Microbiol 79(5):1491–1499. https://doi.org/10.1128/AEM.03075-12
Minekus M, Alminger M, Alvito P, Ballance S, Bohn T, Bourlieu C, Carrière F, Boutrou R, Corredig M, Dupont D, Dufour C, Egger L, Golding M, Karakaya S, Kirkhus B, Le Feunteun S, Lesmes U, Macierzanka A, Mackie A, Marze S, McClements DJ, Ménard O, Recio I, Santos CN, Singh RP, Vegarud GE, Wickham MSJ, Weitschies W, Brodkorb A (2014) A standardised static in vitro digestion method suitable for food – an international consensus. Food Funct 5(6):1113–1124. https://doi.org/10.1039/C3FO60702J
Martín R, Chain F, Miquel S, Lu J, Gratadoux JJ, Sokol H, Verdu EF, Bercik P, Bermúdez-Humarán LG, Langella P (2014) The commensal bacterium Faecalibacterium prausnitzii is protective in DNBS-induced chronic moderate and severe colitis models. Inflamm Bowel Dis 20(3):417–430. https://doi.org/10.1097/01.MIB.0000440815.76627.64
Barone M, Chain F, Sokol H, Brigidi P, Bermúdez-Humarán LG, Langella P, Martín R (2018) A versatile new model of chemically induced chronic colitis using an outbred murine strain. Front Microbiol 9(565). https://doi.org/10.3389/fmicb.2018.00565
Guarner F, Schaafsma GJ (1998) Probiotics. Int J Food Microbiol 39(3):237–238. https://doi.org/10.1016/S0168-1605(97)00136-0
Ren D-Y, Li C, Qin Y-Q, Yin R-L, Du S-W, Ye F, Liu H-F, Wang M-P, Sun Y, Li X, Tian M-Y, Jin N-Y (2013) Lactobacilli reduce chemokine IL-8 production in response to TNF-α and Salmonella challenge of Caco-2 cells. Biomed Res Int 2013:925219–925219. https://doi.org/10.1155/2013/925219
Shakirova L, Grube M, Gavare M, Auzina L, Zikmanis P (2013) Lactobacillus acidophilus La5 and Bifidobacterium lactis Bb12 cell surface hydrophobicity and survival of the cells under adverse environmental conditions. J Ind Microbiol Biotechnol 40(1):85–93. https://doi.org/10.1007/s10295-012-1204-z
Bellon-Fontaine MN, Rault J, van Oss CJ (1996) Microbial adhesion to solvents: a novel method to determine the electron-donor/electron-acceptor or Lewis acid-base properties of microbial cells. Colloid Surface B 7(1):47–53. https://doi.org/10.1016/0927-7765(96)01272-6
Jungersen M, Wind A, Johansen E, Christensen JE, Stuer-Lauridsen B, Eskesen D (2014) The science behind the probiotic strain Bifidobacterium animalis subsp. lactis BB-12®. Microorganisms 2(2):92–110. https://doi.org/10.3390/microorganisms2020092
Vemuri R, Shinde T, Shastri MD, Perera AP, Tristram S, Martoni CJ, Gundamaraju R, Ahuja KDK, Ball M, Eri R (2018) A human origin strain Lactobacillus acidophilus DDS-1 exhibits superior in vitro probiotic efficacy in comparison to plant or dairy origin probiotics. Int J Med Sci 15(9):840–848. https://doi.org/10.7150/ijms.25004
Li S-C, Hsu W-F, Chang J-S, Shih C-K (2019) Combination of Lactobacillus acidophilus and Bifidobacterium animalis subsp. lactis shows a stronger anti-inflammatory effect than individual strains in HT-29 cells. Nutrients 11(5):969. https://doi.org/10.3390/nu11050969
Imaoka A, Shima T, Kato K, Mizuno S, Uehara T, Matsumoto S, Setoyama H, Hara T, Umesaki Y (2008) Anti-inflammatory activity of probiotic Bifidobacterium: enhancement of IL-10 production in peripheral blood mononuclear cells from ulcerative colitis patients and inhibition of IL-8 secretion in HT-29 cells. World J Gastroenterol 14(16):2511–2516. https://doi.org/10.3748/wjg.14.2511
Ruiz L, Ruas-Madiedo P, Gueimonde M, de los Reyes-Gavilán CG, Margolles A, Sánchez B (2011) How do bifidobacteria counteract environmental challenges? Mechanisms involved and physiological consequences. Genes Nutr 6(3):307–318. https://doi.org/10.1007/s12263-010-0207-5
Tamime AY, Saarela M, Wszolek M, Ghoddousi H, Linares DM, Shah NP (2017) Production and maintaining viability of probiotic micro-organisms in dairy products. In: Probiotic Dairy Products, pp 67–164. https://doi.org/10.1002/9781119214137.ch4
Muñoz IB, Verruck S, Canella MHM, Dias CO, Amboni RDMC, Prudencio ES (2018) The use of soft fresh cheese manufactured from freeze concentrated milk as a novelty protective matrix on Bifidobacterium BB-12 survival under in vitro simulated gastrointestinal conditions. LWT 97:725–729. https://doi.org/10.1016/j.lwt.2018.08.009
Kim M, Oh S, Imm J-Y (2018) Buffering capacity of dairy powders and their effect on yoghurt quality. Korean J Food Sci Anim Resour 38(2):273–281. https://doi.org/10.5851/kosfa.2018.38.2.273
Ogita T, Nakashima M, Morita H, Saito Y, Suzuki T, Tanabe S (2011) Streptococcus thermophilus ST28 ameliorates colitis in mice partially by suppression of inflammatory Th17 cells. J Biomed Biotechnol 2011:378417–378419. https://doi.org/10.1155/2011/378417
Uriot O, Denis S, Junjua M, Roussel Y, Dary-Mourot A, Blanquet-Diot S (2017) Streptococcus thermophilus: from yogurt starter to a new promising probiotic candidate? J Funct Foods 37:74–89. https://doi.org/10.1016/j.jff.2017.07.038
Funding
GP scholarship was funded by CAMPUS France Hongrie (896369K). GP is a PhD student funded by the University of Kaposvar.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by GP, ETM, and RM. The first draft of the manuscript was written by GP, ETM, and RM. The manuscript was corrected by RM. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
All animal experiments performed received authorization numbers by the French ministry of Education nationale, enseignement supérieur et recherche (3445-2016010615159974 and 16744-201807061805486).
Code Availability
All used software application or custom code supports their published claims and complies with field standards.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PPT 82 kb)
Rights and permissions
About this article
Cite this article
Pápai, G., Torres-Maravilla, E., Chain, F. et al. The Administration Matrix Modifies the Beneficial Properties of a Probiotic Mix of Bifidobacterium animalis subsp. lactis BB-12 and Lactobacillus acidophilus LA-5. Probiotics & Antimicro. Prot. 13, 484–494 (2021). https://doi.org/10.1007/s12602-020-09702-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-020-09702-2