Abstract
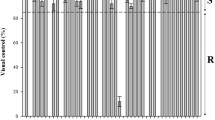
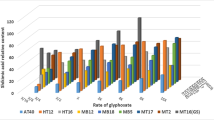
Amaranthus palmeri is a troublesome weed which is growing in importance worldwide. It causes serious competition in many crops. A. palmeri was introduced into Turkey 4–5 years ago and appears to adapt to different environmental conditions. Monoculture cropping systems and repeated use of the same herbicides have led to development of herbicide resistance in A. palmeri to several active ingredients including glyphosate, in USA and in South America. This study was conducted to investigate the evolution of glyphosate resistance in A. palmeri in Turkey. Seeds of 21 A. palmeri populations were collected in Turkish citrus fields where control problems with glyphosate were reported. A potentially glyphosate susceptible A. palmeri population was collected from a maize field (GS1 biotype). Seeds of putatively resistant and potentially susceptible (GS) biotypes were germinated and transplanted into large pots, and then allowed to grow in separate greenhouse chambers to obtain F2 generations. After carrying out a preliminary test experiment to exclude the most susceptible populations, a dose-response experiment was conducted in which glyphosate was applied at the 3–4 true leaf stage at 0, 332.5, 665, 1330, 2660, 5320, 10,640 and 21,280 g a.i. ha−1. Plants were harvested 21 days after treatment and dry weight was determined. Glyphosate applied at the recommended rate (1330 g a.i ha−1) controlled GS A. palmeri biotypes by more than 95% while controlling the GR biotypes at about 45%. Among those biotypes, GR1 and GR2 biotypes were confirmed to have an incipient resistance to glyphosate. The effect of glyphosate on shikimic acid accumulation was determined. Results showed that the GS2 biotype accumulated 3.1 and 1.56 times more shikimic acid than GR2 and GR1 biotypes which demonstrates that there is a lower accumulation of shikimic acid in the alleged resistant biotypes than in GS1. These findings demonstrate some increased tolerance of A. palmeri biotypes to glyphosate, which reinforces the need to implement integrated weed management to control this invasive plant in Turkey.





Similar content being viewed by others
References
Carvalho, S. J. P., Gonçalves Netto, A., Nicolai, M., Cavenaghi, A. L., López-Ovejero, R. F., & Christoffoleti, P. J. (2015). Detection of glyphosate-resistant palmer amaranth (Amaranthus palmeri) in agricultural areas of Mato Grosso, Brazil. Planta Daninha, 33, 579–586.
Chahal, P. S., Varanasi, V. K., Jugulam, M., & Jhala, A. J. (2017). Glyphosate-resistant palmer amaranth (Amaranthus palmeri) in Nebraska: Confirmation, EPSPS gene amplification, and response to POST corn and soybean herbicides. Weed Technology, 31, 80–93.
Chandi, A., Milla-Lewis, S. R., Giacomini, D., Westra, P., Preston, C., Jordan, D. L., York, A. C., Burton, J. D., & Whitaker, J. R. (2012). Inheritance of evolved glyphosate resistance in a North Carolina palmer amaranth (Amaranthus palmeri) biotype. International Journal of Agronomy. https://doi.org/10.1155/2012/176108.
Chatham, L. A., Bradley, K. W., Kruger, G. R., Martin, J. R., Owen, M. D. K., Peterson, D. E., Mithila, J., & Tranel, P. J. (2015). A multistate study of the association between glyphosate resistance and EPSPS gene amplification in waterhemp (Amaranthus tuberculatus). Weed Science, 63, 569–577.
Culpepper, A. S., & York, A. C. (1998). Weed management in glyphosate tolerant cotton. Journal of Cotton Science, 2, 174–185.
Culpepper, A. S., Grey, T. L., Vencill, W. K., Kichler, J. M., Webster, T. M., Brown, S. M., York, A. C., Davis, J. W., & Hanna, W. W. (2006). Glyphosate resistant palmer amaranth (Amaranthus palmeri) confirmed in Georgia. Weed Science, 54, 620–626.
Elmore, C. D. (1990). Weed identification guide. Southern Weed Science Society.
Eren, Ö., Doğan, M. N., Boz, Ö., Türkseven, S., & Özcan, R. (2016). Amaranthus palmeri L. In E. von Raab-Straube & T. Raus (Eds.), Euro+Med-Checklist Notulae, 6 (pp. 423–424). Willdenowia 437–441.
Fernandez-Escalada, M., Gil-Monreal, M., Zabalza, A., & Royuela, M. (2016). Characterization of the Amaranthus palmeri physiological response to glyphosate in susceptible and resistant populations. Journal of Agriculture Food Chemistry, 64, 95–106.
Fernandez-Escalada, M., Zulet-González, A., Gil-Monreal, M., Zabalza, A., Ravel, K., Gaines, T., & Royuela, M. (2017). Effects of EPSPS copy number variation (CNV) and glyphosate application on the aromatic and branched chain amino acid synthesis pathways in Amaranthus palmeri. Frontiers in Plant Science, 8, 1970. https://doi.org/10.3389/fpls.2017.01970.
Gaines, T. A., Zhang, W., Wang, D., Bukun, B., Chisholm, S. T., Shaner, D. L., Nissen, S. J., Patzoldt, W. L., Tranel, P. J., Culpepper, A. S., Grey, T. L., Webster, T. M., Vencill, W. K., Sammons, R. D., Jiang, J., Preston, C., Leach, J. E., & Westra, P. (2010). Gene amplification confers glyphosate resistance in Amaranthus palmeri. Proceedings of the National Academy of Sciences, 107, 1029–1034.
Gaines, T. A., Shaner, D. L., Ward, S. M., Leach, J. E., Preston, C., & Westra, P. (2011). Mechanism of resistance of evolved glyphosate-resistant palmer amaranth (Amaranthus palmeri). Journal of Agriculture Food Chemistry, 59, 5886–5889.
Ge, X., d’Avignon, D. A., Ackerman, J. J. H., & Sammons, R. D. (2010). Rapid vacuolar sequestration: The horseweed glyphosate resistance mechanism. Pest Management Science, 66, 345–348.
Ge, X., d’Avignon D. A., Ackerman J., Ostrander, E., & Sammons, R. D. (2013). Applications of 31P NMR spectroscopy to glyphosate studies in plants: Insights into cellular uptake and vacuolar sequestration correlated to herbicide resistance. In Herbicides: Biological activity, classification and health and environmental implications. Nova Science.
Harring, T., Streibig, J. C., & Husted, S. (1998). Accumulation of shikimic acid: A technique for screening glyphosate efficacy. Journal of Agricultural and Food Chemistry, 46, 4406–4412.
Heap, I. (2020a). The international herbicide-resistant weed database. Online. Sunday, May 10, 2020. Available www.weedscience.org.
Heap, I. (2020b) Criteria for confirmation of herbicide-resistant weeds—with specific emphasis on confirming low level resistance. Available online: http://www.weedscience.org/Documents/ResistanceCriterion.pdf. Accessed 21/10/2020.
Jha, P., & Norsworthy, J. K. (2005). Effect of soybean canopy formation and tillage on temporal emergence of Palmer amaranth. Proceedings Southern Weed Science Society, 58, 214.
Keeley, P. E., Carter, C. H., & Thullen, R. J. (1987). Influence of planting date on growth of palmer amaranth (Amaranthus palmeri). Weed Science, 35, 199–204.
Klingaman, T. E., & Oliver, L. R. (1994). Palmer amaranth (Amaranthus palmeri) interference in soybeans (Glycine max). Weed Science, 42, 523–527.
Leon, R. G. (2020). Of Amaranthus palmeri and other demons. ITEA-Información Técnica Económica Agraria, 116, 241–255. https://doi.org/10.12706/itea.2020.013.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 25, 4:402-408
Massinga, R. A., & Currie, R. S. (2002). Impact of palmer amaranth (Amaranthus palmeri) on corn (Zea mays) grain yield and yield and quality of forage. Weed Technology, 16, 532–536.
Mohseni-Moghadam, M., Schroeder, J., Heerema, R., & Ashigh, J. (2013). Resistance to glyphosate in palmer amaranth (Amaranthus palmeri) populations from New Mexico pecan orchards. Weed Technology, 27, 85–91.
Moss S.R, Clarke J.H, Blair A.M, Culley T.N, Read M.A, Ryan PJ, (1999). The occurrence of herbicide-resistant grass-weeds in the United Kingdom and a new system for designating resistance in screening assays. In: Proc. of the Brighton crop protection conference on weeds. United Kingdom: p. 179–184.
Nandula, V. K. (2010). Glyphosate resistance in crops and weeds: History, development, and management. Wiley.
Nandula, V. K., Reddy, K. N., Koger, C. H., Poston, D. H., Rimando, A. M., Duke, S. O., Bond, J. A., & Ribeiro, D. N. (2012). Multiple resistance to glyphosate and pyrithiobac in palmer amaranth (Amaranthus palmeri) from Mississippi and response to flumiclorac. Weed Science, 60, 179–188.
Nandula, V. K., Wright, A. A., Bond, J. A., Ray, J. D., Eubank, T. W., & Molin, W. T. (2014). EPSPS amplification in glyphosate-resistant spiny amaranth (Amaranthus spinosus): A case of gene transfer via interspecific hybridization from glyphosate-resistant Palmer amaranth (Amaranthus palmeri). Pest Management Science, 70, 1902–1909.
Norsworthy, J. K., Griffith, G. M., Scott, R. C., Smith, K. L., & Oliver, L. R. (2008). Confirmation and control of glyphosate-resistant palmer amaranth (Amaranthus palmeri) in Arkansas. Weed Technology, 22, 108–113.
Patzoldt, W. L., Hager, A. G., McCormick, J. S., & Tranel, P. J. (2006). A codon deletion confers resistance to herbicides inhibiting protoporphyrinogen oxidase. Proceedings of the National Academy of Sciences, 103, 12329–12334.
Pline-Srinc, W. (2006). Physiological mechanisms of glyphosate resistance. Weed Technology, 20, 290–300.
Powles, S. (2008). Evolved glyphosate-resistant weeds around the world: Lessons to be learnt. Management Science, 64, 360–365.
Recasens, J., & Conesa, J. A. (2011). Presencia de la mala hierba Amaranthus palmeri en el NE de la península Ibérica. Una amenaza como potencial invasora de cultivos extensivos de regadío. Boletín de Sanidad Vegetal. Plagas, 37, 129–132.
Ritz, C., & Streibig, J. C. (2005). Bioassay analysis using R. Journal of Statistical Software, 12, 1–22.
Salas, R. A., Dayan, F. E., Pan, Z., Watson, S. B., Dickson, J. W., Scott, R. C., & Burgos, N. R. (2012). EPSPS gene amplification in glyphosate-resistant Italian ryegrass (Lolium perenne ssp. multiflorum) from Arkansas. Pest Management Science, 68, 1223–1230.
Sarangi, D., Sandell, L. D., Knezevic, S. Z., Aulakh, J. S., Lindquist, J. L., Irmak, S., & Jhala, A. J. (2015). Confirmation and control of glyphosate-resistant common waterhemp (Amaranthus rudis) in Nebraska. Weed Technology, 29, 82–92.
Shaner, D. L., Lindenmeyer, R. B., & Ostlie, M. H. (2012). What have the mechanisms of resistance to glyphosate taught us? Pest Management Science, 68, 3–9.
Steckel, L. E., Main, C. L., Ellis, A. T., & Mueller, T. C. (2008). Palmer amaranth (Amaranthus palmeri) in Tennessee has low-level glyphosate resistance. Weed Technology, 22, 119–123.
Torra, J., Royo-Esnal, A., Romano, Y., Osuna, M. D., Leon, R. G., & Recasens, J. (2020). Amaranthus palmeri a new invasive weed in Spain with herbicide resistant biotypes. Agronomy, 10, 993. https://doi.org/10.3390/agronomy10070993.
VanGessel, M. J. (2001). Glyphosate-resistant horseweed from Delaware. Weed Science, 49, 703–705.
Varanasi, V. K., Betha, S., Thompson, C. R., & Jugulam, M. (2015). Target and non-target site multiple herbicide resistance in palmer amaranth (Amaranthus palmeri) from Kansas. Page 81 in proceedings of the 70th north central weed science society. North Central Weed Science Society.
Webster, T. M. (2005). Weed survey—southern states: Broadleaf crops subsection. Proceeding Southern Weed Science Society, 58, 291–304.
Webster, T. M., & Grey, T. L. (2015). Glyphosate-resistant palmer amaranth (Amaranthus palmeri) morphology, growth, and seed production in Georgia. Weed Science, 63, 264–272.
Whitaker, J. R., Burton, J. D., York, A. C., Jordan, D. L., & Chandi, A. (2013). Physiology of glyphosate-resistant and glyphosate-susceptible palmer amaranth (Amaranthus palmeri) biotypes collected from North Carolina. International Journal of Agronomy, 1−6.
Wiersma, S. T., Chisholm, S. T. Godar, A. S. Stahlman, P. W. Leach, J., & Westra, P. (2012). Gene amplification of EPSP synthase in glyphosate resistant Kochia scoparia, p. 97. Proc. of Western Soc. of Weed Sci. Western Society of Weed Science.
Acknowledgments
The authors thank Bayer Agriculture BVBA for funding and editorial assistance to the project. Also, thanks to Sushila Chaudhari, (Assistant Professor, Department of Horticulture, MSU) for her valuable remarks.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict interest
We hereby certify that our article title “Investigating glyphosate resistance in Amaranthus palmeri biotypes from Turkey” comprises original material. The enclosed manuscript is being submitted for possible publication in Phytoparasitica. This paper has not been published and is not being considered for publication elsewhere.
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mennan, H., Kaya-Altop, E., Belvaux, X. et al. Investigating glyphosate resistance in Amaranthus palmeri biotypes from Turkey. Phytoparasitica 49, 1043–1052 (2021). https://doi.org/10.1007/s12600-021-00910-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-021-00910-2