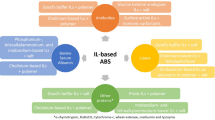
Abstract
Mixtures of ionic liquids (ILs) have attracted our attention because of their extraordinary performances in extraction technologies and in absorbing large amount of CO2 gas. It has been observed that when two or more ILs are mixed in different proportions, a new solvent is obtained which is much better than that of each component of ILs from which the mixture is obtained. Within a mixture of ILs, several unidentified interactions occur among several ions which give rise to unique solvent properties to the mixture. Herein, in this review, we have highlighted the utilization of the advantageous properties of the IL mixtures in protein stability studies. This approach is exceptional and opens new directions to the use of ILs in biotechnology.



Similar content being viewed by others
References
Attri P, Venkatesu P, Kumar A (2012) Water and a protic ionic liquid acted as refolding additives for chemically denatured enzymes. Org Biomol Chem 10:7475–7478
Attri P, Venkatesu P (2012) Influence of protic ionic liquids on the structure and stability of succinylated Con A. Int J Biol Macromol 51:119–128
Attri P, Jha I, Choi EH, Venkatesu P (2014) Variation in the structural changes of myoglobin in the presence of several protic ionic liquid. Int J Biol Macromol 69:114–123
Bisht M, Venkatesu P (2017) Influence of cholinium-based ionic liquids on the structural stability and activity of α-chymotrypsin. New J Chem 41:13902–13911
Bisht M, Kumar A, Venkatesu P (2015) Analysis of the driving force that rule the stability of lysozyme in alkylammonium-based ionic liquids. Int J Biol Macromol 81:1074–1081
Bisht M, Jha I, Venkatesu P (2016) Comprehensive evaluation of biomolecular interactions between protein and amino acid based-ionic liquids: a comparable study between [Bmim][Br] and [Bmim][Gly] ionic liquids. Chemistry Select 1:3510–3519
Bisht M, Mondal D, Pereira MM et al (2017) Long-term protein packaging in cholinium-based ionic liquids: improved catalytic activity and enhanced stability of cytochrome c against multiple stresses. Green Chem 19:4900–4911
Bi YH, Duan ZQ, Li XQ et al (2015) Introducing biobased ionic liquids as the nonaqueous media for enzymatic synthesis of phosphatidylserine. Agric Food Chem 63:1558–1561
Bates ED, Mayton RD, Ntai I et al (2002) CO2 capture by a task-specific ionic liquid. J Am Chem Soc 124:926–927
Benedetto A, Ballone P (2016) Room temperature ionic liquids meet biomolecules: a microscopic view of structure and dynamics. ACS Sus Chem Eng 4:392–412
Chen FF, Huang K, Zhou Y et al (2016) Multi-molar absorption of CO2 by the activation of carboxylate groups in amino acid ionic liquids. Angew Chem 128:7282–7286
Desai R K, Streefland M, Wijffels R H, et al (2016) Extraction of proteins with ABS. In: Freire M. (eds) Ionic-liquid-based aqueous biphasic systems. Green Chemistry and Sustainable Technology. Springer, Berlin, Heidelberg
Daschakraborty S, Biswas R (2014) Dielectric relaxation in ionic liquids: role of ion-ion and ion-dipole interactions, and effects of heterogeneity. J Chem Phys 140:014504
Greaves TL, Drummond CJ (2015) Protic ionic liquids: evolving structure–property relationships and expanding applications. Chem Rev 115:11379–11448
Gupta BS, Taha M, Lee MJ (2016) Extraction of an active enzyme by self-buffering ionic liquids: a green medium for enzymatic research. RSC Adv 6:18567–18576
Gebbie MA, Smith AM, Dobbs HA et al (2017) Long range electrostatic forces in ionic liquids. Chem Commun 53:1214–1224
Ghosh S, Parui S, Jana B et al (2015) Ionic liquid induced dehydration and domain closure in lysozyme: FCS and MD simulation. J Chem Phys 143:125103
Hayes R, Imberti S Dr., Warr GG et al (2013) The nature of hydrogen bonding in protic ionic liquids. Angew Chem 52:4623–4627
Hunt PA, Kirchner B, Welton T (2006) Characterising the electronic structure of ionic liquids: an examination of the 1-butyl-3-methylimidazolium chloride ion pair. Chem Eur J 12:6762–6775
Hunt PA, Gould IR (2006) Structural characterization of the 1-butyl-3-methylimidazolium chloride ion pair using ab initio methods. J Phys Chem A 110:2269–2282
Jha I, Venkatesu P (2016) Unprecedented improvement in the stability of Haemoglobin in the presence of promising green solvent 1-allyl-3-methylimidazolium chloride. ACS Sustain Chem Eng 4:413−421
Jha I, Attri P, Venkatesu P (2014) Unexpected effects of the alteration of structure and stability of myoglobin and hemoglobin in ammonium-based ionic liquids. Phys Chem Chem Phys 16:5514–5526
Jha I, Rani A, Venkatesu P (2017) Sustained stability and activity of lysozyme in choline chloride against pH induced denaturation. ACS Sus Chem Eng 5:8344–8355
Jha I, Bisht M, Venkatesu P (2016) Does 1-allyl-3-methylimidazolium chloride acts as a biocompatible solvent for stem bromelain? J Phys Chem B 120:5625–5633
Kossmann S, Thar J, Kirchner B, Hunt PA (2006) Cooperativity in ionic liquids. J Chem Phys 124:174506–174512
Kumar A, Rani A, Venkatesu P (2015) A comparative study of the Hofmeister series of anions of the ionic salts and ionic liquids on the stability of α-chymotrypsin. New J Chem 39:938–952
Kumar A, Rani A, Venkatesu P et al (2014a) Quantitative evaluation of the ability of ionic liquids to offset the cold-induced unfolding of proteins. Phys Chem Chem Phys 16:15806–15810
Kumar A, Venkatesu P (2013) Prevention of insulin self-aggregation by protic ionic liquid. RSC Adv 3:362–367
Kumar A, Bisht M, Venkatesu P (2016) Exploring the structure and stability of amino acids and glycine peptides in biocompatible ionic liquids. RSC Adv 6:18763–18777
Kumar A, Venkatesu P, Taha M, Lee MJ (2014b) Thermodynamic contribution of amino acids in ionic liquids towards protein stability. Curr Biochem Eng 1:125–140
Kumar A, Venkatesu P (2014) Does the stability of proteins in ionic liquids obeys the Hofmeister series? Int J Biol Macromol 63:244–253
Kumar A, Venkatesu P (2012) Overview of the stability of α-chymotrypsin in different solvent media. Chem Rev 112:4283–4307
Kumar A, Bisht M, Venkatesu P (2017) Biocompatibility of ionic liquids towards protein stability: a comprehensive overview on the current understanding and their implications. Int J Biol Macromol 96:611–651
Katoh R, Hara M, Tsuzuki S (2008) Ion pair formation in [bmim]I ionic liquids. J Phys Chem B 112:15426–15430
Kareem MA, Mjalli FS, Hashim Mohd A, AlNashef IM (2010) Phosphonium-based ionic liquids analogues and their physical properties. J Chem Eng Data 55:4632–4637
Losada-Pérez P, Khorshid M, Renner FU (2016) Interactions of aqueous imidazolium-based ionic liquid mixtures with solid-supported phospholipid vesicles. PLoS One 11:e0163518
Lee SY, Khoiroh I, Ooi CW et al (2017) Recent advances in protein extraction using ionic liquid-based aqueous two-phase systems. Sep Purif Rev 46:291–304
Lei Z (2017) Introduction: Ionic Liquids. Chem Rev 117:6633–6635
Lei Z, Dai C, Chen B (2014) Gas solubility in ionic liquids. Chem Rev 114:1289–1326
Matsumoto K, Hagiwara R, Mazej Z, Benkič P, Žemva B (2006) Crystal structures of frozen room temperature ionic liquids, 1-ethyl-3-methylimidazolium tetrafluoroborate (EMImBF4), hexafluoroniobate (EMImNbF6) and hexafluorotantalate (EMImTaF6), determined by low-temperature X-ray diffraction. Solid State Sci 8:1250–1257
Mai NL, Ahn K, Koo YM (2014) Methods for recovery of ionic liquids—a review. Process Biochem 49:872–881
Mojumdar SS, Chowdhury R, Chattoraj S et al (2012) Role of ionic liquid on the conformational dynamics in the native, molten globule, and unfolded states of cytochrome C: a fluorescence correlation spectroscopy study. J Phys Chem B 116:12189–12198
Niedermeyer H, Hallett JP, Villar-Garcia IJ et al (2012) Mixtures of ionic liquids. Chem Soc Rev 41:7780–7802
Nakajima K, Nakanishi S, Lísal M, Kimura K (2017) Surface structures of binary mixture of ionic liquids. J Mol Liq 230:542–549
Podgoršek A, Jacquemin J, Pádua AAH et al (2016) Mixing enthalpy for binary mixtures containing ionic liquids. Chem Rev 116:6075−6106
Park S, Kazlauskas RJ (2003) Biocatalysis in ionic liquids—advantages beyond green technology. Cur Opin Biotechnol 14:432–437
Reddy PM, Umapathi R, Venkatesu P (2015) A green approach to offset the perturbation action of 1-butyl-3-methylimidazolium iodide on α-chymotrypsin. Phys Chem Chem Phys 17:184–190
Ramdin M, de Loos TW, Vlugt TJH (2012) State-of-the-art of CO2 capture with ionic liquids. Ind Eng Chem Res 51:8149–8177
Sasmal DK, Mondal T, Mojumdar SS et al (2011) An FCS study of unfolding and refolding of CPM-labeled human serum albumin: role of ionic liquid. J Phys Chem B 115:13075–13083
Schröder C (2017) Proteins in ionic liquids: current status of experiments and simulations. Top Curr Chem 375–425
Stolarska O, Pawlowska-Zygarowicz A, Soto A, Rodríguez H et al (2017) Mixtures of ionic liquids as more efficient media for cellulose dissolution. Carbohydr Polym 178:2017
Sivapragasam M, Moniruzzaman M, Goto M (2016) Recent advances in exploiting ionic liquids for biomolecules: solubility, stability and applications. Biotechnol J 11:1000–1013
Smith EL, Abbott AP, Ryder KS (2014) Deep eutectic solvents (DESs) and their applications. Chem Rev 114:11060–11082
Taha ME, Silva FA, Quental MV, Ventura Sónia PM, Freirea MG, Coutinho JAP (2014) Good's buffers as a basis for developing self-buffering and biocompatible ionic liquids for biological research. Green Chem 16:3149–3159
Vijayraghavan R, Pas SJ, Izgorodina EI et al (2013) Diamino protic ionic liquids for CO2 capture. Phys Chem Chem Phys 15:19994–19999
Weight A K, Larson A M, Langer R S, et al (2015) U S Patent No. WO 2015038811 A2, Liquid protein formulations containing ionic liquids
Wu BP, Wen Q, Xu H, Yang Z (2014) Insights into the impact of deep eutectic solvents on horseradish peroxidase: activity, stability and structure. J Mol Catal B Enzym 101:101–107
Wang B, Qin L, Mu T et al (2017) Are ionic liquids chemically stable? Chem Rev 117:7113–7131
Wang M, Rao N, Wang M et al (2018) Properties of ionic liquid mixtures of [NH2e-mim][BF4] and [bmim][BF4] as absorbents for CO2 capture. Greenhouse Gas Sci Technol 0:1–10
Xia Z, Das P, Shakhnovich E et al (2012) Collapse of unfolded proteins in a mixture of denaturants. J Am Chem Soc 134:18266–18274
Xiao D, Rajian JR, Li S et al (2006) Additivity in the optical Kerr effect spectra of binary ionic liquid mixtures: implications for nanostructural organization. J Phys Chem B 110:16174–16178
Xiao D, Rajian JR, Hines LG, Li S et al (2008) Nanostructural organization and anion effects in the optical Kerr effect spectra of binary ionic liquid mixtures. J Phys Chem B 112:13316–13325
Xu P, Du P, Zong MH et al (2016) Combination of deep eutectic solvent and ionic liquid to improve biocatalytic reduction of 2-octanone with Acetobacter pasteurianus GIM1.158 cell. Sci Rep 6:1–10
Zhao H (2016) Protein stabilization and enzyme activation in ionic liquids: specific ion effects. J Chem Technol Biotechnol 91:25–50
Zhang S, Zhang J, Zhang Y et al (2017) Nanoconfined ionic liquids. Chem Rev 117:6755–6833
Zahn S, Uhlig F, Thar J, Spickermann C et al (2008) Intermolecular forces in an ionic liquid ([Mmim][Cl]) versus those in a typical salt (NaCl). Angew Chem 47:3639–3641
Zeng S, Zhang X, Bai L et al (2017) Ionic-liquid-based CO2 capture systems: structure, interaction and process. Chem Rev 117:9625–9673
Acknowledgements
We gratefully acknowledge the Council of Scientific and Industrial Research, New Delhi, India, through the Grant No. 01(2871)/17/EMR-II) for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Awanish Kumar declares that he has no conflicts of interest. Pannuru Venkatesu declares that he has no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
This article is part of a Special Issue on “Ionic Liquids and Biomolecules” edited by Antonio Benedetto and Hans-Joachim Galla.
Rights and permissions
About this article
Cite this article
Kumar, A., Venkatesu, P. Innovative aspects of protein stability in ionic liquid mixtures. Biophys Rev 10, 841–846 (2018). https://doi.org/10.1007/s12551-018-0411-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-018-0411-x