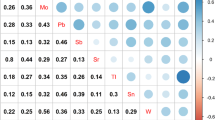
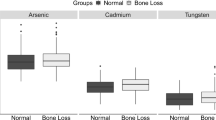
Abstract
Studies have shown that exposure to heavy metals increases the risk of osteoporosis in the elderly population and leads to fractures. However, epidemiologic studies focus on combined effects of multiple metals on bone strength among preschoolers are scarce. Therefore, this study was conducted to examine associations of multiple metals exposure with the risk of insufficient bone strength (IBS) among 1399 children aged 2–6 years in China. Urine concentrations of 23 metals were measured using an inductively coupled plasma mass spectrometer, while bone strength was assessed using a quantitative ultrasound instrument. The weighted quantile sum (WQS) and Bayesian kernel machine regression (BKMR) models were conducted to investigate the associations between exposure to metal mixtures and IBS. In the single metal exposure model, urine cobalt and zinc were identified to be negatively associated with IBS, whereas urine lead was positively associated with IBS. Furthermore, WQS regression and BKMR analyses consistently showed that exposure to metal mixtures was inversely related to IBS, and cobalt was the main contributor to the combined effects. Such associations were more evident in children aged 2–4 years. Our findings suggested that exposure to metal mixtures was associated with bone strength among children, and cobalt was the main contributor to the overall effects.
Graphical Abstract



Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Akesson A, Bjellerup P, Lundh T, Lidfeldt J, Nerbrand C, Samsioe G et al (2006) Cadmium-induced effects on bone in a population-based study of women. Environ Health Perspect 114:830–834. https://doi.org/10.1289/ehp.8763
Alghadir AH, Gabr SA, Al-Eisa ES, Alghadir MH (2016) Correlation between bone mineral density and serum trace elements in response to supervised aerobic training in older adults. Clin Interv Aging 11:265–273. https://doi.org/10.2147/CIA.S100566
Banjabi AA, Kannan K, Kumosani TA, Yousef JM, Abulnaja KO, Moselhy SS (2021) Association of blood heavy metal levels with osteocalcin abnormality and incidence of osteoporosis in Saudi subjects. Braz J Biol 83:e248828. https://doi.org/10.1590/1519-6984.248828
Beier EE, Maher JR, Sheu T-J et al (2013) Heavy metal lead exposure, osteoporotic-like phenotype in an animal model, and depression of Wnt signaling. Environ Health Perspect 121:97–104. https://doi.org/10.1289/ehp.1205374
Bobb JF, Valeri L, Claus Henn B, Christiani DC, Wright RO, Mazumdar M et al (2015) Bayesian kernel machine regression for estimating the health effects of multi-pollutant mixtures. Biostatistics 16:493–508. https://doi.org/10.1093/biostatistics/kxu058
Bobb JF, Claus Henn B, Valeri L, Coull BA (2018) Statistical software for analyzing the health effects of multiple concurrent exposures via Bayesian kernel machine regression. Environ Health 17:67. https://doi.org/10.1186/s12940-018-0413-y
Campbell JR, Auinger P (2007) The association between blood lead levels and osteoporosis among adults–results from the third national health and nutrition examination survey (NHANES III). Environ Health Perspect 115:1018–1022. https://doi.org/10.1289/ehp.9716
Carrico C, Gennings C, Wheeler DC, Factor-Litvak P (2015) Characterization of weighted quantile sum regression for highly correlated data in a risk analysis setting. J Agric Biol Environ Stat 20:100–120. https://doi.org/10.1007/s13253-014-0180-3
Chang L, Shen S, Zhang Z, Song X, Jiang Q (2018) Study on the relationship between age and the concentrations of heavy metal elements in human bone. Ann Transl Med 6:320–327
Chen L, Zhao Y, Liu F, Chen H, Tan T, Yao P et al (2022) Biological aging mediates the associations between urinary metals and osteoarthritis among U.S. adults. BMC Med 20:207–219. https://doi.org/10.1186/s12916-022-02403-3
Chung SM (2022) Long-term sex-specific effects of cadmium exposure on osteoporosis and bone density: a 10-year community-based cohort study. J Clin Med 11:2899–2909. https://doi.org/10.3390/jcm11102899
Di Marcello F, Di Donato G, d’Angelo DM, Breda L, Chiarelli F (2022) Bone health in children with rheumatic disorders: focus on molecular mechanisms, diagnosis, and management. Int J Mol Sci 23:5725–5749. https://doi.org/10.3390/ijms23105725
Dowd TL, Rosen JF, Mints L, Gundberg CM (2001) The effect of Pb2+ on the structure and hydroxyapatite binding properties of osteocalcin. Biochem Biophys Acta 1535:153–163. https://doi.org/10.1016/s0925-4439(00)00094-6
Galvez-Fernandez M, Grau-Perez M, Garcia-Barrera T, Ramirez-Acosta S, Gomez-Ariza JL, Perez-Gomez B et al (2021) Arsenic, cadmium, and selenium exposures and bone mineral density-related endpoints: the HORTEGA study. Free Radic Biol Med 162:392–400. https://doi.org/10.1016/j.freeradbiomed.2020.10.318
Hans D, Šteňová E, Lamy O (2017) The trabecular bone score (TBS) complements DXA and the FRAX as a fracture risk assessment tool in routine clinical practice. Curr Osteoporos Rep 15:521–531. https://doi.org/10.1007/s11914-017-0410-z
Heitland P, Koster HD (2006) Biomonitoring of 30 trace elements in urine of children and adults by ICP-MS. Clin Chim Acta 365:310–318. https://doi.org/10.1016/j.cca.2005.09.013
Howe CG, Margetaki K, Vafeiadi M, Roumeliotaki T, Karachaliou M, Kogevinas M et al (2021) Prenatal metal mixtures and child blood pressure in the Rhea mother-child cohort in Greece. Environ Health 20:1–16. https://doi.org/10.1186/s12940-020-00685-9
Hyun TH, Elizabeth BC, Milne DB (2004) Zinc intakes and plasma concentrations in men with osteoporosis: the Rancho Bernardo Study. Am J Clin Nutr. https://doi.org/10.1556/AAlim.33.2004.3.13
Inose T, Takano T, Nakamura K, Kizuki M, Seino K (2006) Tibial cortical bone properties of preadolescents and their mothers in an urban area associated with lifestyle: a longitudinal study. Acta Paediatr 95:276–282. https://doi.org/10.1080/08035250500352169
Jalili C, Kazemi M, Taheri E, Mohammadi H, Boozari B, Hadi A et al (2020) Exposure to heavy metals and the risk of osteopenia or osteoporosis: a systematic review and meta-analysis. Osteoporos Int 31:1671–1682. https://doi.org/10.1007/s00198-020-05429-6
Katerina T, Fernando R (2019) The genetic architecture of osteoporosis and fracture risk. Bone 126:2–10. https://doi.org/10.1016/j.bone.2019.04.005
Kim ES, Shin S, Lee YJ, Ha IH (2021) Association between blood cadmium levels and the risk of osteopenia and osteoporosis in Korean post-menopausal women. Arch Osteoporos 16:22–32. https://doi.org/10.1007/s11657-021-00887-9
King J, Keen C (1999) Zinc. In: Shils M, Olson J, Shike M (eds) Modern nutrition in health and disease. Williams & Wilkins, Baltimore, pp 223–239
Lequin MH, Rijn RRV, Robben SGF, Hop WCJ, Kuijk CV (2000) Normal values for tibial quantitative ultrasonometry in Caucasian children and adolescents (aged 6 to 19 years). Calcif Tissue Int 67:101–105. https://doi.org/10.1007/s00223001119
Leyssens L, Vinck B, Van Der Straeten C, Wuyts F et al (2017) Cobalt toxicity in humans—a review of the potential sources and systemic health effects. Toxicology 387:43–56. https://doi.org/10.1016/j.tox.2017.05.015
Li JJ, Pang LN, Wu S, Zeng M (2018) Advances in the effect of heavy metals in aquatic environment on the health risks for bone. IOP Conf Ser 186:012057–012066. https://doi.org/10.1088/1755-1315/186/3/012057
Li T, Xie Y, Wang L, Huang G, Cheng Y, Hou D et al (2022) The association between lead exposure and bone mineral density in childhood and adolescence: results from NHANES 1999–2006 and 2011–2018. Nutrients 14:1523–1532. https://doi.org/10.3390/nu14071523
Liang D, Yang M, Guo B, Cao J, Yang L, Guo XD (2012) Zinc upregulates the expression of osteoprotegerin in mouse osteoblasts MC3T3-E1 through PKC/MAPK pathways. Biol Trace Elem Res 146:340–348. https://doi.org/10.1007/s12011-011-9254-z
Lim HS, Lee HH, Kim TH, Lee BR (2016) Relationship between heavy metal exposure and bone mineral density in Korean adult. J Bone Metab 23:223–231
Liu SH, Bobb JF, Claus Henn B, Gennings C, Schnaas L, Tellez-Rojo M et al (2018) Bayesian varying coefficient kernel machine regression to assess neurodevelopmental trajectories associated with exposure to complex mixtures. Stat Med 37:4680–4694. https://doi.org/10.1002/sim.7947
Liu Y, Yu L, Zhu M, Lin W, Liu Y, Li M et al (2022) Associations of exposure to multiple metals with blood pressure and hypertension: a cross-sectional study in Chinese preschool children. Chemosphere 307:135985–136004. https://doi.org/10.1016/j.chemosphere.2022.135985
Maggioli C, Stagi S (2017) Bone modeling, remodeling, and skeletal health in children and adolescents: mineral accrual, assessment and treatment. Ann Pediatr Endocrinol Metab 22:1–5. https://doi.org/10.6065/apem.2017.22.1.1
Mahaffey KR, Vanderveen JE (1979) Nutrient-toxicant interactions: susceptible populations. Environ Health Perspect 29:81–87. https://doi.org/10.1289/ehp.792981
Moradi S, Khorrami-Nezhad L, Ali-Akbar S, Zare F, Alipour T, Azadeh DKB et al (2018) The associations between dietary patterns and bone health, according to the TGF-β1 T869→C polymorphism, in postmenopausal Iranian women. Aging Clin Exp Res 30:563–571. https://doi.org/10.1007/s40520-017-0828-2
Moran JM, Lopez-Arza LG, Lavado-Garcia JM, Pedrera-Canal M, Rey-Sanchez P, Rodriguez-Velasco FJ et al (2013) Hormonal relationships to bone mass in elderly Spanish men as influenced by dietary calcium and vitamin D. Nutrients 5:4924–4937. https://doi.org/10.3390/nu5124924
Oviedo-Rondón E, Ferket PR, Havestein GB (2006) Understanding long bone development in broilers and turkeys. Avian Poultry Biol Rev 17:77–88
Park RM, An Y (2022) Continuous NHANES survey data for environmental ambient and occupational hazard identification-feasibility and preliminary findings for osteoporosis and kidney disease. J Occup Environ Hygiene. https://doi.org/10.1080/15459624.2022.2088769
Paustenbach DJ, Tvermoes BE, Unice KM, Finley BL, Kerger BD (2013) A review of the health hazards posed by cobalt. Crit Rev Toxicol 43:316–362. https://doi.org/10.3109/10408444.2013.779633
Qu X, He Z, Qiao H, Zhai Z, Mao Z, Yu Z et al (2018) Serum copper levels are associated with bone mineral density and total fracture. J Orthop Transl 14:34–44. https://doi.org/10.1016/j.jot.2018.05.001
Razmandeh R, Nasli-Esfahani E, Heydarpour R, Faridbod F, Ganjali MR, Norouzi P et al (2014) Association of zinc, copper and magnesium with bone mineral density in Iranian postmenopausal women—a case control study. J Diabetes Metab Disord 13:43–49. https://doi.org/10.1186/2251-6581-13-43
Rodriguez J, Mandalunis PM (2018) A review of metal exposure and its effects on bone health. J Toxicol 2018:4854152–4854163. https://doi.org/10.1155/2018/4854152
Rondanelli M, Peroni G, Gasparri C, Infantino V, Naso M, Riva A et al (2021) An overview on the correlation between blood zinc, zinc intake, zinc supplementation and bone mineral density in humans. Acta Ortop Mex 35:142–152
Ryz NR, Weiler HA, Taylor CG (2009) Zinc deficiency reduces bone mineral density in the spine of young adult rats: a pilot study. Ann Nutr Metab 54:218–226. https://doi.org/10.1159/000224627
Si L, Winzenberg TM, Jiang Q, Chen M, Palmer AJ (2015) Projection of osteoporosis-related fractures and costs in China: 2010–2050. Osteoporos Int 26:1929–1937. https://doi.org/10.1007/s00198-015-3093-2
Silbergeld EK, Schwartz J, Mahaffey K (1988) Lead and osteoporosis: mobilization of lead from bone in postmenopausal women. Environ Res 47:79–94. https://doi.org/10.1016/s0013-9351(88)80023-9
Simonsen LO, Harbak H, Bennekou P (2012) Cobalt metabolism and toxicology—a brief update. Sci Total Environ 432:210–215. https://doi.org/10.1016/j.scitotenv.2012.06.009
Starcher BC, Hill CH, Madaras JG (1980) Effect of zinc deficiency on bone collagenase and collagen turnover. J Nutr 110:2095–2102. https://doi.org/10.1093/jn/110.10.2095
Wang WJ, Wu CC, Jung WT, Lin CY (2019) The associations among lead exposure, bone mineral density, and FRAX score: NHANES, 2013 to 2014. Bone 128:115045. https://doi.org/10.1016/j.bone.2019.115045
Wang WJ, Huang MN, Wang CK, Yang AM, Lin CY (2021) Zinc status is independently related to the bone mineral density, fracture risk assessment tool result, and bone fracture history: results from a US nationally representative survey. J Trace Elem Med Biol 67:126765. https://doi.org/10.1016/j.jtemb.2021.126765
Weaver CM, Gordon CM, Janz KF, Kalkwarf HJ, Lappe JM, Lewis R et al (2016) The National Osteoporosis Foundation’s position statement on peak bone mass development and lifestyle factors: a systematic review and implementation recommendations. Osteoporos Int 27:1281–1386. https://doi.org/10.1007/s00198-015-3440-3
Wei MH, Cui Y, Zhou HL, Song WJ, Di DS, Zhang RY et al (2021) Associations of multiple metals with bone mineral density: a population-based study in US adults. Chemosphere 282:131150–131158. https://doi.org/10.1016/j.chemosphere.2021.131150
Wu C-C, Wang C-K, Yang A-M, Lu C-S, Lin C-Y (2020) Selenium status is independently related to bone mineral density, FRAX score, and bone fracture history: NHANES, 2013 to 2014. Bone 143:115631. https://doi.org/10.1016/j.bone.2020.115631
Acknowledgements
We thank all study participants for their valuable time and their support of our study. Furthermore, we would like to thank in particular Shiyan Maternal and Child Health Hospital for their help in collecting the biological sample and interviewing the study population participants.
Funding
This work was supported by the National Nature Science Foundation of China [Nos. 81872698 and 71774049]; Innovative Research Team of Hubei Provincial Department of Education [T2021014]; Advantages Discipline Group (Public health) Project in Higher Education of Hubei Province (2021–2025) [2022PHXKQ3]; Starting Fund of Hubei University of Medicine [2020QDJZR029].
Author information
Authors and Affiliations
Contributions
YL and YW have contributed equally to the work and share first authorship. YL and YW: conceptualization, formal analysis, visualization, data curation, writing—original draft preparation. YF, YL, ML, and MH: data curation, investigation, data collection, visualization. YZ, CL and HD: investigation, project administration, resources, supervision. XL and HJ: investigation, data curation, resources, supervision, project administration. JW: conceptualization, writing—review and editing, supervision, project administration, funding acquisition. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
No financial or non-financial benefits have been received or will be received from any party related directly or indirectly to the subject of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Wang, Y., Fu, Y. et al. Associations Between Exposure to Metal Mixtures and Insufficient Bone Strength: A Cross-Sectional Study Among Children Aged 2–6 Years. Expo Health (2023). https://doi.org/10.1007/s12403-023-00591-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12403-023-00591-8