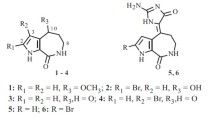
Abstract
Structural reinvestigation of corticatic acid A from a marine sponge, Petrosia sp., resulted in the revision of its structure, which conformed to the structural features of the biosynthetically related linear acetylenes isolated from Petrosia sp. In the process of isolating an authentic corticatic acid A, we isolated three new congeners and determined their structures. We also suggested that the structures of other corticatic acids need revision.






Similar content being viewed by others
References
Van Soest RWM, Fusetani N, Andersen RJ (1998) Straight-chain acetylenes as chemotaxonomic markers of the marine Haplosclerida. In: Watanabe Y, Fusetani N (eds) Sponge sciences. Springer, Tokyo, pp 3–30
Hitora Y, Takada K, Okada S, Ise Y, Matsunaga S (2011) (−)-Duryne and its homologues, cytotoxic acetylenes from a marine sponge Petrosia sp. J Nat Prod 74:1262–1267
Minto RE, Blacklock BJ (2008) Biosynthesis and function of polyacetylenes and allied natural products. Prog Lipid Res 47:233–306
Takayama K, Wang C, Besra GS (2005) Pathway to synthesis and processing of mycolic acid in Mycobacterium tuberculosis. Clin Microbiol Rev 18:81–101
Wright AE, McConnell OJ, Kohmoto S, Lui MS, Thompson W, Snader KM (1987) Duryne, a new cytotoxic agent from the marine sponge Cribrochalina dura. Tetrahedron Lett 28:1377–1380
Li H-Y, Matsunaga S, Fusetani N (1994) Corticatic acids A-C, antifungal acetylenic acids from the marine sponge, Petrosia corticata. J Nat Prod 57:1464–1467
Nishimura S, Matsunaga S, Shibazaki M, Suzuki K, Harada N, Naoki H, Fusetani N (2002) Corticatic acids D and E, polyacetylenic geranylgeranyltransferase type 1 inhibitors, from the marine sponge Petrosia corticata. J Nat Prod 65:1353–1356
Ohtani I, Kusumi T, Kashman Y, Kakisawa H (1991) High-field FT NMR application of Mosher’s Method. The absolute configuration of marine terpenoids. J Am Chem Soc 113:4092–4096
Seo Y, Cho KW, Rho J-R, Shin J (1998) Petrocortynes and petrosiacetylenes, novel polyacetylenes from a sponge of the genus Petrosia. Tetrahedron 54:447–462
Kim JS, Lim YJ, Im KS, Jung JH, Shim CJ, Lee CO, Hong J, Lee H (1999) Cytotoxic polyacetylenes from the marine sponge Petrosia sp. J Nat Prod 62:554–559
Okamoto C, Nakao Y, Fujita T, Iwashita T, van Soest RW, Fusetani N, Matsunaga S (2007) Cytotoxic C47-polyacetylene carboxylic acids from a marine sponge Petrosia sp. J Nat Prod 70:1816–1819
Amagata T (2010) Misassigned structures: case examples from the past decade. In: Mander L, Liu H-W (eds) Comprehensive natural products II: chemistry and biology, vol 2. Elsevier, Amsterdam, pp 581–621
Acknowledgments
This work was partly supported by a Grant-in-Aid for Scientific Research on Innovative Areas “Chemical Biology of Natural Products” and JSPS KAKENHI Grant Numbers 25252037, 25712024, and 25660163 from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
12562_2014_776_MOESM1_ESM.pdf
Supplementary material 1 (PDF 1081 kb). Electronic Supplementary Material Available: 1H NMR, 2D NMR, and FABMS data for corticatic acids and corticatynol. These materials are published in the online version
Rights and permissions
About this article
Cite this article
Takada, K., Okada, S. & Matsunaga, S. Structural reappraisal of corticatic acids, biologically active linear polyacetylenes, from a marine sponge of the genus Petrosia . Fish Sci 80, 1057–1064 (2014). https://doi.org/10.1007/s12562-014-0776-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-014-0776-0