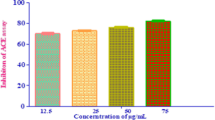
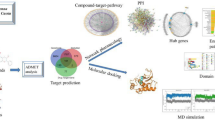
Abstract
Understanding the molecular mode of action of natural product is a key step for developing drugs from them. In this regard, this study is aimed to understand the molecular-level interactions of chemical constituents of Clerodendrum colebrookianum Walp., with anti-hypertensive drug targets using computational approaches. The plant has ethno-medicinal importance for the treatment of hypertension and reported to show activity against anti-hypertensive drug targets—Rho-associated coiled-coil protein kinase (ROCK), angiotensin-converting enzyme, and phosphodiesterase 5 (PDE5). Docking studies showed that three chemical constituents (acteoside, martinoside, and osmanthuside β6) out of 21 reported from the plant to interact with the anti-hypertensive drug targets with good glide score. In addition, they formed H-bond interactions with the key residues Met156/Met157 of ROCK I/ROCK II and Gln817 of PDE5. Further, molecular dynamics (MD) simulation of protein–ligand complexes suggest that H-bond interactions between acteoside/osmanthuside β6 and Met156/Met157 (ROCK I/ROCK II), acteoside and Gln817 (PDE5) were stable. The present investigation suggests that the anti-hypertensive activity of the plant is due to the interaction of acteoside and osmanthuside β6 with ROCK and PDE5 drug targets. The identified molecular mode of binding of the plant constituents could help to design new drugs to treat hypertension.




Similar content being viewed by others
Abbreviations
- ACE:
-
Angiotensin-converting enzyme
- MD:
-
Molecular dynamics
- PDE5:
-
Phosphodiesterase 5
- PE:
-
Potential energy
- RMSD:
-
Root mean square deviations
- RMSF:
-
Root mean square fluctuations
- ROCK:
-
Rho-associated coiled-coil protein kinase
References
Olayiwola A, Vernon H, Hugh S (1991) Conservation of medicinal plants. Cambridge University Press, Cambridge
Ekor M (2014) The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol 4:177
Atanasov AG, Waltenberger B, Pferschy-Wenzig EM, Linder T, Wawrosch C, Uhrin P, Temml V, Wang L, Schwaiger S, Heiss EH, Rollinger JM, Schuster D, Breuss JM, Bochkov V, Mihovilovic MD, Kopp B, Bauer R, Dirsch VM, Stuppner H (2015) Discovery and resupply of pharmacologically active plant-derived natural products: a review. Biotechnol Adv 33(8):1582–1614
Newman DJ, Cragg GM (2007) Natural products as sources of new drugs over the last 25 years. J Nat Prod 70(3):461–477
Shrivastava N, Patel T (2007) Clerodendrum and healthcare: an overview—Part II phytochemistry and biotechnology. Med Aromat Plant Sci Biotechnol 1:209–223
Shrivastava N, Patel T (2007) Clerodendrum and healthcare: an overview Med Aromat Plant Sci. Biotechnol 1:142
Nath CN, Bordoloi DN (1990) Clerodendrum colebrookianum, a folk remedy for the treatment of hypertension in Northeastern India. Int J Pharmacogn 29(2):127–129
Beevers G, Lip GY, O’Brien E (2001) ABC of hypertension: the pathophysiology of hypertension. BMJ 322(7291):912–916
Anchala R, Kannuri NK, Pant H, Khan H, Franco OH, Di Angelantonio E, Prabhakaran D (2014) Hypertension in India: a systematic review and meta-analysis of prevalence, awareness, and control of hypertension. J Hypertens 32(6):1170–1177
Brown NJ, Vaughan DE (1998) Angiotensin-converting enzyme inhibitors. Circulation 97(14):1411–1420
Kuyper LM, Khan NA (2014) Atenolol vs nonatenolol beta-blockers for the treatment of hypertension: a meta-analysis. Can J Cardiol 30(5 Suppl):S47–S53
Sica DA (2004) Minoxidil: an underused vasodilator for resistant or severe hypertension. J Clin Hypertens (Greenwich) 6(5):283–287
Erickson AL, DeGrado JR, Fanikos JR (2010) Clevidipine: a short-acting intravenous dihydropyridine calcium channel blocker for the management of hypertension. Pharmacotherapy 30(5):515–528
Sanoski CA (2009) Aliskiren: an oral direct renin inhibitor for the treatment of hypertension. Pharmacotherapy 29(2):193–212
Kjeldsen SE, Stalhammar J, Hasvold P, Bodegard J, Olsson U, Russell D (2010) Effects of losartan vs candesartan in reducing cardiovascular events in the primary treatment of hypertension. J Hum Hypertens 24(4):263–273
Yang H, Jiang B, Hou AJ, Lin ZW, Sun HD (2000) Colebroside A, a new diglucoside of fatty acid ester of glycerin from Clerodendrum colebrookianum. J Asian Nat Prod Res 2(3):177–185
Janmoni K, Sureshkumar SS, Mohamed LK (2012) Clerodendrum colebrookianum Walp.: a potential folk medicinal plant of North East India. Asian J Pharm Biol Res 2(4):256–261
Rajlakshmi D, Banerjee SK, Sood S, Maulik SK (2003) In-vitro and in vivo antioxidant activity of different extracts of the leaves of Clerodendron colebrookianum Walp in the rat. J Pharm Pharmacol 55(12):1681–1686
Lokesh D, Amitsankar D (2012) Evaluation of mechanism for antihypertensive action of Clerodendrum colebrookianum Walp., used by folklore healers in north-east India. J Ethnopharmacol 143(1):207–212
Kang DG, Lee YS, Kim HJ, Lee YM, Lee HS (2003) Angiotensin converting enzyme inhibitory phenylpropanoid glycosides from Clerodendron trichotomum. J Ethnopharmacol 89(1):151–154
Thai KM, Le DP, Tran NV, Nguyen TT, Tran TD, Le MT (2015) Computational assay of Zanamivir binding affinity with original and mutant influenza neuraminidase 9 using molecular docking. J Theor Biol 385:31–39
Chinta G, Ramya Chandar Charles M, Klopcic I, Sollner Dolenc M, Periyasamy L, Selvaraj Coumar M (2015) In silico and in vitro investigation of the Piperine’s male contraceptive effect: docking and molecular dynamics simulation studies in androgen-binding protein and androgen receptor. Plant Med 81(10):804–812
Durrant JD, McCammon JA (2011) Molecular dynamics simulations and drug discovery. BMC Biol 9:71
Fancui M (2013) Molecular dynamics simulation of VEGFR2 with sorafenib and other urea-substituted aryloxy compounds. J Theor Chem 2013:7
Schrödinger LLC (2011) Maestro, version 9.2. Schrödinger LLC
Kaminski GA, Friesner RA, Tirado-rives J, Jorgensen WL (2001) Evaluation and reparametrization of the OPLS-AA force field for proteins via comparison with accurate quantum chemical calculations on peptides. J Phys Chem 105:6474–6487
Jacobs M, Hayakawa K, Swenson L, Bellon S, Fleming M, Taslimi P, Doran J (2006) The structure of dimeric ROCK I reveals the mechanism for ligand selectivity. J Biol Chem 281(1):260–268
Akama T, Dong C, Virtucio C, Sullivan D, Zhou Y, Zhang YK, Rock F, Freund Y, Liu L, Bu W, Wu A, Fan XQ, Jarnagin K (2013) Linking phenotype to kinase: identification of a novel benzoxaborole hinge-binding motif for kinase inhibition and development of high-potency rho kinase inhibitors. J Pharmacol Exp Ther 347(3):615–625
Natesh R, Schwager SL, Sturrock ED, Acharya KR (2003) Crystal structure of the human angiotensin-converting enzyme-lisinopril complex. Nature 421(6922):551–554
Sung BJ, Hwang KY, Jeon YH, Lee JI, Heo YS, Kim JH, Moon J, Yoon JM, Hyun YL, Kim E, Eum SJ, Park SY, Lee JO, Lee TG, Ro S, Cho JM (2003) Structure of the catalytic domain of human phosphodiesterase 5 with bound drug molecules. Nature 425(6953):98–102
Schrödinger LLC (2011) Prime, version 9.2. Schrödinger LLC
Schrödinger LLC (2011) Glide, version 9.2. Schrödinger LLC
Raj U, Varadwaj PK (2015) Flavonoids as multi-target inhibitors for proteins associated with Ebola virus: in silico discovery using virtual screening and molecular docking studies. Interdiscip Sci 8(2):132–141
Van Der Spoel D, Lindahl E, Hess B, Groenhof G, Mark AE, Berendsen HJ (2005) GROMACS: fast, flexible, and free. J Comput Chem 26(16):1701–1718
Malde AK, Zuo L, Breeze M, Stroet M, Poger D, Nair PC, Oostenbrink C, Mark AE (2011) An automated force field topology builder (ATB) and repository: version 1.0. J Chem Theory Comput 7(12):4026–4037
Walter RPS, Philippe HH, Ilario GT, Alan EM, Salomon RB, Jens F, Andrew ET, Thomas H, Peter K, Wilfred FVG (1999) The GROMOS biomolecular simulation program package. J Phys Chem 103(19):3596–3607
Schmid N, Eichenberger AP, Choutko A, Riniker S, Winger M, Mark AE, van Gunsteren WF (2011) Definition and testing of the GROMOS force-field versions 54A7 and 54B7. Eur Biophys J 40(7):843–856
Hess B, Kutzner C, van der Spoel D, Lindahl E (2008) GROMACS 4: algorithms for highly efficient, load-balanced, and scalable molecular simulation. J Chem Theory Comput 4(3):435–447
Abraham MJ, Gready JE (2011) Optimization of parameters for molecular dynamics simulation using smooth particle-mesh Ewald in GROMACS 4.5. J Comput Chem 32(9):2031–2040
Mueller BK, Mack H, Teusch N (2005) Rho kinase, a promising drug target for neurological disorders. Nat Rev Drug Discov 4(5):387–398
Morgan-Fisher M, Wewer UM, Yoneda A (2013) Regulation of ROCK activity in cancer. J Histochem Cytochem 61(3):185–198
Rath N, Olson MF (2012) Rho-associated kinases in tumorigenesis: re-considering ROCK inhibition for cancer therapy. EMBO Rep 13(10):900–908
Wirth A (2010) Rho kinase and hypertension. Biochim Biophys Acta 1802(12):1276–1284
Liao JK, Seto M, Noma K (2007) Rho kinase (ROCK) inhibitors. J Cardiovasc Pharmacol 50(1):17–24
Hartmann S, Ridley AJ, Lutz S (2015) The function of Rho-associated kinases ROCK1 and ROCK2 in the pathogenesis of cardiovascular disease. Front Pharmacol 6:276
Tripathi SK, Muttineni R, Singh SK (2013) Extra precision docking, free energy calculation and molecular dynamics simulation studies of CDK2 inhibitors. J Theor Biol 334:87–100
Mishra RK, Alokam R, Singhal SM, Srivathsav G, Sriram D, Kaushik-Basu N, Manvar D, Yogeeswari P (2014) Design of novel rho kinase inhibitors using energy based pharmacophore modeling, shape-based screening, in silico virtual screening, and biological evaluation. J Chem Inf Model 54(10):2876–2886
Shen M, Zhou S, Li Y, Pan P, Zhang L, Hou T (2013) Discovery and optimization of triazine derivatives as ROCK1 inhibitors: molecular docking, molecular dynamics simulations and free energy calculations. Mol BioSyst 9(3):361–374
Qin J, Lei B, Xi L, Liu H, Yao X (2010) Molecular modeling studies of Rho kinase inhibitors using molecular docking and 3D-QSAR analysis. Eur J Med Chem 45(7):2768–2776
Chen H, Li S, Hu Y, Chen G, Jiang Q, Tong R, Zang Z, Cai L (2016) An integrated in silico method to discover novel Rock1 inhibitors: multi- complex-based pharmacophore, molecular dynamics simulation and hybrid protocol virtual screening. Comb Chem High Throughput Screen 19(1):36–50
Clarke NE, Turner AJ (2012) Angiotensin-converting enzyme 2: the first decade. Int J Hypertens 2012:307315
Kass DA, Champion HC, Beavo JA (2007) Phosphodiesterase type 5: expanding roles in cardiovascular regulation. Circ Res 101(11):1084–1095
Wilkins MR, Wharton J, Grimminger F, Ghofrani HA (2008) Phosphodiesterase inhibitors for the treatment of pulmonary hypertension. Eur Respir J 32(1):198–209
Corbin JD (2004) Mechanisms of action of PDE5 inhibition in erectile dysfunction. Int J Impot Res 16(Suppl 1):S4–S7
Card GL, England BP, Suzuki Y, Fong D, Powell B, Lee B, Luu C, Tabrizizad M, Gillette S, Ibrahim PN, Artis DR, Bollag G, Milburn MV, Kim SH, Schlessinger J, Zhang KY (2004) Structural basis for the activity of drugs that inhibit phosphodiesterases. Structure 12(12):2233–2247
Rathore PK, Arathy V, Attimarad VS, Kumar P, Roy S (2016) In-silico analysis of gymnemagenin from Gymnema sylvestre (Retz.) R.Br. with targets related to diabetes. J Theor Biol 391:95–101
Acknowledgements
HA thanks University Grants Commission (UGC), Government of India, for Rajiv Gandhi National Fellowship (F1-17.1/2012-13/RGNF-2012-13-SC-RAJ-29230) to pursue his PhD degree, and MSC thanks the Department of Biotechnology (DBT), Government of India for providing financial assistance (DBT’s Twining programme for North East-BT/246/NE/TBP/2011/77).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest in the present work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Arya, H., Syed, S.B., Singh, S.S. et al. In Silico Investigations of Chemical Constituents of Clerodendrum colebrookianum in the Anti-Hypertensive Drug Targets: ROCK, ACE, and PDE5. Interdiscip Sci Comput Life Sci 10, 792–804 (2018). https://doi.org/10.1007/s12539-017-0243-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-017-0243-6