Abstract
The Gruta da Aroeira (Torres Novas, Portugal), with evidence of human occupancy dating back ∼ 400,000 years, is one of very few Middle Pleistocene cave sites to provide a fossil hominin cranium in association with Acheulean bifaces and the by-products of fire usage. Zooarchaeological, taphonomic and tooth-wear analyses suggest that the accumulation of the faunal remains and their modification are anthropogenic. Large game constituted the basis of subsistence, with equids and cervids being preferentially targeted. Woodland and open landscapes formed the ecosystems supporting the populations of the mammals that were preyed upon by the inhabitants of the site. Most of the animal carcasses were carried to, and fully butchered at the site, which was used as a residential base camp. The features of the Aroeira faunal assemblage foreshadow the subsistence strategies developed by the hunter-gatherers of the Middle and the Upper Palaeolithic and testify to their very ancient roots.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Middle Pleistocene of Europe was a critical period in the evolution of modern humans, marked by the development and diffusion between 500 and 600 thousand years ago (ka) of two major innovations: namely, Acheulean technology and the controlled use of fire. The index fossil of the former is the biface or handaxe (Gowlett 1988), employed, among other tasks, for woodworking (Dominguez-Rodrigo et al. 2001; Solodenko et al. 2015) and butchery (Solodenko et al. 2015). The period’s human fossils are indicative of populations that presented significant morphological diversity and reveal the emergence of the diagnostic features of the Neanderthals (Manzi et al. 2011; Arsuaga et al. 2014; de Lumley 2015). European sites containing Acheulean lithics and evidence of the use of fire in association with well-dated human fossils are, however, scarce; but the Middle Pleistocene site of Gruta da Aroeira (Almonda karst system, Torres Novas, Portugal) provides fresh insights. To date, this cave context and its fossil human cranium (Aroeira-3), dated to ∼ 400 ka, are the earliest evidence of the anthropogenic use of fire in association with Acheulean bifaces in western Europe (Daura et al. 2017, 2018; Sanz et al. 2018, 2020).
Several sites indicate that hunting was the major meat-procurement strategy used by humans at this time. Animal species – primarily equids, bovids and cervids – were preferentially targeted from different biotopes, and selective prey choice by age and sex is evident (Rabinovich et al. 2008; Stiner et al. 2011). In western Europe, the subsistence strategies of human groups around MIS 12–11 remain largely elusive (Villa and Lenoir 2009); yet, the faunal remains from a few sites allow subsistence strategies to be reconstructed and aspects of the residents’ social behaviour to be inferred. For example, in the bone bed layer of the TD10.2 sub-unit at Gran Dolina (Atapuerca), large quantities of bison remains are indicative of ∼ 400 ka-old seasonal, communal events of mass hunting in which the cave served as a kill-butchering site (Rodríguez-Hidalgo et al. 2015, 2017). In open-air sites, the production of bone tools and the exploitation of carcasses to obtain marrow have been documented, for example, at Schöningen 13 II-4 (MIS 9) (Germany), in association with butchered bovids (aurochs and bison) (Stahlschmidt et al. 2015; van Kolfschoten et al. 2018), and at Boxgrove (United Kingdom), in association with the butchery of a large female horse (Roberts and Parfitt 1999; Smith 2013; Pope et al. 2020). Similarly, cut marks and the anthropogenic bone breakage of megafaunal remains – including rhinoceros at Caune de l’Arago (Chen and Moigne 2018) and elephant at Áridos 2 (Yravedra et al. 2010) and Ambrona (Villa et al. 2005) – have also been documented. However, it remains unclear whether this evidence is indicative of hunting or of the exploitation of the natural deaths of large animals. Evidence of small game procurement is rare but some – for example, the removal of meat from beaver remains observed at Caune de l’Arago (France) (Lebreton et al. 2017) – indicates that large mammals were not the sole meat resource.
When used as residential sites, Palaeolithic rock shelters and caves constituted the final destination for provisions and, as such, their archaeofaunal records furnish evidence for understanding carcass processing stages, consumption patterns, subsistence strategies and social behaviour. Here, in seeking to shed further light on the palaeoecology and adaptations of Middle Pleistocene humans, we present the faunal assemblage from layer X of Gruta da Aroeira. We undertake zooarchaeological, taphonomic and dental wear analyses of the large mammals from this stratigraphic unit in order to characterize the hunting and subsistence behaviour of the site’s occupants.
Background
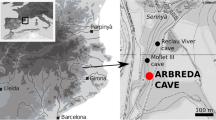
Gruta da Aroeira (henceforth, Aroeira) is a cave located in the Central Limestone Massif of Estremadura, in the west of the Iberian Peninsula (Fig. 1(1–2)). The site is a fossil outlet of the Almonda river (a tributary of the Tagus) (Fig. 1(4)) and lies ∼ 144 m above modern sea level. The cave forms part of the Almonda karst system, a labyrinthine network of passages featuring a number of former entrances with Pleistocene sedimentary infillings sealed by roof collapse, such as Gruta da Oliveira (Zilhão et al. 2021; Zilhão 2024).
The site was discovered in 1991 by the Sociedade Torrejana de Espeleologia e Arqueologia (STEA) (Zilhão et al. 1993) and was first excavated between 1997 and 2002, revealing a rich lithic assemblage of Acheulean bifaces together with faunal remains (Marks et al. 2002a, b) and two human teeth (Aroeira 1 and 2) (Trinkaus et al. 2003). Note that these early publications designated the site as “Galeria Pesada” (Marks et al. 2002b); however, this nomenclature corresponds to the interior conduits of the karst system while the name Gruta da Aroeira corresponds more properly to the cave entrance and the archaeological site (Hoffmann et al. 2013).
Archaeological excavations resumed in 2013 in a 6 m2 area at the back of the cave, with the aim of reaching bedrock and investigating the chronological range of the archaeological deposit, for which the speleothem samples collected and dated between 2006 and 2011 had provided a minimum age of ∼400 ka (Hoffmann et al. 2013). In this area of the site, the stratigraphic sequence spans a thickness of 4 m and comprises three major stratigraphic units. Uppermost Unit 1 is a brecciated infilling capped by flowstone dated to 44.8 ± 2.0 ka (2σ). Unit 2 is a 2.2 m-thick, mud-supported breccia, rich in angular and sub-rounded clasts, corresponding to Acheulean levels X and Xb/c (which are the upper and lower parts of a single unit, excavated 1997–2002 and 2013–2017, respectively) and capped by a second flowstone dated to 418 + 37/ − 27 ka (2σ) (Fig. 1(3)). Basal Unit 3 is an endokarst fluvial deposit comprising two layers: layer XI is a 0.4 m-thick silty sand with scattered gravel and faunal remains but no artefacts, and layer XII is a 0.5 m-thick, archaeologically sterile, slightly gravelly sand. The external layer of a stalagmite column buried by the subsequent accumulation of Units 1 and 2 and which, based on available cross-sectional views, seems to have grown from the top of layer XII, has been dated to 406 ± 30 ka (2σ). This result represents a maximum age for layer Xb/c, whose deposition can therefore be constrained to the 389–436 ka interval.
The lithic assemblage recovered from the latest excavation includes flakes (53%), cores (12%), bifaces (4%), debris (2%) and cobbles (25%) (Daura et al. 2018). The technological analysis of this material revealed no Levallois elements and suggests that on-site knapping consisted of the reduction of centripetal cores. The raw material provenience study suggests that the Acheulean inhabitants of Aroeira primarily frequented the lowlands surrounding the Tagus River and its tributaries. Accordingly, the rocks used in the main were locally available quartz and quartzite, while flint was obtained at sources > 10 km away.
Layer Xb/c also yielded a partial human cranium (Aroeira-3) comprising most of the right half of the calvarium and a fragmentary right maxilla (Daura et al. 2017). Aroeira-3 shows no signs of human modification or any alterations produced by carnivores, and an accident rather than inter-personal conflict or cannibalism is the most plausible explanation for a perimortem-like fracture in the posterior region of the parietal bone (Fig. 2(1–2)) (Sanz et al. 2018). Additionally, the use of fire in the cave is evident owing to the presence of the by-products of combustion, including burnt bones (Fig. 2(3–6)), charcoal and heat-altered cobbles and sediments, the latter allowing the identification of at least two different horizons of fire usage (Sanz et al. 2020).
Materials and methods
Excavation methodology
The archaeofaunal remains analysed here were recovered during fieldwork undertaken between 2013 and 2017. The remains come from the area at the back of the cave where the lower part of Unit 2 (layer Xb/c, henceforth, layer X) was excavated beyond the elevation reached in 2002: grid units H/I/J-6/7/8. In this area of the site, Unit 2 is fully sealed by the flowstone and is, therefore, securely dated. As the archaeological materials were encased in a hard carbonate breccia formed by the precipitation of percolating water, the excavation had to be carried out with demolition hammers. We endeavoured to collect all the remains, but sieving was not possible. Thus, a bias can be expected in terms of the preservation of very small pieces of bone, such as cone flakes; fragile fragments, such as avian bones; and small taxa, such as leporids. Faunal bones were piece-plotted in situ prior to extraction, while small fragments were bagged by grid units of provenience. The breccia blocks extracted from the site with the power tools were broken into small pieces outside the cave. All faunal remains were detached using a pneumatic microhammer and microchisel (Mod. CTS 178) and restored using a normal air scribe (Mod. W 224). Where necessary, bones were consolidated with an acrylic resin (Paraloïd-B72) dissolved with acetone.
Zooarchaeological and taphonomic analyses
Bones were analysed following standard zooarchaeological methods (Stiner 2005; Reitz and Wing 2008). These included identification of the anatomical region (cranial, axial and appendicular), the skeletal element, the portion of the element, side, and age (foetus/neonate, infantile, juvenile, adult or senile). Dental development and degree of tooth wear were used as ageing criteria, with specimens being assigned to one of five age groups: foetal/neonatal, infantile (milk teeth in use), juvenile (deciduous dentition undergoing replacement), adult (permanent teeth) and senile (advanced occlusal tooth wear). The scarce representation of epiphyses prevented us from analysing the degree of fusing as an indicator of age.
The high degree of fragmentation complicated skeletal identification. All non-identifiable fragments were assigned to three categories: flat, long, or spongy bones. The presence of long bone landmarks (e.g. foramens and grooves) facilitated the recognition of small fragments with these unique features (e.g., deer metapodials), but most other fragments were less readily identifiable.
The bones were identified to genus or species level whenever possible, while the large number of unidentified remains were assigned to body size-weight categories following the modified criteria proposed by Bunn (1986): small, < 100 kg (roe deer and ibex); medium, 100–300 kg (red deer); large, 300–1,000 kg (large bovids and horses); and very large, > 1,000 kg (rhinoceros and proboscideans). In the present study, we grouped the different species in broad categories. For example, in line with previous studies (see, for example, Croitor et al. 2019), the three cervids could be individually identified (as Dama, Haploidoceros and Cervus); but, here, unless otherwise justified, we opted to group them together as deer to facilitate interpretation.
The number of remains (NR), the number of identified specimens (NISP), the minimum number of individuals (MNI), the minimum number of elements (MNE) and the skeletal survival rate (% Surv.) were calculated following Brain (1981), Poplin (1981), and Lyman (2008). The following formula was used for the latter: % Surv. of element (i) = (MNEi * 100) / (MNI * number of element(i) in the skeleton). Bone loss from density-mediated attrition was evaluated using the computed tomography (CT)-derived values of bone mineral density (BMD1) for reindeer (Rangifer tarandus) and horse (Equus sp.) as a standard (Lam et al. 1999), and Spearman’s rho test. Taphonomic observations recorded surface modifications in line with Lyman (1994), and Fernández-Jalvo and Andrews (2016).
To determine the effects of post-depositional and biostratinomical (whether carnivore- or hominin-related) processes, breakage type and surface modification were examined at the macroscopic level under a binocular device (MOTIC SFC-121 GG). Gnawing damage signals carnivore modification (Brain 1981; Selvaggio and Wilder 2001), while percussion and cut marks signal anthropogenic modification, namely butchery (Shipman and Rose 1983; Johnson 1985; Blumenschine and Selvaggio 1988). Following Shipman and Rose (1983), cut marks are V-shaped in cross section and may feature shoulder effects, while their orientation, anatomical placement, and type (incisions, sawing marks, and scrape marks) help in distinguishing them from carnivore-inflicted damage and in assessing the butchery function involved (disarticulation, skinning, filleting, viscera removal or defleshing, among others) (Binford 1981; Lyman 1987). Cut marks were analysed at the Centres Científics i Tecnològics of the Universitat de Barcelona using a Quanta 200 scanning electron microscope (SEM).
Breakage patterns were described following the criteria proposed by Villa and Mahieu (1991), namely, fracture angle, outline, and edge. The fracture angle is that between the fracture and the cortical surfaces: right (∼ 90º), oblique (obtuse or acute angles) or mixed (variable angles). The outline is the orientation of the fracture relative to the bone’s long axis: transverse (perpendicular), curved (spiral or V-shapes fracture) or intermediate (multidirectional morphology). The edge criterion refers to the condition of the edge’s surface: smooth (flat surface) or jagged (rough surface). Following Bunn (1983) and Villa and Mahieu (1991), the portion of the bone preserved was described as less than half (of the shaft circumference or of the shaft longitude), more than half, or complete.
Burning damage was described in a previous work, in which the different degrees of burning are described and discussed (Sanz et al. 2020); here, we treat burnt bones as an undifferentiated ensemble. Bone abrasion, including rounding or polishing, may result from biological activity (e.g., use as tools by humans, carnivore activity), or from transport and trampling (Lyman 1994; Fernández-Jalvo and Andrews 2016), and was assessed according to Behrensmeyer’s stages (1978). Abrasion was also categorized according to the degree and location of the rounding: slight (restricted to fractures or edges and observed microscopically), moderate (restricted to some areas and observed macroscopically) and strong (all over with possible loss of original bone morphology), following the criteria proposed by Cáceres (2002) and Fernández-Jalvo and Andrews (2016).
Tooth microwear analysis
Tooth microwear was analysed following the protocol established by Solounias and Semprebon (2002). Tooth occlusal surfaces were first cleaned with acetone and then ethanol 96% to remove consolidants and eliminate any sediment or dust. The occlusal surfaces were moulded using high-resolution dental silicone (Heraeus Kulzer, PROVIL novo Vinylpolysiloxane, Light C.D. 2 regular set) and casts were created using clear epoxy resin (C.T.S. Spain, EPO 150 + K151). All teeth were carefully screened under a stereomicroscope to check and discard those presenting taphonomic alterations (King et al. 1999; Uzunidis et al. 2021). Casts were observed with a Zeiss Stemi 2000 C stereomicroscope at 35× magnification, using the refractive properties of the transparent cast to observe the microfeatures on the enamel. To avoid inter-observer error, the analysis was performed by a single observer (FR).
Microwear features were quantified in two areas using an ocular reticule of 0.16 mm2 on the paracone of the upper molars and the protoconid of the lower molars. We used the classification of features as defined by Solounias and Semprebon (Solounias and Semprebon 2002; Semprebon et al. 2004), which specifically identifies pits and scratches. The presence/absence of other features – cross scratches, hyper-coarse scratches, gouges and large pits – was recorded qualitatively. In addition, scratch textures were assessed using the scratch width score (SWS), which is obtained by giving a score of ‘0’ to a tooth with predominantly fine scratches, ‘1’ to a tooth presenting a mixture of fine and coarse texture types, and ‘2’ to a tooth with predominantly coarse scratches. Individual scores for a sample of teeth were then averaged to obtain the SWS.
Results
Preservation
The taphonomic analysis of the Aroeira-3 human teeth and cranium has been reported elsewhere (Sanz et al. 2018). Table 1 summarizes the main alterations suffered by the faunal assemblage. A total of 22 remains (7%) present initial stages of weathering with bone cracking, while medium and advanced stages are totally absent, pointing to the rapid burial of remains with a low degree of exposure. Thermal alteration is observed on at least 44 bone remains. A brownish patina – from slight to strong – is also recorded (N = 58); yet, few (N = 4) present the yellowish appearance broadly observed in the layer XI bones. Manganese coatings are abundant (28%), but most are slight (24%). Other geological alterations – abrasion (0.7%), desquamation (2%), dissolution (4.3%), sediment pressure (1.3%) and trampling (1.3%) – are scarce. Abrasion is especially infrequent, being responsible for the rounding of just two bones. These bones were recovered close to a stalagmite column, suggesting abrasion may be related to the percolation of water along the interstitial spaces found within the sedimentary column or between the latter and the cave walls. No post-depositional marks attributable to rodents were found.
The impact of human activity is visible on 14.7% of the remains. No evidence of carnivore interference (e.g., pits, furrowing or ravaging) was found in the bone assemblage. No partially digested bones were identified either.
Taxonomic identification, age and body part representation
Cervids are the most abundant category (22%NISP) (Fig. 3(5, 8)), so, unsurprisingly, the non-identifiable bones assigned to medium-sized animals are also the most abundant (36%) (Table 2). Cervids are followed by equids (14%NISP), including two remains of a small size taxon. Other taxa – rhinoceros (1.5%NISP) (Fig. 3(9–10)), bovid, macaque and suid (Fig. 3(4)) (< 1%NISP) – are less frequent. Fragmentation of the remains hinders species assignment; however, some palaeontological features suggest that the rhinoceros remains correspond to Stephanorhinus etruscus.
Carnivores are scarce (2.4%NISP) (Table 2). The most abundant is the bear, represented by six remains, among which the palaeontological features of Ursus deningeri could be identified (Fig. 3(12)). The lynx is represented by one dental remain. Other taxa, namely beaver and tortoise, are also represented (0.9%NISP) (Fig. 3(1)).
The majority of the assemblage is composed of non-identifiable bones (58%). According to body size and in descending NR order, they can be classified as follows: medium (36%), large (16.8%), small (4%) and very large (0.9%). A fragment of a spinous process of a cervical vertebra may belong to a proboscidean. However, it is too fragmentary to be diagnostic. Until additional evidence is obtained, this identification should be considered tentative and the potential presence of the taxon at the site should be treated with caution (See Table 2).
The age at death profile for the individuals recovered in layer X shows a marked predominance of adults in terms of the MNI (equids = 3, cervids = 5, carnivores = 1 for each taxa), with just one juvenile (an equid). Cranial elements constitute the most frequent skeletal part (22%) for all species and size categories (small, medium, and large). Isolated teeth are the most abundant (10%), followed by cervid antlers (7%; one corresponding to a shed antler), maxilla (2%) and mandible (2%) fragments and, finally, fragments of the cranium (1%). Axial skeleton remains – ribs (4%) and vertebrae (6%) – are abundant. Among the remains of the stylopodium, the humerus (6%) is the most numerous, while the femur (2%) is poorly represented. Among the remains of the zeugopodium, the posterior extremity predominates, with the tibia being the most frequent (6%); the anterior extremity is represented by the radius (2%). Metapodium (6%), acropodium (7%) and basipodium (3%) remains are also represented. Scapular and pelvic girdle bones are scarce (1%). Most of the bones are non-identifiable fragments assigned to the long (19%), flat (5%) and spongy (6%) bone categories.
Survival rates were calculated for the two main body size categories identified in layer X: that is, medium and large ungulates. The results of a bivariate test of bone density versus survival rate are not statistically significant for either the medium (Spearman’s r = − 0.037, p = 0.890) or the large categories (Spearman’s r = − 0.096, p = 0.713). Thus, the bias observed in the body part representation pattern is unlikely to derive from differential preservation processes and is more likely to reflect hominin agency.
Carcass butchery
The bone assemblage from layer X bears anthropogenic modification marks caused by stone-tool cutting, breakage, percussion and burning (Table 3). Among the ungulate bones, incisions are seen on thirty-three, impact flakes on five, percussion marks on three, and scrape marks on eight. Bones with cut marks are present throughout, the highest frequency coinciding with the greater concentrations of stone tools (two main horizons can be recognized; Fig. 4(3)).
Butchery patterns can be inferred from the abundance of large and medium-sized ungulates. Skinning is observed on the diaphysis of metapodials, skinning and/or disarticulation of joints is observed on an equid phalanx (Fig. 5(2)) and a sesamoid, and there is other evidence of disarticulation. The latter includes a cut mark on the glenoid cavity of a deer scapula that may correspond to the disarticulation of the humerus, while cut marks on the distal epiphysis of a humerus, on the proximal epiphysis of a femur, and on the tibial crest of deer tibiae would also seem to correspond to disarticulation. Scrape marks associated with the removal of the periosteum and meat, indicative of defleshing (filleting), are present on limb bones and on a rib. The bulk of the incisions are located on shaft portions, which is also indicative of the removal of meat. Evisceration can be inferred from cut marks on some rib bones (Fig. 5(4)). Incisions on the mandibles of two medium-sized ungulates (Fig. 5(5)) suggest the cutting and peeling away of the cheek and tongue. Percussion marks are also present on three of the remains; two of them (a shed antler and a humerus shaft) could have served as knapping tools or ‘retouchers’. These remains, together with three bones presenting abrasion-smoothed ends, are currently under study to determine their possible use as tools.
Fracture morphologies – with a predominance of oblique angles (92%), curved outlines (94%), and smooth fracture surfaces (98%) – are consistent with the breakage of fresh bone (Fig. 6). These breaks are likely to be anthropogenic and made to extract marrow. No cone flakes are present; however, this might reflect the constraints on recovery posed by the hard breccia in which the finds were encased. Indeed, 90% of the shafts analysed preserve less than half their original circumference and 89% less than half their bone length (vs. 6 and 11%, respectively, that preserve more than half), while the number of complete sections (4%) is trivial and full-length bones are totally absent. In combination with the morphology of the fractures, these attributes are unambiguously suggestive of an anthropogenic assemblage cracked open and thoroughly processed for the extraction of within-bone nutrients.
Finally, as mentioned above, 13% of the bones from layer X present signs of heating. Different degrees of combustion are observed: partially burned (1%), carbonized (8%) and calcined (4%) (Sanz et al. 2020).
Dental microwear analysis
The ungulate teeth display a well-preserved microwear pattern: of the twenty-one teeth initially sampled, only three had to be discarded due to taphonomic damage to the occlusal surface.
The microwear pattern of the equids (Equus sp.) is characterized by a large number of scratches, few pits and no gouges; however, large pits are present on roughly half of the specimens (Table 4). The high SWS indicates that dental scratches are predominantly coarse; additionally, hypercoarse scratches are observed on roughly half of the specimens. A comparison with extant ungulates indicates that the Aroeira equids belong to the grazer trophic phase (Fig. 7).
The microwear pattern of the Praedama cf. savini teeth is characterized by a small number of both pits and scratches (Table 4); however, large pits and gouges are numerous, coarse scratches predominate, and there are a few hypercoarse scratches. As such, these teeth compare well to those of extant leaf browsers (Fig. 7); moreover, these characteristics suggest that, as well as the leaves of trees or bushes, Praedama cf. savini also ingested significant amounts of twigs.
The teeth of Cervus elaphus present few scratches and a large number of pits. Moreover, they are characterized by numerous large pits and gouges, and by both coarse and hypercoarse scratches. A comparison with extant ungulates indicates that the Aroeira red deer plot among the leaf browsers (Fig. 7). In common with Praedama cf. savini, they probably fed on the twigs and leaves of trees and bushes, but the number of pits suggests that grit and dust were also ingested. It seems certain, therefore, that these two deer taxa fed in different areas or off different plant parts – ones that, in the case of the Cervus niche, contained leaves that accumulated considerable amounts of grit and dust.
The dental microwear pattern of the Haploidoceros mediterraneus, in common with that of the Praedama cf. savini, presents few pits and scratches. However, while no hypercoarse scratches and gouges are present, a considerable number of fine scratches are detected. The sample from Aroeira plots among extant leaf browsers (Fig. 7), but unlike the other two Aroeira cervids, Mediterranean deer seem to have fed exclusively on the leaves of trees or bushes.
In short, the dental wear analyses carried out on four ungulate species (cervids and equids) from layer X of Aroeira are indicative of different diets. The cervids (Praedama cf. savini, Cervus elaphus and Haploidoceros mediterraneus) appear to have been browsers whereas the horse was a grazer. Of particular relevance is the lack of overlap in the microwear patterns of the four taxa, which is indicative of inter-species niche partitioning, in particular as regards the three cervids.
Discussion
Taphonomy
Although the preparation of the remains was sufficient to guarantee the success of the zooarchaeological, taphonomic and dental microwear analyses, the smaller, more fragile remains are most certainly underrepresented. This, as discussed, can be attributed to the assemblage’s brecciated matrix and the recovery techniques resorted to in having to overcome this constraint. Any evidence of damage attributable to weathering is negligible, which is indicative of minimal exposure prior to burial and the protection afforded by the cave setting and/or the rapid burial of the remains. There is little evidence of post-depositional movement – dry fractures of bones resulting from sediment pressure are present in just 2% of the assemblage – and of trampling – present in just 4% of the assemblage.
The karst depositional environment may also be responsible for the infrequent presence of damage attributable to abrasions (≤ 1%), desquamations (2%), and dissolution (4%). Additionally, the manganese coating giving some of the bones a distinctive black colour (25%) reflects the nature of the burial environment. Very few bones present the extensive, homogeneous yellow coating (1%) that is characteristic of the remains from underlying layer XI and which has been related to post-burial oxidation caused by percolating water, as observed in other karst contexts (Zunino et al. 2012). The brownish colouration of a number of remains (17%) needs further examination, but might derive from fire damage or other characteristics of the depositional environment (Fernández-Jalvo and Andrews 2016; Pollock et al. 2018; Sanz et al. 2020). Several unambiguous by-products of burning (including bone, charcoal and, possibly, cobbles) have, however, been recorded. In short, the faunal assemblage from layer X of Aroeira is characterized by the scarce representation of alterations of a biostratinomic, post-burial, or post-depositional nature.
Evaluation of the state of preservation of the assemblage is critical in ascertaining the stratigraphic integrity of the artefact assemblages and, hence, in making inferences about hominin behaviour. The taphonomy of the faunal remains analysed here strongly supports the homogeneity of the layer X deposit, in agreement with the geochronological data and the results of the studies previously conducted on the lithics and the paleoanthropological remains. Yet, owing to certain idiosyncrasies of the lithic assemblage – namely, the absence of cleavers and other large tools characteristic of the Acheulean – some authors have suggested that the handaxes recovered in layer X might be intrusive (Méndez-Quintas et al. 2018; Santonja and Pérez-González 2021; Santonja et al. 2022). Flake cleavers and bifaces are commonly found in fluvial terraces and at other open-air sites where hominin activities seem to have differed from those carried out in residential camps such as Aroeira. Thus, the lack of cleavers in the latter’s lithic assemblage may have a functional explanation. Whatever the case, the evidence unambiguously rejects the hypothesis that the specific features of the Aroeira assemblage reflect post-depositional disturbance processes.
Biological agents
The anthropogenic marks and breakage patterns observed on the bones recovered from layer X indicate that human groups were the primary agent involved in the accumulation and modification of the assemblage. Although two carnivore species (bear and lynx) are represented in the layer, their few remains – primarily teeth – show no evidence of modification. And while their presence might be related to human activities, it should be noted that both species are cave dwellers. The presence of lynx in caves and their use of the latter as dens have been widely documented (see Brugal et al. 2012; Rodríguez-Hidalgo et al. 2020; Gabucio et al. 2024), while, among bears, reiterative use of caves both for hibernation and birthing has likewise been broadly documented in the Pleistocene record (Stiner 1998; Marks et al. 2002a; Sanz and Daura 2018; Grandal-d’Anglade et al. 2019). Indeed, bears have been documented at other sites in the Almonda karst system, namely in the Middle Palaeolithic Gruta da Oliveira, in association with hibernation deaths (Zilhão et al. 2010). Yet, the idea that the carnivores recovered in layer X of Aroeira might in some way be associated with the economic activities of Palaeolithic hunter-gatherers cannot a priori be rejected. Indeed, while the predation of carnivores is rarely documented in the Middle Pleistocene, there is some evidence in the archaeological record, including at TD10.1 (Atapuerca), where a lion was probably hunted and processed for food (Blasco et al. 2010), and at Schöningen, where a large cat bone was employed as a knapping tool (Van Kolfschoten et al. 2015).
Prior to human occupation, Aroeira was frequented by carnivores as evidenced by two separate horizons: (i) basal Unit 3 (layers XI and XII), which presents no lithics or anthropogenic faunal remains, contains beaver bones related to the natural death of animals whose habitat extended to the underground stretches of the river, upstream from the then-extant spring (Cuenca‑Bescós et al. 2021); and (ii) the boundary between layers X and XI, characterized by an accumulation of coprolites. The evidence from layer X allows us to rule out the possibility that carnivores had a role in the formation of the faunal assemblage as either bone modifiers or bone accumulators. Assessing the likelihood of other explanations, for example, the sporadic use of the cave by carnivores as a refuge, is difficult in the absence of direct indicators of activity (Villa et al. 2004). It is our conjecture, therefore, that humans inhabited the cave with little or no overlap with carnivores, the latter’s absence probably being a direct result of the intensity and frequency of the presence of human groups.
Due to the limited number of remains recovered (the result, as discussed, of the hard breccia encasing and of the fragility of the bones), we are unable to ascertain the origin of the Lagomorpha remains and those of other small prey, or more specifically whether they might be associated with the activity of the lynx. However, the microfaunal remains from an area near the collapsed cave entrance excavated between 1997 and 2002 implicate the golden eagle or the Eurasian eagle-owl in the accumulation of rabbit remains and, as such, rule out the involvement of humans or carnivores, including that of the lynx (Marks et al. 2002a; Pelletier 2018).
Subsistence strategies
Based on the anthropogenic bone modifications observed, cervids and equids were the main prey of the Aroeira humans. Indeed, in all likelihood, most of the non-identifiable fragments assigned to the medium-sized and large categories also belong to these taxa. At the site, the entire carcass processing sequence – skinning, removal of the viscera, disarticulation, defleshing and filleting, and, finally, marrow extraction – can be identified. This pattern is consistent with the introduction and on-site processing of complete carcasses (at least, in the case of cervids) and of their on-site consumption too. These butchery practices sought not only the reduction of the carcasses to portable, shareable portions, but also the exploitation of grease, bone marrow, meat, and hides. Moreover, the anthropogenic marks and breakage patterns indicate regular hunts, intensive exploitation of carcasses, and primary access to prey. The other taxa recovered – including suid, bovid and rhino – are scarce in number and neither cut marks nor carnivore damage have been identified on their bones. However, the parsimonious explanation is that they too were exploited by human groups, as documented in other Lower Palaeolithic assemblages (Moncel et al. 2012; Rodríguez-Hidalgo et al. 2016; Chen and Moigne 2018). However, it is not possible to propose an origin for the macaque mandible found at the cave. Although several European Lower Palaeolithic sites provide evidence of the coexistence of macaques and humans (Elton and O’Regan 2014; Konidaris et al. 2022), direct evidence of their exploitation by humans remains elusive (but see the reports from Cova Bolomor) (Blasco and Fernández Peris 2012).
The high frequency of cut marks on the bones recovered from layer X is notable (∼ 10%) and in close agreement with the frequencies documented in other layers of the cave (8–16%) (Marks et al. 2002a). Similar frequencies have been documented at other Lower Palaeolithic sites, including both Gran Dolina (Atapuerca) (Rodríguez-Hidalgo et al. 2015) and Qesem Cave (Stiner et al. 2009). Traditionally, archaeofaunal interpretations emphasize the total frequencies of cut-marked specimens within a given assemblage at the expense of other qualitative variables. Yet, cut mark frequencies can vary widely, due to the broad spectrum of variables determining whether bones will be modified in this way (Lyman 1987), and as such this indicator is of uncertain value. Other relevant variables impacting cut-mark frequency include skeletal element representation, time of day, number of butchers, biological agents involved, carcass size, and fragmentation (Lyman 2005; Dominguez-Rodrigo and Yravedra 2009). What is critical here is whether these variables can be archaeologically measured and their behavioural significance ascertained. Variables such as the degree of expertise of butchers, carcass processing and site functionality are difficult to control and cannot be measured from the assemblages. In contrast, the percentage of cut-marked long bones, the size of the prey, and the presence of tools play a crucial function in interpreting cut-mark frequencies (Lupo and O’Connell 2002; Egeland 2003; Dominguez-Rodrigo and Yravedra 2009).
The number of cut marks in relation to tool type and butchering activities has been addressed in experimental studies. Their findings indicate that handaxes produce a higher number of marks in disarticulation and skinning processes, while retouched flakes are largely responsible for the marks made when defleshing (de Juana et al. 2010; Galán and Domínguez-Rodrigo 2013). However, a direct correlation cannot be drawn between tool types and cut-mark percentages because of the other factors that might impact the patterns. For example, in Qesem Cave, the presence and distinctive features of cut-marked bones apparently correlate not to tool types, but rather to variations in the social modes of food consumption and cooperation that occur within a co-resident group (Stiner et al. 2009). Thus, in butchery, multiple hands might be involved, including the less experienced, as opposed to the skilled butchering efforts of a single or just a few individuals.
In the case of Aroeira, defleshing would seem to be the most frequent activity and this, in turn, may well be related to the significant representation of flakes (∼ 50%), including the flakes used as blanks for tools present among the 2013–2017 layer X lithics. Skinning is less readily identifiable, in potential agreement with the somewhat scarce representation of bifacial tools (∼ 5%) in these same lithics (Daura et al. 2018). The high frequency of butchery marks indicates that the cave served as a base camp and reflects the intensive exploitation of the carcasses, an activity that may have taken place in dedicated areas. However, the limited size of the 2013–2017 trench does not allow us to assess intra-site distribution patterns.
The presence of human remains in layer X does not seem to be related to the subsistence practices observed at the site. As discussed, the absence of anthropogenic marks points to either an accidental or a natural, disease-related death event (Sanz et al. 2018). Similarly, there is no evidence that the cranium served a symbolic purpose or that it underwent some kind of processing (Saladié et al. 2012).
Paleoenvironmental reconstruction
The Aroeira faunal record is indicative of open woodland and a relatively mild, semi-humid climate. Based on the chronological constraints provided by U-Th dating, layer X can be dated to the MIS 11 interglacial, which presented a higher mean annual temperature (+ 2.7 to + 0.3 °C) and a higher mean annual precipitation (+ 311.7 to + 74.4 mm) than today and, in the case of Iberia, a significant expanse of woodland (Blain et al. 2015; López-García et al. 2018). However, the earlier limit of the interval indicated by U-Th dating also includes the last twelve millennia of the MIS 12 glacial phase (i.e., 424–436 ka). As discussed elsewhere (Zilhão 2023), a late MIS 12 chronology is more consistent with the clast-supported nature of the deposit, in which the fine elements represent the infilling of the interstitial spaces of an open-work accumulation, and with the palaeoclimatic implications of the microfaunal assemblage, which includes the yellow-necked mouse (Apodemus flavicollis), a species never recorded in Portugal and today restricted to northern Iberia, above 41ºN (Torre et al. 2009).
The most abundant prey were the cervids that fed off the surrounding woodlands, followed in abundance by equids, which grazed in an otherwise open landscape. Mediterranean tortoise and other small mammals were also harvested, given the existence of a semi-humid environment and as indicated by the composition of the rodent assemblage (López-García et al. 2018). Finally, the presence of the macaque (Alba et al. 2019) and other, less abundant taxa, including wild boar, point to notable areas of woodland in the immediate vicinity of the cave.
Based on dental microwear analyses, the deer of Aroeira were essentially browsers; however, different browsing diets can be observed within this rich cervid community. Mediterranean deer (Haploidoceros mediterraneus) fed exclusively on the leaves of trees or bushes, and present a microwear pattern similar to that observed for the taxon at Cova del Rinoceront, near Barcelona (Rivals et al. 2016). For the cervids as a whole, the dental wear patterns suggest sympatry with niche partitioning, which allowed the three taxa to browse alongside each other in the same woodland environments. Fallow deer (Dama cf. vallonnetensis) are also present at Aroeira, but are represented by just two antlers, one of these being the proximal part of a shed antler that may have been brought to the site by humans for technological purposes. Given the evidence from Cova del Rinoceront (Sanz et al. 2014; Rivals et al. 2016; Croitor et al. 2020), it is likely that fallow deer would have competed with Mediterranean deer (H. mediterraneus) for the same food resources.
Based on the strontium isotope evidence collected from the Middle and Upper Palaeolithic sites of the Almonda karst, the fauna analysed (ibex, red deer, horse and rhinoceros) would have inhabited areas within a short distance of the sites, albeit perhaps on a seasonal basis in the case of ibex, though not in that of cervids and equids (Linscott et al. 2023). The situation cannot have been much different in the Lower Palaeolithic, when deer and horses were also the primary animals targeted by the human dwellers of the caves associated with the spring of the Almonda river. Thus, animal procurement would, in the main, have been limited to the local environment, i.e., the low elevation areas at the base of the Almonda escarpment and, further north, the higher reaches of the Serra d’Aire, and, to the south, the plains of the Tagus basin downstream of the spring.
Conclusions
The faunal assemblage from layer X at the Gruta da Aroeira derives primarily from anthropogenic activities conducted during the Lower Palaeolithic. The subsistence of these human groups was based essentially on large game, as evidenced by the assemblage’s adult-dominated mortality profile and anthropogenic modifications, which are similar to those observed at Middle and Upper Palaeolithic sites. Indeed, the predominance of medium-sized deer and equids suggests that the inhabitants selected their prey and transported the carcasses whole to the site for subsequent processing, in what was a highly standardized exploitation strategy targeting the most valuable parts of the animals. These mammal populations appear to have been sustained by woodland and open landscape ecosystems. The limited occurrence of large animal remains – including rhinoceros and, possibly, a proboscidean – indicates that only the most nutritious parts of these heavier prey would have been transported back to the cave. Thus, the layer X occupation may be interpreted as a residential camp in which a wide range of activities would have been conducted; among them, the complete butchering of ungulate carcasses. This conclusion is supported by various lines of evidence: including, the use of fire; the abundant, cervid- and equid-dominated animal bone accumulation; the scarcity of the remains of carnivores; the latter’s almost total lack of involvement in the accumulation or modification of the ungulate remains; and the high frequency of anthropogenic modifications seen on the bones. In short, the features of the Aroeira faunal assemblage – butchery patterns, deliberate prey selection, transport decisions, mortality profiles, predominance of large game – foreshadow the subsistence strategies developed by the hunter-gatherer groups of the Middle and the Upper Palaeolithic and bear witness to the latter’s very ancient roots.
The Gruta da Aroeria cave site. 1–2. Map showing the location of the Gruta da Aroeira (red star) in the Iberian Peninsula (map extracted from OpenStreetMap). 3. North stratigraphic profile of the 2013–2017 trench. 4. The Almonda escarpment; the position of the site is indicated (photo: Pedro Souto). 5. Panoramic view of the valley located above the escarpment
(1) Endocranial view of the Aroeira-3 human cranium indicating the perimortem fracture (white box) on the posterior region of the parietal bone. (2) Detailed view of the cortical delamination. 3–6. Burnt bones: fully calcined rib (3), carbonized distal condyle of a deer metapodial (4), fully carbonized fragmented epiphysis (5) and carbonized tortoise bone plate (6)
Faunal remains from Aroeira. (1) Tortoise pygal scute (ARO-798). (2) Horse navicular bone (ARO-304). (3) Horse lower tooth (ARO-53). (4) Suid canine (ARO-221). (5) Red deer antler tine fragment (ARO-102). (6) Horse metapodial distal condyle (ARO-756). (7) Equid cuboid (ARO-761). (8) Haploidoceros mediterraneus left maxilla (ARO-308). (9) Rhinoceros femur, proximal epiphysis (ARO-854). (10) Rhinoceros, third metatarsal (ARO-597). 11. Lynx, upper canine (ARO-764). 12. Bear, lower p4 (ARO-571)
Anthropogenic bone modifications. (1) Humerus shaft showing impact notches and filleting marks on the cortical surface (ARO-274). 1a and 1b. Detailed SEM views of cut marks. The longitudinally oriented marks intersect with some of the marks that are oblique to the bone’s main axis. (2) Equid phalanx showing cut marks associated with skinning (ARO-66), and detailed SEM view (2a). (3) Femur with cut marks on the shaft (ARO-853). (4) Rib with several incisions on the body (ARO-895). (5) Mandible with cut marks (ARO-462) with detailed SEM view
Breakage patterns present in layer X bones (Aroeira) compared to breakage patterns of green (Fontbrégoua) and dry bones (Sarrians and Besouze) following Villa and Mahieu (1991) so as to differentiate between peri- (green) and postmortem (dry) fractures
Bivariate plot of the average numbers of pits and scratches presented by the ungulates in layer X (Aroeira). Error bars correspond to the mean standard error (± 1 SEM) for each sample. Plain ellipses correspond to the Gaussian confidence ellipses (p = 0.95) on the centroid for the extant leaf browsers (LB) and grazers (G), based on the Solounias and Semprebon (2002) reference database
Data availability
No datasets were generated or analysed during the current study.
References
Alba DM, Daura J, Sanz M, Santos E, Yagüe AS, Delson E, Zilhão J (2019) New macaque remains from the middle pleistocene of Gruta da Aroeira (Almonda karst system, Portugal). J Hum Evol 131:40–47. https://doi.org/10.1016/j.jhevol.2019.02.003
Arsuaga JL, Martinez I, Arnold LJ et al (2014) Neandertal roots: cranial and chronological evidence from Sima De Los Huesos. Sci (80-) 344:1358–1363. https://doi.org/10.1126/science.1253958
Behrensmeyer AK (1978) Taphonomic and ecologic information from bone weathering. Paleobiology 4:150–162
Binford LR (1981) Bones: ancient men and modern myths. New York
Blain H-A, Lozano-Fernández I, Ollé A et al (2015) The continental record of marine isotope stage 11 (Middle Pleistocene) on the Iberian peninsula characterized by herpetofaunal assemblages. J Quat Sci 30:667–678. https://doi.org/10.1002/jqs.2799
Blasco R, Fernández Peris J (2012) A uniquely broad spectrum diet during the middle Pleistocene at Bolomor cave (Valencia, Spain). Quat Int 252:16–31. https://doi.org/10.1016/j.quaint.2011.03.019
Blasco R, Rosell J, Arsuaga JL, Bermúdez de Castro JM, Carbonell E (2010) The hunted hunter: the capture of a lion (Panthera leo fossilis) at the Gran Dolina site, Sierra De Atapuerca, Spain. J Archaeol Sci 37:2051–2060
Blumenschine RJ, Selvaggio MM (1988) Percussion marks on bone surfaces as a new diagnostic of hominid behaviour. Nature 333:763–765
Brain CK (1981) The hunters or the hunted? An introduction to African cave taphonomy. University of Chicago Press, Chicago
Brugal J-P, Argant J, Crispim JA et al (2012) The Complex Carnivore-rich assemblages from Furninha (Peniche, Portugal): a multidisciplinary approach. J Taphon 10:417–438
Bunn HT (1983) Comparative analysis of modern bone assemblages from a San hunter-gatherer camp in the Kalahari Desert, Botswana, and from a spotted hyena den near Nairobi, Kenya. Anim Archaeol 1:143–148
Bunn HT (1986) Patterns of skeletal representation and hominid subsistence activities at Olduvai Gorge, Tanzania, and Koobi Fora, Kenya. J Hum Evol 15:673–690. https://doi.org/10.1016/S0047-2484(86)80004-5
Cáceres I (2002) Tafonomía de yacimientos antrópicos en karst. Complejo Galería (Sierra de Atapuerca, Burgos), Vanguard Cave (Gibraltar) y Abric Romaní (Capellades, Barcelona). Ph.D.Thesis, Universitat Rovira Virgili
Chen X, Moigne A (2018) Rhinoceros (Stephanorhinus hemitoechus) exploitation in level F at the Caune de l’Arago (Tautavel, Pyrénées-Orientales, France) during MIS 12. Int J Osteoarchaeol 28:669–680. https://doi.org/10.1002/oa.2682
Croitor R, Sanz M, Daura J (2019) Deer remains from the middle pleistocene site of Gruta da Aroeira (Portugal): Iberian faunal endemism and implications for hominin paleobiogeography. Quat Sci Rev 225. https://doi.org/10.1016/j.quascirev.2019.106022
Croitor R, Sanz M, Daura J (2020) The endemic deer Haploidoceros mediterraneus (Bonifay) (Cervidae, Mammalia) from the late pleistocene of Cova Del Rinoceront (Iberian Peninsula): origin, ecomorphology, and paleobiology. Hist Biol 32:409–427. https://doi.org/10.1080/08912963.2018.1499018
Cuenca–Bescós G, Sanz M, Daura J, Zilhão J (2021) The fossils of Castor fiber from the middle pleistocene site of Gruta da Aroeira (Portugal) and human-beaver interaction. Quaternaire 32:01–10. https://doi.org/10.4000/quaternaire.14975
Daura J, Sanz M, Arsuaga JL, Zilhão J (2017) New middle pleistocene hominin cranium from Gruta da Aroeira (Portugal). Proc Natl Acad Sci 114:3397–3402. https://doi.org/10.1073/pnas.1619040114
Daura J, Sanz M, Deschamps M et al (2018) A 400,000-year-old acheulean assemblage associated with the Aroeira-3 human cranium (Gruta da Aroeira, Almonda karst system, Portugal). CR Palevol 17:594–615. https://doi.org/10.1016/j.crpv.2018.03.003
de Juana S, Galán AB, Domínguez-Rodrigo M (2010) Taphonomic identification of cut marks made with lithic handaxes: an experimental study. J Archaeol Sci 37:1841–1850
de Lumley M-A (2015) L’homme de Tautavel. Un Homo erectus européen évolué. Homo erectus tautavelensis. Anthropologie 119:303–348. https://doi.org/10.1016/j.anthro.2015.06.001
Dominguez-Rodrigo M, Yravedra J (2009) Why are cut mark frequencies in archaeofaunal assemblages so variable? A multivariate analysis. J Archaeol Sci 36:884–894
Dominguez-Rodrigo M, Serrallonga J, Juan-Tresserras J et al (2001) Woodworking activities by early humans: a plant residue analysis on Acheulian stone tools from Peninj (Tanzania). J Hum Evol 40:289–299
Egeland CP (2003) Carcass processing intensity and cutmark creation: an experimental approach. Plains Anthropol 48:39–51. https://doi.org/10.1080/2052546.2003.11949279
Elton S, O’Regan HJ (2014) Macaques at the margins: the biogeography and extinction of Macaca sylvanus in Europe. Quat Sci Rev 96:117–130. https://doi.org/10.1016/j.quascirev.2014.04.025
Fernández-Jalvo Y, Andrews P (2016) Atlas of taphonomic identifications. Springer Netherlands, Dordrecht
Gabucio MJ, Sanz M, Daura J (2024) Lynxes and foxes among hyenas, bears and humans. Unit 3 of the Cova Del Coll Verdaguer Upper Pleistocene site (Barcelona, Iberian Peninsula). Quat Sci Rev 333:108671. https://doi.org/10.1016/j.quascirev.2024.108671
Galán AB, Domínguez-Rodrigo M (2013) An experimental study of the Anatomical Distribution of Cut Marks Created by Filleting and Disarticulation on Long Bone ends. Archaeometry 55:1132–1149. https://doi.org/10.1111/j.1475-4754.2012.00730.x
Gowlett JAJ (1988) A case of developed Oldowan in the Acheulean? World Archaeol 20:13–26. https://doi.org/10.1080/00438243.1988.9980053
Grandal-d’Anglade A, Pérez-Rama M, García-Vázquez A, González-Fortes GM (2019) The cave bear’s hibernation: reconstructing the physiology and behaviour of an extinct animal. Hist Biol 31:429–441. https://doi.org/10.1080/08912963.2018.1468441
Hoffmann DL, Pike AWG, Wainer K, Zilhão J (2013) New U-series results for the speleogenesis and the palaeolithic archaeology of the Almonda karstic system (Torres Novas, Portugal). Quat Int 294:168–182. https://doi.org/10.1016/j.quaint.2012.05.027
Johnson E (1985) Current developments in bone technology. Advances in archaeological method and theory. Elsevier, pp 157–235
King T, Andrews P, Boz B (1999) Effect of taphonomic processes on dental microwear. Am J Phys Anthropol 108:359–373. https://doi.org/10.1002/(SICI)1096-8644(199903)108:3%3C359::AID-AJPA10%3E3.0.CO;2-9
Konidaris GE, Athanassiou A, Panagopoulou E, Harvati K (2022) First record of Macaca (Cercopithecidae, Primates) in the middle pleistocene of Greece. J Hum Evol 162:103104. https://doi.org/10.1016/j.jhevol.2021.103104
Lam YM, Chen X, Pearson OM (1999) Intertaxonomic variability in patterns of bone density and the differential representation of Bovid, Cervid, and Equid elements in the archaeological record. Am Antiq 64:343–362. https://doi.org/10.2307/2694283
Lebreton L, Moigne AM, Filoux A, Perrenoud C (2017) A specific small game exploitation for lower paleolithic: the beaver (Castor fiber) exploitation at the Caune de l’Arago (Pyrénées-Orientales, France). J Archaeol Sci Rep. https://doi.org/10.1016/j.jasrep.2016.11.023
Linscott B, Pike AWG, Angelucci DE et al (2023) Reconstructing Middle and Upper Paleolithic human mobility in Portuguese Estremadura through laser ablation strontium isotope analysis. Proc Natl Acad Sci USA 120. https://doi.org/10.1073/pnas.2204501120
López-García JM, Blain H-A, Sanz M, Daura j, Zilhão j (2018) Refining the environmental and climatic background of the middle pleistocene human cranium from Gruta da Aroeira (Torres Novas, Portugal). Quat Sci Rev 200:367–375. https://doi.org/10.1016/j.quascirev.2018.10.003
Lupo KD, O’Connell JF (2002) Cut and tooth mark distributions on large animal bones: ethnoarchaeological data from the Hadza and their implications for current ideas about early human carnivory. J Archaeol Sci 29:85–109. https://doi.org/10.1006/jasc.2001.0690
Lyman L (1987) Archaeofaunas and butchery studies: a taphonomic perspective. Adv Archaeol Method Theory 10:249–337. https://doi.org/10.2307/20210090
Lyman RL (1994) Vertebrate taphonomy. Cambridge University Press, Cambridge
Lyman RL (2005) Analyzing cut marks: lessons from artiodactyl remains in the northwestern United States. J Archaeol Sci 32:1722–1732. https://doi.org/10.1016/j.jas.2005.06.003
Lyman RL (2008) Quantitative paleozoology. Cambridge University Press, Cambridge
Manzi G, Magri D, Palombo MR (2011) Early-middle pleistocene environmental changes and human evolution in the Italian peninsula. Quat Sci Rev 30:1420–1438. https://doi.org/10.1016/j.quascirev.2010.05.034
Marks AE, Brugal J-P, Chabai VP et al (2002a) Le gisement pléistocène moyen de Galeria Pesada, (Estrémadure, Portugal): premiers résultats. Paléo 14:77–100
Marks AE, Monigal K, Chabai VP et al (2002b) Excavations at the late Middle Pleistocene cave of Galeria Pesada: 1997/1999. O Arqueólogo Port 20:7–38
Méndez-Quintas E, Santonja M, Pérez-González A et al (2018) First evidence of an extensive Acheulean large cutting tool accumulation in Europe from Porto Maior (Galicia, Spain). Sci Rep 8:3082. https://doi.org/10.1038/s41598-018-21320-1
Moncel M-H, Moigne A-M, Combier J (2012) Towards the Middle Palaeolithic in Western Europe: the case of Orgnac 3 (southeastern France). J Hum Evol 63:653–666. https://doi.org/10.1016/j.jhevol.2012.08.001
Pelletier M (2018) Évolution morphométrique et Biogéographie des Léporidés dans les environnements méditerranéens Au Pléistocène implications socio-économiques pour les sociétés humaines. Ph.D.Thesis, Aix*Marseille Université
Pollock CR, Pokines JT, Bethard JD (2018) Organic staining on bone from exposure to wood and other plant materials. Forensic Sci Int 283:200–210. https://doi.org/10.1016/j.forsciint.2017.12.021
Pope M, Parfitt S, Roberts M et al (2020) The horse butchery site. A high resolution record of lower palaeolithic hominin behaviour at Boxgrove, UK. SpoilHeap Publications, UK
Poplin F (1981) Un problème d’ostéologie quantitative: calcul d’effectif initial d’après appariements. Généralisation aux autres types de Remontages Et à d’autres matériels archéologiques. Rev d’Archéométrie 5:159–165
Rabinovich R, Gaudzinski-Windheuser S, Goren-Inbar N (2008) Systematic butchering of fallow deer (Dama) at the early middle Pleistocene Acheulian site of Gesher Benot Ya‘aqov (Israel). J Hum Evol 54:134–149. https://doi.org/10.1016/j.jhevol.2007.07.007
Reitz EJ, Wing ES (2008) Zooarchaeology. Cambridge University Press, Cambridge
Rivals F, Sanz M, Daura J (2016) First reconstruction of the dietary traits of the Mediterranean deer (Haploidoceros mediterraneus) from the Cova Del Rinoceront (NE Iberian Peninsula). Palaeogeogr Palaeoclimatol Palaeoecol 449:101–107. https://doi.org/10.1016/j.palaeo.2016.02.014
Roberts MB, Parfitt SA (1999) Boxgrove. A middle pleistocene hominid site at Eartham Quarry, Boxgrove, West Sussex. English Heritage, London
Rodríguez-Hidalgo A, Saladié P, Ollé A, Carbonell E (2015) Hominin subsistence and site function of TD10.1 bone bed level at Gran Dolina site (Atapuerca) during the late Acheulean. J Quat Sci. https://doi.org/10.1002/jqs.2815
Rodríguez-Hidalgo A, Rivals F, Saladié P, Carbonell E (2016) Season of bison mortality in TD10.2 bone bed at Gran Dolina site (Atapuerca): integrating tooth eruption, wear, and microwear methods. J Archaeol Sci Rep 6:780–789. https://doi.org/10.1016/j.jasrep.2015.11.033
Rodríguez-Hidalgo A, Saladié P, Ollé A et al (2017) Human predatory behavior and the social implications of communal hunting based on evidence from the TD10.2 bison bone bed at Gran Dolina (Atapuerca, Spain). J Hum Evol 105:89–122. https://doi.org/10.1016/j.jhevol.2017.01.007
Rodríguez-Hidalgo A, Sanz M, Daura J, Sánchez-Marco A (2020) Taphonomic criteria for identifying Iberian lynx dens in quaternary deposits. Sci Rep 10:7225. https://doi.org/10.1038/s41598-020-63908-6
Saladié P, Huguet R, Rodríguez-Hidalgo A et al (2012) Intergroup cannibalism in the European early pleistocene: the range expansion and imbalance of power hypotheses. J Hum Evol 63:682–695. https://doi.org/10.1016/j.jhevol.2012.07.004
Santonja M, Pérez-González A (2021) Revisión del componente achelense de la industria lítica de Galería (Atapuerca, Burgos, España). Trab Prehist 78:26–48. https://doi.org/10.3989/tp.2021.12263
Santonja M, Pérez-González A, Baena J et al (2022) The Acheulean of the Upper Guadiana River Basin (Central Spain). Morphostratigraphic Context and Chronology. Front Earth Sci 10. https://doi.org/10.3389/feart.2022.912007
Sanz M, Daura J (2018) Carnivore involvement in bone assemblages based on taphonomic and zooarchaeological analyses of Cova del Coll Verdaguer site (Barcelona, Iberian Peninsula). Hist Biol 30:807–820. https://doi.org/10.1080/08912963.2017.1351561
Sanz M, Daura J, Brugal J-P (2014) First occurrence of the extinct deer haploidoceros in the Iberian Peninsula in the Upper Pleistocene of the Cova Del Rinoceront (Castelldefels, Barcelona). CR Palevol 13:27–40. https://doi.org/10.1016/j.crpv.2013.06.005
Sanz M, Sala N, Daura J et al (2018) Taphonomic inferences about middle pleistocene hominins: the human cranium of Gruta da Aroeira (Portugal). Am J Phys Anthropol 167:615–627. https://doi.org/10.1002/ajpa.23689
Sanz M, Daura J, Cabanes D et al (2020) Early evidence of fire in south-western Europe: the Acheulean site of Gruta da Aroeira (Torres Novas, Portugal). Sci Rep 10:12053. https://doi.org/10.1038/s41598-020-68839-w
Selvaggio MM, Wilder J (2001) Identifying the involvement of multiple Carnivore taxa with archaeological bone assemblages. J Archaeol Sci 28:465–470
Semprebon GM, Godfrey LR, Solounias N et al (2004) Can low-magnification stereomicroscopy reveal diet? J Hum Evol 47:115–144. https://doi.org/10.1016/j.jhevol.2004.06.004
Shipman P, Rose J (1983) Early hominid hunting, butchering, and carcass-processing behaviors: approaches to the fossil record. J Anthropol Archaeol 2:57–98
Smith GM (2013) Taphonomic resolution and hominin subsistence behaviour in the Lower Palaeolithic: differing data scales and interpretive frameworks at Boxgrove and Swanscombe (UK). J Archaeol Sci 40:3754–3767
Solodenko N, Zupancich A, Cesaro SN et al (2015) Fat residue and use-wear found on acheulian biface and scraper associated with butchered elephant remains at the site of Revadim, Israel. PLoS ONE 10:e0118572. https://doi.org/10.1371/journal.pone.0118572
Solounias N, Semprebon G (2002) Advances in the Reconstruction of Ungulate Ecomorphology with application to early Fossil equids. Am Museum Novit 3366:1–49. https://doi.org/10.1206/0003-0082(2002)366%3C0001:AITROU%3E2.0.CO;2
Stahlschmidt MC, Miller CE, Ligouis B et al (2015) The depositional environments of Schöningen 13 II-4 and their archaeological implications. J Hum Evol 89:71–91. https://doi.org/10.1016/j.jhevol.2015.07.008
Stiner MC (1998) Mortality analysis of pleistocene bears and its paleoanthropological relevance. J Hum Evol 34:303–326
Stiner MC (2005) The faunas of Hayonim Cave, Israel: a 200,000-year record of Paleolithic diet, demography, and society. Harvard University Press, USA
Stiner MC, Barkai R, Gopher A (2009) Cooperative hunting and meat sharing 400 – 200 kya at Qesem Cave, Israel. Proc Natl Acad Sci USA 106:13207–13212
Stiner MC, Gopher A, Barkai R (2011) Hearth-side socioeconomics, hunting and paleoecology during the late Lower Paleolithic at Qesem Cave, Israel. J Hum Evol 60:213–233
Torre I, Ribas A, Flaquer C, Arrizabalaga A (2009) Nuevos datos sobre la distribución y abundancia del ratón leonado Apodemus flavicollis (Melchior, 1834) en Catalunya. Galemys. Span J Mammal 21:3–11. https://doi.org/10.7325/galemys.2009.ne.a1
Trinkaus E, Marks AE, Brugal J-P et al (2003) Later middle Pleistocene human remains from the Almonda Karstic System, Torres Novas, Portugal. J Hum Evol 45:219–226. https://doi.org/10.1016/j.jhevol.2003.07.001
Uzunidis A, Pineda A, Jiménez-Manchón S et al (2021) The impact of sediment abrasion on tooth microwear analysis: an experimental study. Archaeol Anthropol Sci 13. https://doi.org/10.1007/s12520-021-01382-5
Van Kolfschoten T, Buhrs E, Verheijen I (2015) The larger mammal fauna from the Lower Paleolithic Schöningen Spear site and its contribution to hominin subsistence. J Hum Evol 89:138–153. https://doi.org/10.1016/j.jhevol.2015.09.014
Van Kolfschoten T, Knul M, Buhrs E, Gielen M (2018) Butchered large bovids (Bos primigenius and Bison priscus) from the Palaeolithic throwing spear site Schöningen 13 II-4 (Germany). In: A Bouquet of Archaeozoological Studies, pp 33–46
Villa P, Lenoir M (2009) Hunting and hunting weapons of the lower and middle paleolithic of Europe. In: Vertebrate paleobiology and paleoanthropology, pp 59–85
Villa P, Mahieu E (1991) Breakage patterns of human long bones. J Hum Evol 21:27–48
Villa P, Castel J-C, Beauval C et al (2004) Human and carnivore sites in the European Middle and Upper Paleolithic: similarities and differences in bone modification and fragmentation. In: Brugal, J.-P. & Fosse P (ed) Actes du XIV Congrès UISPP ‘Hommes & Carnivores au Paléolithique’, Liège 2001. Revue de Paléobiologie, pp 705–730
Villa P, Soto E, Santonja M et al (2005) New data from Ambrona: closing the hunting versus scavenging debate. Quat Int 126–128:223–250. https://doi.org/10.1016/j.quaint.2004.03.001
Yravedra J, Dominguez-Rodrigo M, Santonja M et al (2010) Cut marks on the Middle Pleistocene elephant carcass of Aridos 2 (Madrid, Spain). J Archaeol Sci 37:2469–2476
Zilhão J (2023) Portugal na Idade do Gelo: Território E Habitantes. Fundação Francisco Manuel dos Santos, Lisboa
Zilhão J (2024) The palaeolithic archaeology of the Almonda karst system (Portugal): a window into half-a-million years of western Iberian prehistory. In: Uthmeier T, Maier A (eds) STONE AGE. Studying technologies of non-analogous environments and glacial ecosystems. Papers in Honor of Jürgen Richter. Dr. Rudolf Habelt GmbH, Bonn, pp 117–134
Zilhão J, Maurício J, Souto P (1993) Jazidas arqueológicas do sistema cársico da nascente do Almonda. Nov Augusta 7:35–54
Zilhão J, Angelucci DE, Argant J et al (2010) Humans and hyenas in the Middle Paleolithic of Gruta da Oliveira (Almonda karstic system, Torres Novas, Portugal). In: Baquedano E, Rosell J (eds) 1a Reunión de científicos sobre cubiles de hiena (y otros grandes carnívoros) en los yacimientos arqueológicos de la Península Ibérica. Zona Arqueológica 13, pp 298–308
Zilhão J, Angelucci DE, Arnold LJ et al (2021) A revised, last interglacial chronology for the Middle palaeolithic sequence of Gruta da Oliveira (Almonda karst system, Torres Novas, Portugal). Quat Sci Rev 258:106885. https://doi.org/10.1016/j.quascirev.2021.106885
Zunino M, Pavia M, Fernández-López SR, Pavia G (2012) Taphonomic analysis of the Lower Pleistocene Pirro Nord locality (Pirro 10 site, Puglia, Southern Italy): a depositional model for vertebrate assemblages in a karstic environment. Palaios 27:3–18. https://doi.org/10.2110/palo.2011.p11-029r
Acknowledgements
This research benefited from financial support from the following agencies: Fundação para a Ciência e Tecnologia (FCT), Portugal (grants UIDB/00698/2020, UIDP/00698/2020, and project PTDC/HAR-ARQ/30413/2017 – ARQEVO, Archaeology and Evolution of Early Humans in the Western Façade of Iberia); Ramon y Cajal postdoctoral grant (RYC2021-032999-I, M.S.) with financial sponsorship from the Ministerio de Ciencia e Innovación and the European Union NextGenerationEU; AGAUR Generalitat de Catalunya (SGR2021-00337 and SGR2021-01237); Ministerio de Ciencia e Innovación, Government of Spain (PID2020-113960GB-100/AEI/10.13039/501100011033 and PID2022-138590NB-C41/AEI/10.13039/501100011033) and CNRS-INEE International Research Network program (grant no. IRN0871TaphEN). Over the years, the archaeological field work carried out in the Almonda karst has benefited from the logistical support provided by a number of different institutions, primarily the following: Câmara Municipal de Torres Novas, RENOVA–Fábrica de Papel do Almonda, and Sociedade Torrejana de Espeleologia e Arqueologia (STEA). We also thank Paleoalmonda, Associação de Estudos Científicos, of which the authors M.S., J.D. and J.Z are members.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. M. Sanz was sponsored by a Ramon y Cajal postdoctoral grant (RYC2021-032999-I, M.S.) with financial sponsorship from the Ministerio de Ciencia e Innovación and the European Union NextGenerationEU.
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
M.S. collected the data, designed and conducted the research, wrote the main manuscript with contributions from the other authors (J.D. and J.Z.: archaeological context and chronology, F.R.: tooth microwear analyses). M.S., J.D. and J.Z. directed the fieldwork. All authors reviewed, commented on, and participated in improving the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sanz, M., Daura, J., Rivals, F. et al. The residential occupation of the Gruta da Aroeira (Almonda, Portugal) cave site: shedding light on hunting and subsistence practices in the Middle Pleistocene of western Eurasia. Archaeol Anthropol Sci 16, 139 (2024). https://doi.org/10.1007/s12520-024-02026-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12520-024-02026-0