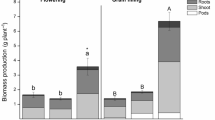
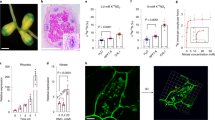
Abstract
Leguminous plants form root nodule organs with soil rhizobia bacteria, which can fix atmospheric nitrogen (N2) and supply ammonia to the host plant. It is established that the symbiotic N fixation efficiency is substantially influenced by plant nutrients, such as molybdenum (Mo), phosphorus (P), copper (Cu), and zinc (Zn). Potassium (K+) is the most abundant cation in plant cells; however, little evidence regarding the potential link between K nutrition and symbiotic N fixation efficiency is available to date. Here, we showed that K+ deficiency reduced the efficiency of symbiotic nitrogen fixation, and inoculated with rhizobia strain USDA110 could improve plant K+ acquisition. Furthermore, we identified a potassium transporter gene (GmHAK5) that was highly expressed in the root steles and in the vasculature cells of nodules. The GmHAK5 protein was localized at the plasma membrane and could rescue the growth phenotype of yeast K+ uptake-defective mutant. The results obtained from this study provides new insights for the understanding of the potential role of K+ nutrition in modulating symbiotic N fixation in soybean.








Similar content being viewed by others
References
Abreu I, Saez A, Castro-Rodriguez R, Escudero V, Rodriguez-Haas B, Senovilla M, Larue C, Grolimund D, Tejada-Jimenez M, Imperial J, Gonzalez-Guerrero M (2017) Medicago truncatula zinc–iron permease6 provides zinc to rhizobia-infected nodule cells. Plant Cell Environ 40(11):2706–2719. https://doi.org/10.1111/pce.13035
Ahn SJ, Shin R, Schachtman DP (2004) Expression of KT/KUP genes in Arabidopsis and the role of root hairs in K+ uptake. Plant Physiol 134(3):1135–1145. https://doi.org/10.1104/pp.103.034660
Amrutha RN, Sekhar PN, Varshney RK, Kishor PB (2007) Genome-wide analysis and identification of genes related to potassium transporter families in rice (Oryza sativa L.). Plant Sci 172:708–721
Bernstein JD, Okamoto Y, Kim M, Shikano S (2013) Potential use of potassium efflux-deficient yeast for studying trafficking signals and potassium channel functions. FEBS Open Bio 3:196–203. https://doi.org/10.1016/j.fob.2013.04.002
Blevins DG, Barnett NM, Frost WB (1978) Role of potassium and malate in nitrate uptake and translocation by wheat seedlings. Plant Physiol 62(5):784–788. https://doi.org/10.1104/pp.62.5.784
Cakmak IHC, Marschner H (1994) Partitioning of shoot and root dry matter and carbohydrates in bean plants suffering from phosphorus, potassium and magnesium deficiency. J Exp Bot 45(9):1245–1250. https://doi.org/10.1093/jxb/45.9.1245
Chaudhary MI, Adu-Gyamfi JJ, Saneoka H, Nguyen NT, Suwa R, Kanai S, El-Shemy HA, Lightfoot DA, Fujita K (2008) The effect of phosphorus deficiency on nutrient uptake, nitrogen fixation and photosynthetic rate in mashbean, mungbean and soybean. Acta Physiol Plant 30(4):537–544. https://doi.org/10.1007/s11738-008-0152-8
Chen L, Liao H (2017) Engineering crop nutrient efficiency for sustainable agriculture. J Integr Plant Biol 59(10):710–735. https://doi.org/10.1111/jipb.12559
Chen G, Hu Q, Luo L, Yang T, Zhang S, Hu Y, Yu L, Xu G (2015) Rice potassium transporter OsHAK1 is essential for maintaining potassium-mediated growth and functions in salt tolerance over low and high potassium concentration ranges. Plant Cell Environ 38(12):2747–2765. https://doi.org/10.1111/pce.12585
Chen L, Qin L, Zhou L, Li X, Chen Z, Sun L, Wang W, Lin Z, Zhao J, Yamaji N, Ma JF, Gu M, Xu G, Liao H (2019) A nodule-localized phosphate transporter GmPT7 plays an important role in enhancing symbiotic N2 fixation and yield in soybean. New Phytol 221(4):2013–2025. https://doi.org/10.1111/nph.15541
Chen C, Chen H, Zhang Y, Thomas HR, Frank MH, He Y, Xia R (2020) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13(8):1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Clemente TE, Cahoon EB (2009) Soybean oil: genetic approaches for modification of functionality and total content. Plant Physiol 151(3):1030–1040. https://doi.org/10.1104/pp.109.146282
Desbrosses GKC, Ott T, Udvardi MK (2004) Lotus japonicus LjKUP is induced late during nodule development and encodes a potassium transporter of the plasma membrane. Mol Plant Microbe Interact 17(7):789–797. https://doi.org/10.1094/MPMI.2004.17.7.789
Downie JA (2014) Legume nodulation. Curr Biol 24(5):R184-190. https://doi.org/10.1016/j.cub.2014.01.028
Drain A, Thouin J, Wang L, Boeglin M, Pauly N, Nieves-Cordones M, Gaillard I, Very AA, Sentenac H (2020) Functional characterization and physiological roles of the single Shaker outward K(+) channel in Medicago truncatula. Plant J 102(6):1249–1265. https://doi.org/10.1111/tpj.14697
Fedorova EE, Coba de la Pena T, Lara-Dampier V, Trifonova NA, Kulikova O, Pueyo JJ, Lucas MM (2021) Potassium content diminishes in infected cells of Medicago truncatula nodules due to the mislocation of channels MtAKT1 and MtSKOR/GORK. J Exp Bot 72(4):1336–1348. https://doi.org/10.1093/jxb/eraa508
Gierth M, Maser P, Schroeder JI (2005) The potassium transporter AtHAK5 functions in K(+) deprivation-induced high-affinity K(+) uptake and AKT1 K(+) channel contribution to K(+) uptake kinetics in Arabidopsis roots. Plant Physiol 137(3):1105–1114. https://doi.org/10.1104/pp.104.057216
Gil-Diez P, Tejada-Jimenez M, Leon-Mediavilla J, Wen J, Mysore KS, Imperial J, Gonzalez-Guerrero M (2019) MtMOT1.2 is responsible for molybdate supply to Medicago truncatula nodules. Plant Cell Environ 42(1):310–320. https://doi.org/10.1111/pce.13388
Gonzalez-Guerrero M, Matthiadis A, Saez A, Long TA (2014) Fixating on metals: new insights into the role of metals in nodulation and symbiotic nitrogen fixation. Front Plant Sci 5:45. https://doi.org/10.3389/fpls.2014.00045
Gupta M, Qiu X, Wang L, Xie W, Zhang C, Xiong L, Lian X, Zhang Q (2008) KT/HAK/KUP potassium transporters gene family and their whole-life cycle expression profile in rice (Oryza sativa). Mol Genet Genom 280(5):437. https://doi.org/10.1007/s00438-008-0377-7
Han M, Wu W, Wu WH, Wang Y (2016) Potassium transporter KUP7 is involved in K(+) acquisition and translocation in arabidopsis root under K(+)-limited conditions. Mol Plant 9(3):437–446. https://doi.org/10.1016/j.molp.2016.01.012
Hardy RWHR, Jackson EK, Burns RC (1968) The acetylene-ethylene assay for N2 fixation: laboratory and field evaluation. Plant Physiol 43(8):1185–1207. https://doi.org/10.1104/pp.43.8.1185
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci 11(12):610–617. https://doi.org/10.1016/j.tplants.2006.10.007
Hirsch RE, Lewis BD, Spalding EP, Sussman MR (1998) A role for the AKT1 potassium channel in plant nutrition. Science 280(5365):918–921. https://doi.org/10.1126/science.280.5365.918
Horie T, Brodsky DE, Costa A, Kaneko T, Lo Schiavo F, Katsuhara M, Schroeder JI (2011) K+ transport by the OsHKT2;4 transporter from rice with atypical Na+ transport properties and competition in permeation of K+ over Mg2+ and Ca2+ ions. Plant Physiol 156(3):1493–1507. https://doi.org/10.1104/pp.110.168047
Kanai S, Ohkura K, Adu-Gyamfi JJ, Mohapatra PK, Nguyen NT, Saneoka H, Fujita K (2007) Depression of sink activity precedes the inhibition of biomass production in tomato plants subjected to potassium deficiency stress. J Exp Bot 58(11):2917–2928. https://doi.org/10.1093/jxb/erm149
Karandashov V, Nagy R, Wegmuller S, Amrhein N, Bucher M (2004) Evolutionary conservation of a phosphate transporter in the arbuscular mycorrhizal symbiosis. Proc Natl Acad Sci USA 101(16):6285–6290. https://doi.org/10.1073/pnas.0306074101
Kereszt A, Li D, Indrasumunar A, Nguyen CD, Nontachaiyapoom S, Kinkema M, Gresshoff PM (2007) Agrobacterium rhizogenes-mediated transformation of soybean to study root biology. Nat Protoc 2(4):948–952. https://doi.org/10.1038/nprot.2007.141
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Kunz HH, Gierth M, Herdean A, Satoh-Cruz M, Kramer DM, Spetea C, Schroeder JI (2014) Plastidial transporters KEA1, -2, and -3 are essential for chloroplast osmoregulation, integrity, and pH regulation in Arabidopsis. Proc Natl Acad Sci USA 111(20):7480–7485. https://doi.org/10.1073/pnas.1323899111
Lebaudy A, Very AA, Sentenac H (2007) K+ channel activity in plants: genes, regulations and functions. FEBS Lett 581(12):2357–2366. https://doi.org/10.1016/j.febslet.2007.03.058
Leigh RA, Wyn Jones RG (1984) A hypothesis relating critical potassium concentrations for growth to the distribution and functions of this ion in the plant cell. New Phytol 97:1–13
Li J, Long Y, Qi GN, Li J, Xu ZJ, Wu WH, Wang Y (2014) The Os-AKT1 channel is critical for K+ uptake in rice roots and is modulated by the rice CBL1-CIPK23 complex. Plant Cell 26(8):3387–3402. https://doi.org/10.1105/tpc.114.123455
Li W, Xu G, Alli A, Yu L (2018) Plant HAK/KUP/KT K(+) transporters: function and regulation. Semin Cell Dev Biol 74:133–141. https://doi.org/10.1016/j.semcdb.2017.07.009
Li R, Feng Y, Chen H, Zhang C, Huang Y, Chen L, Hao Q, Cao D, Yuan S, Zhou X (2020) Whole-genome sequencing of Bradyrhizobium diazoefficiens 113-2 and comparative genomic analysis provide molecular insights into species specificity and host specificity. Front Microbiol 11:576800. https://doi.org/10.3389/fmicb.2020.576800
Limpens E, Ivanov S, van Esse W, Voets G, Fedorova E, Bisseling T (2009) Medicago N2-fixing symbiosomes acquire the endocytic identity marker Rab7 but delay the acquisition of vacuolar identity. Plant Cell 21(9):2811–2828. https://doi.org/10.1105/tpc.108.064410
Liu J, Liu J, Liu J, Cui M, Huang Y, Tian Y, Chen A, Xu G (2019) The potassium transporter SlHAK10 is involved in mycorrhizal potassium uptake. Plant Physiol 180(1):465–479. https://doi.org/10.1104/pp.18.01533
Luan M, Tang RJ, Tang Y, Tian W, Hou C, Zhao F, Lan W, Luan S (2017) Transport and homeostasis of potassium and phosphate: limiting factors for sustainable crop production. J Exp Bot 68(12):3091–3105. https://doi.org/10.1093/jxb/erw444
Lynd JQ, Odell GV, McNew RW (2008) Soil potassium effects on nitrogenase activity with associated nodule components of hairy vetch at anthesis. J Plant Nutr 4(3):303–318. https://doi.org/10.1080/01904168109362920
Maathuis FJ (2009) Physiological functions of mineral macronutrients. Curr Opin Plant Biol 12(3):250–258. https://doi.org/10.1016/j.pbi.2009.04.003
Mengel K, Haghparast MR, Koch K (1974) The effect of potassium on the fixation of molecular nitrogen by root nodules of Vicia faba. Plant Physiol 54(4):535–538. https://doi.org/10.1104/pp.54.4.535
Murray JD, Liu CW, Chen Y, Miller AJ (2017) Nitrogen sensing in legumes. J Exp Bot 68(8):1919–1926. https://doi.org/10.1093/jxb/erw405
Nieves-Cordones M, Lara A, Rodenas R, Amo J, Rivero RM, Martinez V, Rubio F (2019) Modulation of K(+) translocation by AKT1 and AtHAK5 in Arabidopsis plants. Plant Cell Environ 42(8):2357–2371. https://doi.org/10.1111/pce.13573
Pettigrew WT (2008) Potassium influences on yield and quality production for maize, wheat, soybean and cotton. Physiol Plant 133(4):670–681. https://doi.org/10.1111/j.1399-3054.2008.01073.x
Premaratne KP, Oertli JJ (1994) The influence of potassium supply on nodulation, nitrogenase activity and nitrogen accumulation of soybean (Glycine max L. Merrill) grown in nutrient solution. Fertil Res 38:95–99
Qi Z, Hampton CR, Shin R, Barkla BJ, White PJ, Schachtman DP (2008) The high affinity K+ transporter AtHAK5 plays a physiological role in planta at very low K+ concentrations and provides a caesium uptake pathway in Arabidopsis. J Exp Bot 59(3):595–607. https://doi.org/10.1093/jxb/erm330
Qin L, Zhao J, Tian J, Chen L, Sun Z, Guo Y, Lu X, Gu M, Xu G, Liao H (2012) The high-affinity phosphate transporter GmPT5 regulates phosphate transport to nodules and nodulation in soybean. Plant Physiol 159(4):1634–1643. https://doi.org/10.1104/pp.112.199786
Rehman HM, Nawaz MA, Shah ZH, Daur I, Khatoon S, Yang SH, Chung G (2017) In-depth genomic and transcriptomic analysis of five K(+) transporter gene families in soybean confirm their differential expression for nodulation. Front Plant Sci 8:804. https://doi.org/10.3389/fpls.2017.00804
Rosolem CA, Nakagawa JJNCiA (2001) Residual and annual potassic fertilization for soybeans. 59(2):143–149
Senovilla M, Castro-Rodriguez R, Abreu I, Escudero V, Kryvoruchko I, Udvardi MK, Imperial J, Gonzalez-Guerrero M (2018) Medicago truncatula copper transporter 1 (MtCOPT1) delivers copper for symbiotic nitrogen fixation. New Phytol 218(2):696–709. https://doi.org/10.1111/nph.14992
Shabala S, Shabala L (2011) Ion transport and osmotic adjustment in plants and bacteria. Biomol Concepts 2(5):407–419. https://doi.org/10.1515/BMC.2011.032
Tejada-Jimenez M, Gil-Diez P, Leon-Mediavilla J, Wen J, Mysore KS, Imperial J, Gonzalez-Guerrero M (2017) Medicago truncatula molybdate transporter type 1 (MtMOT1.3) is a plasma membrane molybdenum transporter required for nitrogenase activity in root nodules under molybdenum deficiency. New Phytol 216(4):1223–1235. https://doi.org/10.1111/nph.14739
Thomas RJ, Hungria M (1988) Effect of potassium on nitrogen fixation, nitrogen transport, and nitrogen harvest index of bean. J Plant Nutr 11(2):175–188. https://doi.org/10.1080/01904168809363794
Toth K, Batek J, Stacey G (2016) Generation of soybean (Glycine max) transient transgenic roots. Curr Protoc Plant Biol 1(1):1–13. https://doi.org/10.1002/cppb.20017
Udvardi MK, Day DA (1997) Metabolite transport across symbiotic membranes of legume nodules. Annu Rev Plant Physiol Plant Mol Biol 48:493–523. https://doi.org/10.1146/annurev.arplant.48.1.493
Udvardi M, Poole PS (2013) Transport and metabolism in legume-rhizobia symbioses. Annu Rev Plant Biol 64:781–805. https://doi.org/10.1146/annurev-arplant-050312-120235
Valkov VT, Rogato A, Alves LM, Sol S, Noguero M, Leran S, Lacombe B, Chiurazzi M (2017) The nitrate transporter family protein LjNPF8.6 controls the N-fixing nodule activity. Plant Physiol 175(3):1269–1282. https://doi.org/10.1104/pp.17.01187
Vance CP (2001) Symbiotic nitrogen fixation and phosphorus acquisition. Plant nutrition in a world of declining renewable resources. Plant Physiol 127(2):390–397
Walker DJ, Leigh RA, Miller AJ (1996) Potassium homeostasis in vacuolate plant cells. Proc Natl Acad Sci USA 93(19):10510–10514. https://doi.org/10.1073/pnas.93.19.10510
Wang Y, Wu WH (2013) Potassium transport and signaling in higher plants. Annu Rev Plant Biol 64:451–476. https://doi.org/10.1146/annurev-arplant-050312-120153
Wei J, Zheng Y, Feng H, Qu H, Fan X, Yamaji N, Ma JF, Xu G (2018) OsNRT2.4 encodes a dual-affinity nitrate transporter and functions in nitrate-regulated root growth and nitrate distribution in rice. J Exp Bot 69(5):1095–1107. https://doi.org/10.1093/jxb/erx486
Yang Z, Gao Q, Sun C, Li W, Gu S, Xu C (2009) Molecular evolution and functional divergence of HAK potassium transporter gene family in rice (Oryza sativaL.). J Genet Genomics 36:161–172
Yang T, Zhang S, Hu Y, Wu F, Hu Q, Chen G, Cai J, Wu T, Moran N, Yu L, Xu G (2014) The role of a potassium transporter OsHAK5 in potassium acquisition and transport from roots to shoots in rice at low potassium supply levels. Plant Physiol 166(2):945–959. https://doi.org/10.1104/pp.114.246520
Yang T, Feng H, Zhang S, Xiao H, Hu Q, Chen G, Xuan W, Moran N, Murphy A, Yu L, Xu G (2020) The potassium transporter OsHAK5 alters rice architecture via ATP-dependent transmembrane auxin fluxes. Plant Commun 1(5):100052. https://doi.org/10.1016/j.xplc.2020.100052
Funding
This work was supported by grants from the National Natural Science Foundation of China, Young Scientists Fund (32002123, 42007120). University Natural Science Research Project of Anhui Province (KJ2021A0892). Special fund for scientific innovation strategy-construction of high-level Academy of Guangdong academy of agriculture science (R2017YJ-YB1001).
Author information
Authors and Affiliations
Contributions
JJL, JHL and AQC planned and designed the research; JJL, JHL and MMC performed the experiments; JJL, JLL and XC analyzed the data; JJL wrote the manuscript. GHX and AQC revised to the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, J., Liu, J., Cui, M. et al. Investigate the Effect of Potassium on Nodule Symbiosis and Uncover an HAK/KUP/KT Member, GmHAK5, Strongly Responsive to Root Nodulation in Soybean. J. Plant Biol. 65, 459–471 (2022). https://doi.org/10.1007/s12374-022-09364-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-022-09364-3