Abstract
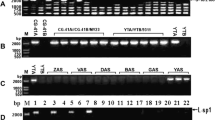
A cytoplasmic male sterile line (designated as M2BS) was obtained from an indica rice maintainer M2B induced by partial-length HcPDIL5-2a (Hibiscus cannabinus protein disulfide isomerase-like) transformation. The anther of M2BS was short, slender, hygrophanous, and fissured. I2-KI staining method showed that there was typical and spherical abortion in pollen grains. M2BS was found abortive at middle and late stage of monocyte by the modified carbol fuchsin stained observation and paraffin section observation. The tapetum was observed pre-degenerated in M2BS. Hereditary analysis indicated that the male sterility of M2BS was a maternally inherited inability after six backcross generations with M2B and the combinations of M2BS hybridized with other two male fertile materials. The M2BS could be affirmed a cytoplasmic male sterile (CMS) type. Moreover, it was a transgenic plant confirmed by PCR, Southern blot and RT-PCR detection. M2BS could be distinguished from M2B and its CMS line M2A by RFLP analysis. The overall mitochondrial genome sequencing results showed, that in M2BS, the main differences of mitochondrial gene sequence were located in nad4, nad5, nad7, orf194 and intergenic region, relatively to those of M2A. The obtained results indicate that M2BS is a novel cytoplasmic male sterile line.
Similar content being viewed by others
References
Beckett JB (1971) Classification of male sterile cytoplasms in maize(Zea mays L.). Crop Sci 11:724–726
Cai YM (2014) Construction of MSH1 RNAi expression vector and its transformation to Brassica Juncea. Master. thesis. Southwest University, Chongqing
Chase CD (2006) Cytoplasmic male sterility: a window to the world of plant mitochondrial-nuclear interactions. Trends Genet 23:81–90
Cooper GM, Nickerson DA, Eichler EE (2007) Mutational and selective effects on copy-number variants in the human genome. Nat Genet 39:S22–S29
Doyle JJ, Doyle JL (1990) A rapid total DNA preparation procedure for fresh plant tissue. Focus 12:13–15
Fujii S, Toriyama K (2009) Suppressed expression of retrograderegulated male sterility restores pollen fertility in cytoplasmic male sterile rice plants. Proc Natl Acad Sci USA 106:9513–9518
Hanson MR (1991) Plant mitochongdrial mutations and sterility. Annu Rev Genet 25:461–486
Hanson MR, Bentolila S (2004) Interactions of mitochondrial and nuclear genes that affect male gametophyte development. Plant Cell 16:S154–169
He S, Abad AR, Gelvin SB, Mackenzie SA (1996) A cytoplasmic male sterility-associated mitochondrial protein causes pollen disruption in transgenic tobacco. Proc Natl Acad Sci USA 93:11763–11768
Jin G (2011) Discovery of SNP markers associated with CMS cytoplasm and male sterility-related genes in Kenaf (Hibiscus Cannabinus L.). Ph.D. thesis. GuangXi University, Nanning
Ji JL, Sheng CZ,Shi M, An CJ, Wu XF, Li DS, Du RQ (2002) A study on the transformation of wheat with salt-tolerant gene HVA1 by method deflating pollen tube. J.Triticeae Crops 22:10–13
Kim DH, Kang JG, Kim B (2007) Isolation and characterization of the cytoplasmic male sterility associated orf456 gene of chili pepper (Capsicum annuum L.). Plant Mol Biol 63:519–532
Li G (2008) Differential analysis of mitochondrial proteome and discovery of sterile genes in the cytoplasmic male sterility of Kenaf (Hibiscus Cannabinus L.). Ph.D. thesis. GuangXi University, Nanning
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–60
Li XQ, Wan BH, Liu JL, Li JS, Xu SZ, Zheng YL (2004) Classification of male sterile cytoplasms of WBMs in maize (Zea mays L.). Acta Agron.Sin 30:304–307
Li YH, Xiao XG, Zhao GR, Nie XL, Zhang AM, Yan LF (1999) Preliminary study of transferring new engineered male sterile gene into cultivated wheat. J. Agric.Biotech 7:255–258
Liu DM (2014) Innovation of male sterile germplasm and identification of male sterile related genes in cotton. Ph.D. thesis. GuangXi University, Nanning
Mariani C, Beuckeleer MD, Truettner J, Leemans J, Goldberg RB (1990) Induction of male sterility in plants by a chimaeric ribonucleases gene. Nature 347:737–741
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, DePristo MA (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303
Ondzighi CA, Christopher DA, Cho EJ, Chang SC, Staehelin LA (2008) Arabidopsis protein disulfide isomerase-5 inhibits cysteine proteases during trafficking to vacuoles before programmed cell death of the endothelium in developing seeds. Plant Cell 20:2205–2220
Rosellini D, Pezzotti M, Veronesi F (2001) Characterization of transgenic male sterility in alfalfa. Euphytica 118:313–319
Sandhu APS, Abdelnoor RV, Mackenzie SA (2007) Transgenic induction of mitochondrial rearrangements for cytoplasmic male sterility in crop plants. PNAS 104:1766–1770
Schnable PS, Wise RP (1998) The molecular basis of cytoplasmic male sterility and fertility restoration. Trends Plant Sci 3:175–180
Sun RF, Fang ZY, Zhang SJ, Li F, Niu XK, Wu FY (2000) Biochemical analysis of cold-tolerant Ogura CMS chinese cabbage lines. Acta Hortic.Sin 27:187–192
van der Meer IM, Stam ME, van Tunen AJ, Mol JN, Stuitje AR (1992) Antisense inhibition of flavonoid biosynthesis in petunia anthers results in male sterility. Plant Cell 4:253–262
Wang K, Gao F, Ji YX, Liu Y, Dan ZW, Yang PF, Zhu YG, Li SQ (2013) ORFH79 impairs mitochondrial function via interaction with a subunit of electron transport chain complex III in Honglian cytoplasmic male sterile rice. New Phytol 198:408–418
Wang LS, Li YZ, Ma XY (2006) Breeding of plant male sterile line and hybrid seed production techniques. J. Anhui Agric Sci 34:2362–2363
Wang Q, Yang HP, Zou M, Song HY (2013) Procurement of transgenic male sterile mustard plants induced by TA29-Barnase transformation. China Veget 4:26–31
Yang JH, Liu XY, Yang XD, Zhang MF (2010) Mitochondriallytargeted expression of a cytoplasmic male sterility-associated orf220 gene causes male sterility in Brassica juncea. BMC Plant Bio 10:231
Yu PT, Wang W, He YK, Shen RJ (2000) Transformation of Barnase gene in chinese cabbage. Acta Agric. Shanghai 16:17–19
Yuan LP (1997) Current status and developing prospects in two-line hybrid rice research in China. Reas.Agric Modern 18:1–3
Yuen CYL, Wong K, Christopher DA (2015) Phylogenetic characterization and promoter expression analysis of a novel hybrid protein disulfide isomerase/cargo receptor subfamily unique to plants and chromalveolates. Mol Genet Genomics 291:455–469
Zhang H, Wang B, Xue AQ, Cao J, Li BJ, Tan ZP, Huang WR (1998) The construction and tomato transformation of male sterile chimeric gene. HEREDITAS 20:5–7
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Tang, D., Chen, P., Jin, G. et al. Identification of a novel cytoplasmic male sterile line M2BS induced by partial-length HcPDIL5-2a transformation in rice (Oryza sativa L.). J. Plant Biol. 60, 146–153 (2017). https://doi.org/10.1007/s12374-016-0371-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-016-0371-2