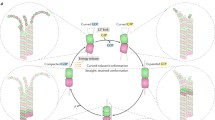
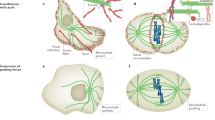
Abstract
Tubulins are essential proteins, which are conserved across all eukaryotic species. They polymerize to form microtubules, cytoskeletal components of paramount importance for cellular mechanics. The microtubules combine an extraordinarily high flexural rigidity and a non-equilibrium behavior, manifested in their intermittent assembly and disassembly. These chemically fueled dynamics allow microtubules to generate significant pushing and pulling forces at their ends to reposition intracellular organelles, remodel membranes, bear compressive forces, and transport chromosomes during cell division. In this article, we review classical and recent studies, which have allowed the quantification of microtubule-generated forces. The measurements, to which we owe most of the quantitative information about microtubule forces, were carried out in biochemically reconstituted systems in vitro. We also discuss how mathematical and computational modeling has contributed to the interpretations of these results and shaped our understanding of the mechanisms of force production by tubulin polymerization and depolymerization.







Similar content being viewed by others
References
Akiyoshi B, Sarangapani KK, Powers AF, Nelson CR, Reichow SL, Arellano-Santoyo H, Gonen T, Ranish JA, Asbury CL, Biggins S (2010) Tension directly stabilizes reconstituted kinetochore-microtubule attachments. Nature 468:576–579. https://doi.org/10.1038/nature09594
Alexandrova VV, Anisimov MN, Zaitsev AV, Mustyatsa VV, Popov VV, Ataullakhanov FI, Gudimchuk NB (2022) Theory of tip structure–dependent microtubule catastrophes and damage-induced microtubule rescues. Proc Natl Acad Sci 119:e2208294119. https://doi.org/10.1073/pnas.2208294119
Alkemade C, Wierenga H, Volkov VA, Preciado López M, Akhmanova A, ten Wolde PR, Dogterom M, Koenderink GH (2022) Cross-linkers at growing microtubule ends generate forces that drive actin transport. Proc Natl Acad Sci 119:e2112799119. https://doi.org/10.1073/pnas.2112799119
Amini R, Volkov VA, Dogterom M (2023) Dynamic instability of force-generating bacterial microtubules. https://doi.org/10.1101/2023.08.02.551647
Asbury CL, Gestaut DR, Powers AF, Franck AD, Davis TN (2006) The Dam1 kinetochore complex harnesses microtubule dynamics to produce force and movement. Proc Natl Acad Sci 103:9873–9878. https://doi.org/10.1073/pnas.0602249103
Beckett D, Voth GA (2023) Unveiling the catalytic mechanism of GTP hydrolysis in microtubules. Proc Natl Acad Sci 120:e2305899120. https://doi.org/10.1073/pnas.2305899120
Bowne-Anderson H, Zanic M, Kauer M, Howard J (2013) Microtubule dynamic instability: a new model with coupled GTP hydrolysis and multistep catastrophe. BioEssays News Rev Mol Cell Dev Biol 35:452–461. https://doi.org/10.1002/bies.201200131
Brangwynne CP, MacKintosh FC, Kumar S, Geisse NA, Talbot J, Mahadevan L, Parker KK, Ingber DE, Weitz DA (2006) Microtubules can bear enhanced compressive loads in living cells because of lateral reinforcement. J Cell Biol 173:733–741. https://doi.org/10.1083/jcb.200601060
Carlier MF, Pantaloni D (1981) Kinetic analysis of guanosine 5’-triphosphate hydrolysis associated with tubulin polymerization. Biochemistry 20:1918–1924
Castle BT, McCubbin S, Prahl LS, Bernens JN, Sept D, Odde DJ (2017) Mechanisms of kinetic stabilization by the drugs paclitaxel and vinblastine. Mol Biol Cell 28:1238–1257. https://doi.org/10.1091/mbc.E16-08-0567
Chrétien D, Fuller SD, Karsenti E (1995) Structure of growing microtubule ends: two-dimensional sheets close into tubes at variable rates. J Cell Biol 129:1311–1328
Coombes CE, Yamamoto A, Kenzie MR, Odde DJ, Gardner MK (2013) Evolving tip structures can explain age-dependent microtubule catastrophe. Curr Biol 23:1342–1348. https://doi.org/10.1016/j.cub.2013.05.059
Coue M, Lombillo VA, McIntosh JR (1991) Microtubule depolymerization promotes particle and chromosome movement in vitro. J Cell Biol 112:1165–1175
Daga RR, Yonetani A, Chang F (2006) Asymmetric microtubule pushing forces in nuclear centering. Curr Biol 16:1544–1550. https://doi.org/10.1016/j.cub.2006.06.026
De Simone A, Spahr A, Busso C, Gönczy P (2018) Uncovering the balance of forces driving microtubule aster migration in C. elegans zygotes. Nat Commun 9:938. https://doi.org/10.1038/s41467-018-03118-x
Desai A, Mitchison TJ (1997) Microtubule polymerization dynamics. Annu Rev Cell Dev Biol 13:83–117. https://doi.org/10.1146/annurev.cellbio.13.1.83
Dogterom M, Kerssemakers JW, Romet-Lemonne G, Janson ME (2005) Force generation by dynamic microtubules. Curr Opin Cell Biol 17:67–74. https://doi.org/10.1016/j.ceb.2004.12.011
Dogterom M, Yurke B (1997) Measurement of the force-velocity relation for growing microtubules. Science 278:856–860. https://doi.org/10.1126/science.278.5339.856
Driver JW, Geyer EA, Bailey ME, Rice LM, Asbury CL (2017) Direct measurement of conformational strain energy in protofilaments curling outward from disassembling microtubule tips. eLife 6. https://doi.org/10.7554/eLife.28433
Duellberg, C., Cade, N.I., Holmes, D., Surrey, T., 2016. The size of the EB cap determines instantaneous microtubule stability. eLife 5:e13470. https://doi.org/10.7554/eLife.13470
Efremov A, Grishchuk EL, McIntosh JR, Ataullakhanov FI (2007) In search of an optimal ring to couple microtubule depolymerization to processive chromosome motions. Proc Natl Acad Sci U S A 104:19017–19022. https://doi.org/10.1073/pnas.0709524104
Farmer VJ, Zanic M (2023) Beyond the GTP-cap: elucidating the molecular mechanisms of microtubule catastrophe. BioEssays 45:2200081. https://doi.org/10.1002/bies.202200081
Franck AD, Powers AF, Gestaut DR, Gonen T, Davis TN, Asbury CL (2007) Tension applied through the Dam1 complex promotes microtubule elongation providing a direct mechanism for length control in mitosis. Nat Cell Biol 9:832–837. https://doi.org/10.1038/ncb1609
Gard DL, Kirschner MW (1987) A microtubule-associated protein from Xenopus eggs that specifically promotes assembly at the plus-end. J Cell Biol 105:2203–2215. https://doi.org/10.1083/jcb.105.5.2203
Gardner MK, Zanic M, Gell C, Bormuth V, Howard J (2011) Depolymerizing kinesins Kip3 and MCAK shape cellular microtubule architecture by differential control of catastrophe. Cell 147:1092–1103. https://doi.org/10.1016/j.cell.2011.10.037
Garzon-Coral C, Fantana HA, Howard J (2016) A force-generating machinery maintains the spindle at the cell center during mitosis. Science 352:1124–1127. https://doi.org/10.1126/science.aad9745
Gittes F, Meyhöfer E, Baek S, Howard J (1996) Directional loading of the kinesin motor molecule as it buckles a microtubule. Biophys J 70:418–429. https://doi.org/10.1016/S0006-3495(96)79585-1
Gittes F, Mickey B, Nettleton J, Howard J (1993) Flexural rigidity of microtubules and actin filaments measured from thermal fluctuations in shape. J Cell Biol 120:923–934. https://doi.org/10.1083/jcb.120.4.923
Grigoriev I, Gouveia SM, van der Vaart B, Demmers J, Smyth JT, Honnappa S, Splinter D, Steinmetz MO, Putney JW, Hoogenraad CC, Akhmanova A (2008) STIM1 is a MT-plus-end-tracking protein involved in remodeling of the ER. Curr Biol 18:177–182. https://doi.org/10.1016/j.cub.2007.12.050
Grishchuk E, McIntosh R, Molodtsov M, Ataullakhanov F (2012) Force generation by dynamic microtubule polymers. Comprehensive Biophysics. https://doi.org/10.1016/B978-0-12-374920-8.00409-4
Grishchuk EL, Efremov AK, Volkov VA, Spiridonov IS, Gudimchuk N, Westermann S, Drubin D, Barnes G, McIntosh JR, Ataullakhanov FI (2008a) The Dam1 ring binds microtubules strongly enough to be a processive as well as energy-efficient coupler for chromosome motion. Proc Natl Acad Sci 105:15423–15428. https://doi.org/10.1073/pnas.0807859105
Grishchuk EL, McIntosh JR (2006) Microtubule depolymerization can drive poleward chromosome motion in fission yeast. EMBO J 25:4888–4896
Grishchuk EL, Molodtsov MI, Ataullakhanov FI, McIntosh JR (2005) Force production by disassembling microtubules. Nature 438:384–388. https://doi.org/10.1038/nature04132
Grishchuk EL, Spiridonov IS, Volkov VA, Efremov A, Westermann S, Drubin D, Barnes G, Ataullakhanov FI, McIntosh JR (2008b) Different assemblies of the DAM1 complex follow shortening microtubules by distinct mechanisms. Proc Natl Acad Sci U S A 105:6918–6923. https://doi.org/10.1073/pnas.0801811105
Gudimchuk NB, McIntosh JR (2021) Regulation of microtubule dynamics, mechanics and function through the growing tip. Nat Rev Mol Cell Biol 22:777–795. https://doi.org/10.1038/s41580-021-00399-x
Gudimchuk NB, Ulyanov EV, O’Toole E, Page CL, Vinogradov DS, Morgan G, Li G, Moore JK, Szczesna E, Roll-Mecak A, Ataullakhanov FI, Richard McIntosh J (2020) Mechanisms of microtubule dynamics and force generation examined with computational modeling and electron cryotomography. Nat Commun 11:3765. https://doi.org/10.1038/s41467-020-17553-2
Guo Y, Li D, Zhang S, Yang Y, Liu J-J, Wang X, Liu C, Milkie DE, Moore RP, Tulu US, Kiehart DP, Hu J, Lippincott-Schwartz J, Betzig E, Li D (2018) Visualizing intracellular organelle and cytoskeletal interactions at nanoscale resolution on millisecond timescales. Cell. https://doi.org/10.1016/j.cell.2018.09.057
Hamilton GE, Helgeson LA, Noland CL, Asbury CL, Dimitrova YN, Davis TN (2020) Reconstitution reveals two paths of force transmission through the kinetochore. eLife 9:e56582. https://doi.org/10.7554/eLife.56582
Hill TL (1987) Linear aggregation theory in cell biology, Springer Series in Molecular Biology. Springer, New York, NY. https://doi.org/10.1007/978-1-4612-4736-4
Hotani H, Miyamoto H (1990) Dynamic features of microtubules as visualized by dark-field microscopy. Adv Biophys 26:135–156. https://doi.org/10.1016/0065-227x(90)90010-q
Huis in’t Veld PJ, Volkov VA, Stender ID, Musacchio A, Dogterom M (2019) Molecular determinants of the Ska-Ndc80 interaction and their influence on microtubule tracking and force-coupling. eLife 8:e49539. https://doi.org/10.7554/eLife.49539
Hyman AA, Salser S, Drechsel DN, Unwin N, Mitchison TJ (1992) Role of GTP hydrolysis in microtubule dynamics: information from a slowly hydrolyzable analogue. GMPCPP Mol Biol Cell 3:1155–1167
Iwaki M, Wickham SF, Ikezaki K, Yanagida T, Shih WM (2016) A programmable DNA origami nanospring that reveals force-induced adjacent binding of myosin VI heads. Nat Commun 7:13715. https://doi.org/10.1038/ncomms13715
Janson ME, Dogterom M (2004) Scaling of microtubule force-velocity curves obtained at different tubulin concentrations. Phys Rev Lett 92:248101. https://doi.org/10.1103/PhysRevLett.92.248101
Joglekar AP, Hunt AJ (2002) A simple, mechanistic model for directional instability during mitotic chromosome movements. Biophys J 83:42–58. https://doi.org/10.1016/S0006-3495(02)75148-5
Kaneko T, Itoh TJ, Hotani H (1998) Morphological transformation of liposomes caused by assembly of encapsulated tubulin and determination of shape by microtubule-associated proteins (MAPs)11Edited by M. F. Moody. J Mol Biol 284:1671–1681. https://doi.org/10.1006/jmbi.1998.2251
Kanfer G, Peterka M, Arzhanik VK, Drobyshev AL, Ataullakhanov FI, Volkov VA, Kornmann B (2017) CENP-F couples cargo to growing and shortening microtubule ends. Mol Biol Cell 28:2400–2409. https://doi.org/10.1091/mbc.E16-11-0756
Kerssemakers JWJ, Laura Munteanu E, Laan L, Noetzel TL, Janson ME, Dogterom M (2006) Assembly dynamics of microtubules at molecular resolution. Nature 442:709–712. https://doi.org/10.1038/nature04928
Koshland DE, Mitchison TJ, Kirschner MW (1988) Polewards chromosome movement driven by microtubule depolymerization in vitro. Nature 331:499–504. https://doi.org/10.1038/331499a0
Laan L, Husson J, Munteanu EL, Kerssemakers JWJ, Dogterom M (2008) Force-generation and dynamic instability of microtubule bundles. Proc Natl Acad Sci 105:8920–8925. https://doi.org/10.1073/pnas.0710311105
Laan L, Pavin N, Husson J, Romet-Lemonne G, van Duijn M, López MP, Vale RD, Jülicher F, Reck-Peterson SL, Dogterom M (2012) Cortical dynein controls microtubule dynamics to generate pulling forces that position microtubule asters. Cell 148:502–514. https://doi.org/10.1016/j.cell.2012.01.007
Li Y, Kučera O, Cuvelier D, Rutkowski DM, Deygas M, Rai D, Pavlovič T, Vicente FN, Piel M, Giannone G, Vavylonis D, Akhmanova A, Blanchoin L, Théry M (2023) Compressive forces stabilize microtubules in living cells. Nat Mater 22:913–924. https://doi.org/10.1038/s41563-023-01578-1
Lomakin AJ, Kraikivski P, Semenova I, Ikeda K, Zaliapin I, Tirnauer JS, Akhmanova A, Rodionov V (2011) Stimulation of the CLIP-170--dependent capture of membrane organelles by microtubules through fine tuning of microtubule assembly dynamics. Mol Biol Cell 22:4029–4037. https://doi.org/10.1091/mbc.E11-03-0260
Lomakin AJ, Semenova I, Zaliapin I, Kraikivski P, Nadezhdina E, Slepchenko BM, Akhmanova A, Rodionov V (2009) CLIP-170-dependent capture of membrane organelles by microtubules initiates minus-end directed transport. Dev Cell 17:323–333. https://doi.org/10.1016/j.devcel.2009.07.010
Lombillo VA, Stewart RJ, McIntosh JR (1995) Minus-end-directed motion of kinesin-coated microspheres driven by microtubule depolymerization. Nature 373:161–164. https://doi.org/10.1038/373161a0
Maleki NA, Huis in’t Veld PJ, Akhmanova A, Dogterom M, Volkov VA (2022) Estimation of microtubule-generated forces using a DNA origami nanospring. J Cell Sci 136:jcs260154. https://doi.org/10.1242/jcs.260154
Margolin G, Gregoretti IV, Cickovski TM, Li C, Shi W, Alber MS, Goodson HV (2012) The mechanisms of microtubule catastrophe and rescue: implications from analysis of a dimer-scale computational model. Mol Biol Cell 23:642–656. https://doi.org/10.1091/mbc.E11-08-0688
McIntosh JR, Grishchuk EL, Morphew MK, Efremov AK, Zhudenkov K, Volkov VA, Cheeseman IM, Desai A, Mastronarde DN, Ataullakhanov FI (2008) Fibrils connect microtubule tips with kinetochores: a mechanism to couple tubulin dynamics to chromosome motion. Cell 135:322–333. https://doi.org/10.1016/j.cell.2008.08.038
McIntosh JR, Molodtsov MI, Ataullakhanov FI (2012) Biophysics of mitosis. Q Rev Biophys 45:147–207. https://doi.org/10.1017/S0033583512000017
McIntosh JR, O’Toole E, Morgan G, Austin J, Ulyanov E, Ataullakhanov F, Gudimchuk N (2018) Microtubules grow by the addition of bent guanosine triphosphate tubulin to the tips of curved protofilaments. J Cell Biol 217:2691–2708. https://doi.org/10.1083/jcb.201802138
McIntosh JR, O’Toole E, Zhudenkov K, Morphew M, Schwartz C, Ataullakhanov FI, Grishchuk EL (2013) Conserved and divergent features of kinetochores and spindle microtubule ends from five species. J Cell Biol 200:459–474. https://doi.org/10.1083/jcb.201209154
McIntosh JR, Volkov V, Ataullakhanov FI, Grishchuk EL (2010) Tubulin depolymerization may be an ancient biological motor. J Cell Sci 123:3425–3434. https://doi.org/10.1242/jcs.067611
Miller MP, Asbury CL, Biggins S (2016) A TOG protein confers tension sensitivity to kinetochore-microtubule attachments. Cell 165:1428. https://doi.org/10.1016/j.cell.2016.04.030
Miranda JJ, De Wulf P, Sorger PK, Harrison SC (2005) The yeast DASH complex forms closed rings on microtubules. Nat Struct Mol Biol 12:138–143. https://doi.org/10.1038/nsmb896
Mogilner A, Oster G (1999) The polymerization ratchet model explains the force-velocity relation for growing microtubules. Eur Biophys J 28:235–242. https://doi.org/10.1007/s002490050204
Mogilner A, Oster G (1996) Cell motility driven by actin polymerization. Biophys J 71:3030–3045. https://doi.org/10.1016/S0006-3495(96)79496-1
Molodtsov MI, Ermakova EA, Shnol EE, Grishchuk EL, McIntosh JR, Ataullakhanov FI (2005a) A molecular-mechanical model of the microtubule. Biophys J 88:3167–3179. https://doi.org/10.1529/biophysj.104.051789
Molodtsov MI, Grishchuk EL, Efremov AK, McIntosh JR, Ataullakhanov FI (2005b) Force production by depolymerizing microtubules: a theoretical study. Proc Natl Acad Sci U S A 102:4353–4358. https://doi.org/10.1073/pnas.0501142102
Molodtsov MI, Mieck C, Dobbelaere J, Dammermann A, Westermann S, Vaziri A (2016) A force-induced directional switch of a molecular motor enables parallel microtubule bundle formation. Cell 167:539–552.e14. https://doi.org/10.1016/j.cell.2016.09.029
Murray LE, Kim H, Rice LM, Asbury CL (2022) Working strokes produced by curling protofilaments at disassembling microtubule tips can be biochemically tuned and vary with species. eLife 11:e83225. https://doi.org/10.7554/eLife.83225
Nogales E, Downing KH, Amos LA, Löwe J (1998) Tubulin and FtsZ form a distinct family of GTPases. Nat Struct Biol 5:451–458. https://doi.org/10.1038/nsb0698-451
Oguchi Y, Uchimura S, Ohki T, Mikhailenko SV, Ishiwata S (2011) The bidirectional depolymerizer MCAK generates force by disassembling both microtubule ends. Nat Cell Biol 13:846–852. https://doi.org/10.1038/ncb2256
Olmsted JB, Borisy GG (1973) Microtubules. Annu Rev Biochem 42:507–540. https://doi.org/10.1146/annurev.bi.42.070173.002451
Pegoraro AF, Janmey P, Weitz DA (2017) Mechanical properties of the cytoskeleton and cells. Cold Spring Harb Perspect Biol 9:a022038. https://doi.org/10.1101/cshperspect.a022038
Peskin CS, Odell GM, Oster GF (1993) Cellular motions and thermal fluctuations: the Brownian ratchet. Biophys J 65:316–324
Polley S, Müschenborn H, Terbeck M, De Antoni A, Vetter IR, Dogterom M, Musacchio A, Volkov VA, Huis in’t Veld PJ (2023) Stable kinetochore-microtubule attachment requires loop-dependent Ndc80-Ndc80 binding. EMBO J 42:e112504. https://doi.org/10.15252/embj.2022112504
Ramey VH, Wang H-W, Nakajima Y, Wong A, Liu J, Drubin D, Barnes G, Nogales E (2011) The Dam1 ring binds to the E-hook of tubulin and diffuses along the microtubule. Mol Biol Cell 22:457–466. https://doi.org/10.1091/mbc.e10-10-0841
Rodríguez-García R, Volkov VA, Chen C-Y, Katrukha EA, Olieric N, Aher A, Grigoriev I, López MP, Steinmetz MO, Kapitein LC, Koenderink G, Dogterom M, Akhmanova A (2020) Mechanisms of motor-independent membrane remodeling driven by dynamic microtubules. Curr Biol 30:972–987.e12. https://doi.org/10.1016/j.cub.2020.01.036
Romani A, Scarpa A (1992) Regulation of cell magnesium. Arch Biochem Biophys 298:1–12. https://doi.org/10.1016/0003-9861(92)90086-c
Schaedel L, Triclin S, Chrétien D, Abrieu A, Aumeier C, Gaillard J, Blanchoin L, Théry M, John K (2019) Lattice defects induce microtubule self-renewal. Nat Phys 15:830–838. https://doi.org/10.1038/s41567-019-0542-4
Schek HT, Gardner MK, Cheng J, Odde DJ, Hunt AJ (2007) Microtubule assembly dynamics at the nanoscale. Curr Biol 17:1445–1455. https://doi.org/10.1016/j.cub.2007.07.011
Schek HT, Hunt AJ (2005) Micropatterned structures for studying the mechanics of biological polymers. Biomed Microdevices 7:41–46. https://doi.org/10.1007/s10544-005-6170-z
Schmidt JC, Arthanari H, Boeszoermenyi A, Dashkevich NM, Wilson-Kubalek EM, Monnier N, Markus M, Oberer M, Milligan RA, Bathe M, Wagner G, Grishchuk EL, Cheeseman IM (2012) The kinetochore-bound Ska1 complex tracks depolymerizing microtubules and binds to curved protofilaments. Dev Cell 23:968–980. https://doi.org/10.1016/j.devcel.2012.09.012
Schwietert F, Volkov VA, Huis In’t Veld PJ, Dogterom M, Musacchio A, Kierfeld J (2022) Strain stiffening of Ndc80 complexes attached to microtubule plus ends. Biophys J 121:4048–4062. https://doi.org/10.1016/j.bpj.2022.09.039
Simmons RM, Finer JT, Chu S, Spudich JA (1996) Quantitative measurements of force and displacement using an optical trap. Biophys J 70:1813–1822. https://doi.org/10.1016/S0006-3495(96)79746-1
Sissoko GB, Tarasovetc EV, Grishchuk EL, Cheeseman IM (2023) Higher-order assembly is a regulatory switch that promotes outer kinetochore recruitment. https://doi.org/10.1101/2023.05.22.541649
Son J, Orkoulas G, Kolomeisky AB (2005) Monte Carlo simulations of rigid biopolymer growth processes. J Chem Phys 123:124902. https://doi.org/10.1063/1.2013248
Stamenović D, Mijailovich SM, Tolić-Nørrelykke IM, Chen J, Wang N (2002) Cell prestress. II. Contribution of microtubules. Am J Physiol Cell Physiol 282:C617–C624. https://doi.org/10.1152/ajpcell.00271.2001
Stukalin EB, Kolomeisky AB (2004) Simple growth models of rigid multifilament biopolymers. J Chem Phys 121:1097–1104. https://doi.org/10.1063/1.1759316
Svoboda K, Block SM (1994a) Biological applications of optical forces. Annu Rev Biophys Biomol Struct 23:247–285. https://doi.org/10.1146/annurev.bb.23.060194.001335
Svoboda K, Block SM (1994b) Force and velocity measured for single kinesin molecules. Cell 77:773–784. https://doi.org/10.1016/0092-8674(94)90060-4
Tanaka K, Kitamura E, Kitamura Y, Tanaka TU (2007) Molecular mechanisms of microtubule-dependent kinetochore transport toward spindle poles. J Cell Biol 178:269–281. https://doi.org/10.1083/jcb.200702141
Tarasovetc EV, Allu PK, Wimbish RT, DeLuca JG, Cheeseman IM, Black BE, Grishchuk EL (2021) Permitted and restricted steps of human kinetochore assembly in mitotic cell extracts. Mol Biol Cell 32:1241–1255. https://doi.org/10.1091/mbc.E20-07-0461
Tolić-Nørrelykke IM, Sacconi L, Stringari C, Raabe I, Pavone FS (2005) Nuclear and division-plane positioning revealed by optical micromanipulation. Curr Biol 15:1212–1216. https://doi.org/10.1016/j.cub.2005.05.052
Tolić-Nørrelykke IM, Sacconi L, Thon G, Pavone FS (2004) Positioning and elongation of the fission yeast spindle by microtubule-based pushing. Curr Biol 14:1181–1186. https://doi.org/10.1016/j.cub.2004.06.029
Torvi, J.R., Wong, J., Serwas, D., Moayed, A., Drubin, D.G., Barnes, G., 2022. Reconstitution of kinetochore motility and microtubule dynamics reveals a role for a kinesin-8 in establishing end-on attachments. eLife 11, e78450. https://doi.org/10.7554/eLife.78450
Tran PT, Joshi P, Salmon ED (1997) How tubulin subunits are lost from the shortening ends of microtubules. J Struct Biol 118:107–118. https://doi.org/10.1006/jsbi.1997.3844
Tran PT, Marsh L, Doye V, Inoué S, Chang F (2001) A mechanism for nuclear positioning in fission yeast based on microtubule pushing. J Cell Biol 153:397–411. https://doi.org/10.1083/jcb.153.2.397
Traut TW (1994) Physiological concentrations of purines and pyrimidines. Mol Cell Biochem 140:1–22. https://doi.org/10.1007/BF00928361
Trushko A, Schäffer E, Howard J (2013) The growth speed of microtubules with XMAP215-coated beads coupled to their ends is increased by tensile force. Proc Natl Acad Sci U S A 110:14670–14675. https://doi.org/10.1073/pnas.1218053110
van Doorn GS, Tanase C, Mulder BM, Dogterom M (2000) On the stall force for growing microtubules. Eur Biophys J EBJ 29:2–6. https://doi.org/10.1007/s002490050245
VanBuren V, Cassimeris L, Odde DJ (2005) Mechanochemical model of microtubule structure and self-assembly kinetics. Biophys J 89:2911–2926. https://doi.org/10.1529/biophysj.105.060913
VanBuren V, Odde DJ, Cassimeris L (2002) Estimates of lateral and longitudinal bond energies within the microtubule lattice. Proc Natl Acad Sci U S A 99:6035–6040. https://doi.org/10.1073/pnas.092504999
Vleugel M, Kok M, Dogterom M (2016) Understanding force-generating microtubule systems through in vitro reconstitution. Cell Adh Migr 10:475–494. https://doi.org/10.1080/19336918.2016.1241923
Volkov VA, Grissom PM, Arzhanik VK, Zaytsev AV, Renganathan K, McClure-Begley T, Old WM, Ahn N, McIntosh JR (2015) Centromere protein F includes two sites that couple efficiently to depolymerizing microtubules. J Cell Biol 209:813–828. https://doi.org/10.1083/jcb.201408083
Volkov VA, Huis in t Veld PJ, Dogterom M, Musacchio A (2018) Multivalency of NDC80 in the outer kinetochore is essential to track shortening microtubules and generate forces. eLife 7:e36764. https://doi.org/10.7554/eLife.36764
Volkov VA, Zaytsev AV, Gudimchuk N, Grissom PM, Gintsburg AL, Ataullakhanov FI, McIntosh JR, Grishchuk EL (2013) Long tethers provide high-force coupling of the Dam1 ring to shortening microtubules. Proc Natl Acad Sci U S A 110:7708–7713. https://doi.org/10.1073/pnas.1305821110
Wang S, Romano FB, Field CM, Mitchison TJ, Rapoport TA (2013) Multiple mechanisms determine ER network morphology during the cell cycle in Xenopus egg extracts. J Cell Biol 203:801–814. https://doi.org/10.1083/jcb.201308001
Waterman-Storer CM, Gregory J, Parsons SF, Salmon ED (1995) Membrane/microtubule tip attachment complexes (TACs) allow the assembly dynamics of plus ends to push and pull membranes into tubulovesicular networks in interphase Xenopus egg extracts. J Cell Biol 130:1161–1169. https://doi.org/10.1083/jcb.130.5.1161
Waterman-Storer CM, Salmon ED (1998) Endoplasmic reticulum membrane tubules are distributed by microtubules in living cells using three distinct mechanisms. Curr Biol 8:798–807. https://doi.org/10.1016/S0960-9822(98)70321-5
Westermann S, Avila-Sakar A, Wang H-W, Niederstrasser H, Wong J, Drubin DG, Nogales E, Barnes G (2005) Formation of a dynamic kinetochore- microtubule interface through assembly of the Dam1 ring complex. Mol Cell 17:277–290. https://doi.org/10.1016/j.molcel.2004.12.019
Westermann S, Wang H-W, Avila-Sakar A, Drubin DG, Nogales E, Barnes G (2006) The Dam1 kinetochore ring complex moves processively on depolymerizing microtubule ends. Nature 440:565–569. https://doi.org/10.1038/nature04409
Wieczorek M, Bechstedt S, Chaaban S, Brouhard GJ (2015) Microtubule-associated proteins control the kinetics of microtubule nucleation. Nat Cell Biol 17:907–916. https://doi.org/10.1038/ncb3188
Wimbish RT, DeLuca JG (2020) Hec1/Ndc80 tail domain function at the kinetochore-microtubule interface. Front Cell Dev Biol 8:43. https://doi.org/10.3389/fcell.2020.00043
Yaffe MP, Stuurman N, Vale RD (2003) Mitochondrial positioning in fission yeast is driven by association with dynamic microtubules and mitotic spindle poles. Proc Natl Acad Sci 100:11424–11428. https://doi.org/10.1073/pnas.1534703100
Zakharov P, Gudimchuk N, Voevodin V, Tikhonravov A, Ataullakhanov FI, Grishchuk EL (2015) Molecular and mechanical causes of microtubule catastrophe and aging. Biophys J 109:2574–2591. https://doi.org/10.1016/j.bpj.2015.10.048
Zhao T, Graham OS, Raposo A, Johnston DS (2012) Growing microtubules push the oocyte nucleus to polarize the drosophila dorsal-ventral axis. Science 336:999–1003. https://doi.org/10.1126/science.1219147
Acknowledgements
We thank J. R. McIntosh for the critical reading of the manuscript and helpful suggestions.
Funding
Analysis of in vitro studies of microtubules was supported by the Russian Science Foundation grant # 23-74-00007, https://rscf.ru/project/23-74-00007/. Analysis of mathematical and computational models was supported by the Scientific and Educational Mathematical Center “Sofia Kovalevskaya Northwestern Center for Mathematical Research” (agreement no. 075-02-2023-937, 16.02.2023).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gudimchuk, N.B., Alexandrova, V.V. Measuring and modeling forces generated by microtubules. Biophys Rev 15, 1095–1110 (2023). https://doi.org/10.1007/s12551-023-01161-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-023-01161-7