Abstract
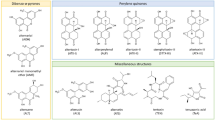
Due to climate change, mycotoxins are expected to become a specific concern worldwide. In the future, predicted changes in environmental conditions will affect the growth of crops and may favor the development of fungi and, therefore, the presence of mycotoxins. In addition to direct human oral exposure to mycotoxins through cereal food products, potential human exposure may also occur as a result of crop contamination with mycotoxins via animal feed and consumption of meat or milk products. Fungi can produce numerous compounds, many of which have not yet been characterized, including in terms of their toxicological potency. A large number of mycotoxins and their metabolites have not been evaluated for their toxicity so far. In this study, an innovative combined strategy based on several validated in silico tools was used to assess specific toxicity endpoints. From a list of 552 mycotoxins, 12 mycotoxins were clustered together based on physico-chemical parameters. On this specific cluster, firstly quantitative structure–activity relationship (QSAR) tools were used to assess the mutagenic and carcinogenic potential of each compound. From this analysis, 12 mycotoxins were found to have a potential activity in cancer promotion. The link between these compounds and cancer activity was further investigated by two complementary approaches: identification of gene pathways involved in the toxic response and a datamining search. Altogether, the results point to a potential association between these mycotoxins and lung cancer.





adapted from the KEGG representation
Similar content being viewed by others
Data Availability
List of 552 Mycotoxins and associated Smiles and JOELib descriptors are available on Zenondo (https://zenodo.org), on https://doi.org/10.5281/zenodo.5977517.
Code Availability
Not applicable.
References
Ahn M-Y (2018) HDAC inhibitor apicidin suppresses murine oral squamous cell carcinoma cell growth in vitro and in vivo via inhibiting HDAC8 expression. Oncol Lett 16:6552–6560. https://doi.org/10.3892/ol.2018.9468
AlMatar M, Makky EA (2016) Cladosporium cladosporioides from the perspectives of medical and biotechnological approaches. 3 Biotech 6:4. https://doi.org/10.1007/s13205-015-0323-4
Alonso-Jauregui M, Font M, González-Peñas E, López de Cerain A, Vettorazzi A (2021) Prioritization of mycotoxins based on their genotoxic potential with an in silico-in vitro strategy. Toxins (basel) 13:734. https://doi.org/10.3390/toxins13100734
Arcangeli G, Traversini V, Tomasini E, Baldassarre A, Lecca LI, Galea RP, Mucci N (2020) Allergic anaphylactic risk in farming activities: a systematic review. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph17144921
Backman TWH, Cao Y, Girke T (2011) ChemMine tools: an online service for analyzing and clustering small molecules. Nucleic Acids Res 39:W486-491. https://doi.org/10.1093/nar/gkr320
Banerjee P, Eckert AO, Schrey AK, Preissner R (2018) ProTox-II: a webserver for the prediction of toxicity of chemicals. Nucleic Acids Res 46:W257–W263. https://doi.org/10.1093/nar/gky318
Benfenati E, Manganaro A, Gini G (2013) VEGA-QSAR: AI inside a platform for predictive toxicology. In: CEUR Workshop Proceedings. Presented at the 2nd Workshop on Popularize Artificial Intelligence, PAI 2013, Held in Conjunction with AI*IA 2013, CEUR-WS. pp 21–28
Bernacki DT, Bryce SM, Bemis JC, Dertinger SD (2019) Aneugen molecular mechanism assay: proof-of-concept with 27 reference chemicals. Toxicol Sci 170:382–393. https://doi.org/10.1093/toxsci/kfz123
Brazelle W, Kreahling JM, Gemmer J, Ma Y, Cress WD, Haura E, Altiok S (2010) Histone deacetylase inhibitors downregulate checkpoint kinase 1 expression to induce cell death in non-small cell lung cancer cells. PLoS ONE 5:e14335. https://doi.org/10.1371/journal.pone.0014335
Carvaillo J-C, Barouki R, Coumoul X, Audouze K (2019) Linking bisphenol S to adverse outcome pathways using a combined text mining and systems biology approach. Environ Health Perspect 127:47005. https://doi.org/10.1289/EHP4200
Chang J, Varghese DS, Gillam MC, Peyton M, Modi B, Schiltz RL, Girard L, Martinez ED (2012) Differential response of cancer cells to HDAC inhibitors trichostatin A and depsipeptide. Br J Cancer 106:116–125. https://doi.org/10.1038/bjc.2011.532
Chen B-F, Tsai M-C, Jow G-M (2006) Induction of calcium influx from extracellular fluid by beauvericin in human leukemia cells. Biochem Biophys Res Commun 340:134–139. https://doi.org/10.1016/j.bbrc.2005.11.166
Chiarini A, Whitfield JF, Pacchiana R, Marconi M, Armato U, Dal Prà I (2010) Calphostin C, a remarkable multimodal photodynamic killer of neoplastic cells by selective nuclear lamin B1 destruction and apoptogenesis (Review). Oncol Rep 23:887–892. https://doi.org/10.3892/or_00000711
Claeys L, Romano C, De Ruyck K, Wilson H, Fervers B, Korenjak M, Zavadil J, Gunter MJ, De Saeger S, De Boevre M, Huybrechts I (2020) Mycotoxin exposure and human cancer risk: a systematic review of epidemiological studies. Compr Rev Food Sci Food Saf 19:1449–1464. https://doi.org/10.1111/1541-4337.12567
De Saeger S, Logrieco A (2017) Report from the 1st MYCOKEY International Conference Global Mycotoxin Reduction in the Food and Feed Chain Held in Ghent, Belgium, 11–14 September 2017. Toxins (basel). https://doi.org/10.3390/toxins9090276
Dornetshuber R, Heffeter P, Kamyar M-R, Peterbauer T, Berger W, Lemmens-Gruber R (2007) Enniatin exerts p53-dependent cytostatic and p53-independent cytotoxic activities against human cancer cells. Chem Res Toxicol 20:465–473. https://doi.org/10.1021/tx600259t
EFSA Panel (2014) Scientific opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed. EFSA J 12:3802. https://doi.org/10.2903/j.efsa.2014.3802
Fung F, Clark RF (2004) Health effects of mycotoxins: a toxicological overview. J Toxicol Clin Toxicol 42:217–234. https://doi.org/10.1081/clt-120030947
Gammelsrud A, Solhaug A, Dendelé B, Sandberg WJ, Ivanova L, Kocbach Bølling A, Lagadic-Gossmann D, Refsnes M, Becher R, Eriksen G, Holme JA (2012) Enniatin B-induced cell death and inflammatory responses in RAW 267.4 murine macrophages. Toxicol Appl Pharmacol 261:74–87. https://doi.org/10.1016/j.taap.2012.03.014
Girke T, Cheng L-C, Raikhel N (2005) ChemMine. A compound mining database for chemical genomics. Plant Physiol 138:573–577. https://doi.org/10.1104/pp.105.062687
Glück J, Buhrke T, Frenzel F, Braeuning A, Lampen A (2018) In silico genotoxicity and carcinogenicity prediction for food-relevant secondary plant metabolites. Food Chem Toxicol 116:298–306. https://doi.org/10.1016/j.fct.2018.04.024
Hagiwara R, Kageyama K, Niioka K, Takayasu S, Tasso M, Daimon M (2021) Involvement of histone deacetylase 1/2 in adrenocorticotropic hormone synthesis and proliferation of corticotroph tumor AtT-20 cells. Peptides 136:170441. https://doi.org/10.1016/j.peptides.2020.170441
Honma M, Kitazawa A, Cayley A, Williams RV, Barber C, Hanser T, Saiakhov R, Chakravarti S, Myatt GJ, Cross KP, Benfenati E, Raitano G, Mekenyan O, Petkov P, Bossa C, Benigni R, Battistelli CL, Giuliani A, Tcheremenskaia O, DeMeo C, Norinder U, Koga H, Jose C, Jeliazkova N, Kochev N, Paskaleva V, Yang C, Daga PR, Clark RD, Rathman J (2019) Improvement of quantitative structure-activity relationship (QSAR) tools for predicting Ames mutagenicity: outcomes of the Ames/QSAR international challenge project. Mutagenesis 34:3–16. https://doi.org/10.1093/mutage/gey031
Ivanova L, Egge-Jacobsen WM, Solhaug A, Thoen E, Fæste CK (2012) Lysosomes as a possible target of enniatin B-induced toxicity in Caco-2 cells. Chem Res Toxicol 25:1662–1674. https://doi.org/10.1021/tx300114x
Jaderson M, Park J-H (2020) Effect of storage temperature and duration on concentrations of 27 fungal secondary metabolites spiked into floor dust from an office building. J Occup Environ Hyg 17:220–230. https://doi.org/10.1080/15459624.2020.1734205
Jiang Z, Kong C, Zhang Z, Zhu Y, Zhang Y, Chen X (2015) Reduction of protein kinase C α (PKC-α) promote apoptosis via down-regulation of Dicer in bladder cancer. J Cell Mol Med 19:1085–1093. https://doi.org/10.1111/jcmm.12503
Jornod F, Rugard M, Tamisier L, Coumoul X, Andersen HR, Barouki R, Audouze K (2020) AOP4EUpest: mapping of pesticides in adverse outcome pathways using a text mining tool. Bioinformatics 36:4379–4381. https://doi.org/10.1093/bioinformatics/btaa545
Juan-García A, Tolosa J, Juan C, Ruiz M-J (2019) Cytotoxicity, genotoxicity and disturbance of cell cycle in HepG2 cells exposed to OTA and BEA: single and combined actions. Toxins (basel). https://doi.org/10.3390/toxins11060341
Kachuri L, Harris MA, MacLeod JS, Tjepkema M, Peters PA, Demers PA (2017) Cancer risks in a population-based study of 70,570 agricultural workers: results from the Canadian census health and environment cohort (CanCHEC). BMC Cancer 17:343. https://doi.org/10.1186/s12885-017-3346-x
Kalfalah FM, Mielke C, Christensen MO, Baechler S, Marko D, Boege F (2011) Genotoxicity of dietary, environmental and therapeutic topoisomerase II poisons is uniformly correlated to prolongation of enzyme DNA residence. Mol Nutr Food Res 55(Suppl 1):S127-142. https://doi.org/10.1002/mnfr.201000509
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30. https://doi.org/10.1093/nar/28.1.27
Kanehisa M, Furumichi M, Sato Y, Ishiguro-Watanabe M, Tanabe M (2021) KEGG: integrating viruses and cellular organisms. Nucleic Acids Res 49:D545–D551. https://doi.org/10.1093/nar/gkaa970
Kearney GD, Arcury TA, Quandt SA, Talton JW, Arnold TJ, Sandberg JC, Wiggins MF, Daniel SS (2020) Respiratory health and suspected asthma among hired Latinx child farmworkers in rural North Carolina. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph17217939
Kouri K, Duchen MR, Lemmens-Gruber R (2005) Effects of beauvericin on the metabolic state and ionic homeostasis of ventricular myocytes of the guinea pig. Chem Res Toxicol 18:1661–1668. https://doi.org/10.1021/tx050096g
Kovalsky-Paris MP, Liu Y-J, Nahrer K, Binder EM (2015) Climate change impacts on mycotoxin production. Climate change and mycotoxins. De Gruyter, Berlin
Kubicek S, Gilbert JC, Fomina-Yadlin D, Gitlin AD, Yuan Y, Wagner FF, Holson EB, Luo T, Lewis TA, Taylor B, Gupta S, Shamji AF, Wagner BK, Clemons PA, Schreiber SL (2012) Chromatin-targeting small molecules cause class-specific transcriptional changes in pancreatic endocrine cells. Proc Natl Acad Sci U S A 109:5364–5369. https://doi.org/10.1073/pnas.1201079109
Liao Y, Wang J, Jaehnig EJ, Shi Z, Zhang B (2019) WebGestalt 2019: gene set analysis toolkit with revamped UIs and APIs. Nucleic Acids Res 47:W199–W205. https://doi.org/10.1093/nar/gkz401
Lin H-I, Lee Y-J, Chen B-F, Tsai M-C, Lu J-L, Chou C-J, Jow G-M (2005) Involvement of Bcl-2 family, cytochrome c and caspase 3 in induction of apoptosis by beauvericin in human non-small cell lung cancer cells. Cancer Lett 230:248–259. https://doi.org/10.1016/j.canlet.2004.12.044
Lu C-L, Lin H-I, Chen B-F, Jow G-M (2016) Beauvericin-induced cell apoptosis through the mitogen-activated protein kinase pathway in human nonsmall cell lung cancer A549 cells. J Toxicol Sci 41:429–437. https://doi.org/10.2131/jts.41.429
Madden JC, Enoch SJ, Paini A, Cronin MTD (2020) A Review of in silico tools as alternatives to animal testing: principles, resources and applications. Altern Lab Anim 48:146–172. https://doi.org/10.1177/0261192920965977
Madia F, Kirkland D, Morita T, White P, Asturiol D, Corvi R (2020) EURL ECVAM genotoxicity and carcinogenicity database of substances eliciting negative results in the Ames test: construction of the database. Mutat Res 854–855:503199. https://doi.org/10.1016/j.mrgentox.2020.503199
Maunz A, Gütlein M, Rautenberg M, Vorgrimmler D, Gebele D, Helma C (2013) lazar: a modular predictive toxicology framework. Front Pharmacol 4:38. https://doi.org/10.3389/fphar.2013.00038
Ndaw S, Jargot D, Antoine G, Denis F, Melin S, Robert A (2021) Investigating multi-mycotoxin exposure in occupational settings: a biomonitoring and airborne measurement approach. Toxins (basel). https://doi.org/10.3390/toxins13010054
Nielsen KF, Smedsgaard J (2003) Fungal metabolite screening: database of 474 mycotoxins and fungal metabolites for dereplication by standardised liquid chromatography-UV-mass spectrometry methodology. J Chromatogr A 1002:111–136. https://doi.org/10.1016/s0021-9673(03)00490-4
Ojcius DM, Zychlinsky A, Zheng LM, Young JD (1991) Ionophore-induced apoptosis: role of DNA fragmentation and calcium fluxes. Exp Cell Res 197:43–49. https://doi.org/10.1016/0014-4827(91)90477-c
Olivo M, Ali-Seyed M (2007) Apoptosis signalling mechanisms in human cancer cells induced by Calphostin-PDT. Int J Oncol 30:537–548
Park H, Im JY, Kim J, Choi WS, Kim HS (2008) Effects of apicidin, a histone deacetylase inhibitor, on the regulation of apoptosis in H-ras-transformed breast epithelial cells. Int J Mol Med 21:325–333
Pócsi I, Giacometti F, Ambrus Á, Logrieco AF (2020) Editorial: Aspergillus-derived mycotoxins in the feed and food chain. Front Microbiol 11:606108. https://doi.org/10.3389/fmicb.2020.606108
Poli G, Di Fabio R, Ferrante L, Summa V, Botta M (2017) Largazole analogues as histone deacetylase inhibitors and anticancer agents: an overview of structure-activity relationships. ChemMedChem 12:1917–1926. https://doi.org/10.1002/cmdc.201700563
Prosperini A, Juan-García A, Font G, Ruiz MJ (2013) Reactive oxygen species involvement in apoptosis and mitochondrial damage in Caco-2 cells induced by enniatins A, A1, B and B1. Toxicol Lett 222:36–44. https://doi.org/10.1016/j.toxlet.2013.07.009
Rugard M, Coumoul X, Carvaillo J-C, Barouki R, Audouze K (2020) Deciphering adverse outcome pathway network linked to bisphenol F using text mining and systems toxicology approaches. Toxicol Sci 173:32–40. https://doi.org/10.1093/toxsci/kfz214
Sauvé J-F, Locke SJ, Josse PR, Stapleton EM, Metwali N, Altmaier RW, Andreotti G, Thorne PS, Hofmann JN, Beane Freeman LE, Friesen MC (2020) Characterization of inhalable endotoxin, glucan, and dust exposures in Iowa farmers. Int J Hyg Environ Health 228:113525. https://doi.org/10.1016/j.ijheh.2020.113525
Shephard GS, Burger H-M, Gambacorta L, Gong YY, Krska R, Rheeder JP, Solfrizzo M, Srey C, Sulyok M, Visconti A, Warth B, van der Westhuizen L (2013) Multiple mycotoxin exposure determined by urinary biomarkers in rural subsistence farmers in the former Transkei, South Africa. Food Chem Toxicol 62:217–225. https://doi.org/10.1016/j.fct.2013.08.040
Sulyok M, Stadler D, Steiner D, Krska R (2020) Validation of an LC-MS/MS-based dilute-and-shoot approach for the quantification of > 500 mycotoxins and other secondary metabolites in food crops: challenges and solutions. Anal Bioanal Chem 412:2607–2620. https://doi.org/10.1007/s00216-020-02489-9
Tinwell H, Ashby J (1994) Genetic toxicity and potential carcinogenicity of taxol. Carcinogenesis 15:1499–1501. https://doi.org/10.1093/carcin/15.8.1499
Tolosa J, Barba FJ, Pallarés N, Ferrer E (2020) Mycotoxin identification and in silico toxicity assessment prediction in Atlantic Salmon. Mar Drugs 18:E629. https://doi.org/10.3390/md18120629
Tran VN, Viktorova J, Augustynkova K, Jelenova N, Dobiasova S, Rehorova K, Fenclova M, Stranska-Zachariasova M, Vitek L, Hajslova J, Ruml T (2020) In silico and in vitro studies of mycotoxins and their cocktails; their toxicity and its mitigation by silibinin pre-treatment. Toxins (basel) 12:E148. https://doi.org/10.3390/toxins12030148
Tual S, Lemarchand C, Boulanger M, Dalphin J-C, Rachet B, Marcotullio E, Velten M, Guizard A-V, Clin B, Baldi I, Lebailly P (2017) Exposure to farm animals and risk of lung cancer in the AGRICAN cohort. Am J Epidemiol 186:463–472. https://doi.org/10.1093/aje/kwx125
Ulrich S, Gottschalk C, Biermaier B, Bahlinger E, Twarużek M, Asmussen S, Schollenberger M, Valenta H, Ebel F, Dänicke S (2021) Occurrence of type A, B and D trichothecenes, zearalenone and stachybotrylactam in straw. Arch Anim Nutr 75:105–120. https://doi.org/10.1080/1745039X.2021.1877075
Vippila MR, Ly PK, Cuny GD (2015) Synthesis and antiproliferative activity evaluation of the disulfide-containing cyclic peptide thiochondrilline C and derivatives. J Nat Prod 78:2398–2404. https://doi.org/10.1021/acs.jnatprod.5b00428
Wang J, Duncan D, Shi Z, Zhang B (2013) WEB-based GEne SeT AnaLysis toolkit (WebGestalt): update 2013. Nucleic Acids Res 41:W77-83. https://doi.org/10.1093/nar/gkt439
Wätjen W, Debbab A, Hohlfeld A, Chovolou Y, Kampkötter A, Edrada RA, Ebel R, Hakiki A, Mosaddak M, Totzke F, Kubbutat MHG, Proksch P (2009) Enniatins A1, B and B1 from an endophytic strain of Fusarium tricinctum induce apoptotic cell death in H4IIE hepatoma cells accompanied by inhibition of ERK phosphorylation. Mol Nutr Food Res 53:431–440. https://doi.org/10.1002/mnfr.200700428
Wu C-C, Chen T-H, Liu B-L, Wu L-C, Chen Y-C, Tzeng Y-M, Hsu S-L (2013) Destruxin B Isolated from entomopathogenic fungus Metarhizium anisopliae induces apoptosis via a Bcl-2 family-dependent mitochondrial pathway in human nonsmall cell lung cancer cells. Evid Based Complement Alternat Med 2013:548929. https://doi.org/10.1155/2013/548929
Wu Q, Patocka J, Kuca K (2019) Beauvericin, a Fusarium mycotoxin: anticancer activity, mechanisms, and human exposure risk assessment. Mini Rev Med Chem 19:206–214. https://doi.org/10.2174/1389557518666180928161808
Xiong G, Wu Z, Yi J, Fu L, Yang Z, Hsieh C, Yin M, Zeng X, Wu C, Lu A, Chen X, Hou T, Cao D (2021) ADMETlab 2.0: an integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic Acids Res 49:W5–W14. https://doi.org/10.1093/nar/gkab255
Yoo EJ, Lee BM (2005) Comparative mutagenicity of apicidin and apicidin derivatives (SD-0203 and SD-2007), histone deacetylase inhibitors. J Toxicol Environ Health A 68:2097–2109. https://doi.org/10.1080/15287390500182511
Zeiger E (2019) The test that changed the world: the Ames test and the regulation of chemicals. Mutat Res/genet Toxicol Environ Mutagenesis 841:43–48. https://doi.org/10.1016/j.mrgentox.2019.05.007
Zhang J, Lai Z, Huang W, Ling H, Lin M, Tang S, Liu Y, Tao Y (2017) Apicidin inhibited proliferation and invasion and induced apoptosis via mitochondrial pathway in non-small cell lung cancer GLC-82 cells. Anticancer Agents Med Chem 17:1374–1382. https://doi.org/10.2174/1871520617666170419120044
Zhao G, Ronda E, Cea L, Pulido J, Barrio G, Regidor E (2019) Mortality by cause of death and risk behaviors in farmers versus non-farmers: the importance of avoiding the healthy worker effect. Int Arch Occup Environ Health 92:599–608. https://doi.org/10.1007/s00420-018-1396-2
Funding
This research was supported by the European Union Interreg program, Agritox project EAPA-998-2018, and by ANSES.
Author information
Authors and Affiliations
Contributions
DH: investigation, visualization, writing—original draft. DH and PL: formal analysis. DH, LMB and VF: writing—review & editing, project administration. DH and VF: conceptualization.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no-competing interests.
Ethical Approval
Not applicable.
Research Involving Human and Animal Participants
Not applicable.
Permission
Permission to use KEGG pathway map image was kindly granted by “Kanehisa Laboratories”. KEGG Copyright Permission Number: 220152.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Habauzit, D., Lemée, P., Botana, L.M. et al. Toxicity Predictions for Mycotoxins: A Combined In Silico Approach on Enniatin-Like Cluster. Expo Health 15, 315–331 (2023). https://doi.org/10.1007/s12403-022-00492-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12403-022-00492-2