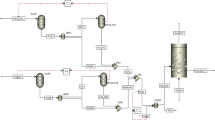
Abstract
Thermal balance of steam boilers can be done by two ways, the direct or indirect method; the direct method is not accurate. The reason why it is not accurate is that some operating parameters of biomass boilers cannot be obtained by direct measurements, like the measurement of fuel flow. These facts make it difficult to apply the direct method in the heat balance and force to use indirect one to determine these parameters and the boiler efficiency; hence, the indirect method is generally used for heat balance. Indirect method provides more accurate values and additionally, quantifies each of the energy losses, allowing to determinate causes of low efficiency of the equipment. Large amount of data involved implies the use of more affective calculation means; being iterative, assumed values for fuel flow must be compared with a value calculated until both match. In this work, the indirect method has been programmed in MATLAB; and for the preliminary fuel flow evaluation an optimization method could be used to prevent having to assume a value. Calculation details and a MATLAB algorithm are also presented.

Similar content being viewed by others
Abbreviations
- α :
-
Excess air coefficient
- β :
-
Bunte coefficient (β = 22.413e−3 m3/mol for sugarcane bagasse)
- η :
-
Gross efficiency of the steam generator (%)
- a arr :
-
Fraction of inorganic material in the dragged ashes
- a cen :
-
Fraction of inorganic material in the ashes removed by the scrubber
- a esc :
-
Fraction of inorganic material in the slag
- A arr :
-
Inorganic material in the dragged ashes (%)
- Aesc :
-
Inorganic material in the ashes removed by the scrubber (%)
- Acen :
-
Inorganic material in the slag (%)
- At :
-
Ash according the elemental analysis of fuel on working moisture (%)
- \({\text{A}}_{\text{dry}}^{t}\) :
-
Ash according the elemental analysis of fuel on laboratory moisture (%)
- c :
-
Specific heat of the fuel (kJ/kg K)
- c esc :
-
Specific heat of the slag (kJ/kg K)
- Ct :
-
Carbon according the elemental analysis of fuel on working moisture (%)
- Carr :
-
Carbon content in the dragged ashes (%)
- Ccen :
-
Carbon content in the ashes removed by the scrubber (%)
- \({\text{C}}_{\text{dry}}^{t}\) :
-
Carbon according the elemental analysis of fuel on laboratory moisture (%)
- Cesc :
-
Carbon content in the slag (%)
- CO:
-
Carbon monoxide on a dry basis in the combustion products (%)
- \({\text{CO}}_{2}^{\hbox{max} }\) :
-
Maximum volume fraction of carbon dioxide
- CO2 :
-
Volume fraction of carbon dioxide in the combustion products
- h aa :
-
Enthalpy of feedwater (kJ/kg)
- \(h_{\text{air}}^{{T_{\text{g}} }}\) :
-
Air enthalpy in the temperature of the exhaust gases (kJ/m3)
- \(h_{\text{air}}^{o}\) :
-
Air enthalpy in the temperature of the exhaust gases (kJ/kg)
- \(h_{{_{\text{ash}} }}^{{T_{\text{g}} }}\) :
-
Ash enthalpy in the temperature of the exhaust gases (kJ/kg)
- \(h_{{{\text{CO}}_{2} }}^{{T_{\text{g}} }}\) :
-
CO2 enthalpy in the temperature of the exhaust gases (kJ/m3)
- \(h_{\text{g}}^{o}\) :
-
Enthalpy of the exhaust gas (stoichiometric quantity) (kJ/kg)
- h ge :
-
Enthalpy of the exhaust gases (kJ/kg)
- \(h_{{{\text{H}}_{2} {\text{O}}}}^{{T_{\text{g}} }}\) :
-
H2O enthalpy in the temperature of the exhaust gases (kJ/m3)
- \(h_{{{\text{N}}_{2} }}^{{T_{\text{g}} }}\) :
-
N2 enthalpy in the temperature of the exhaust gases (kJ/m3)
- h vs :
-
Enthalpy of superheated steam (kJ/kg)
- Ht :
-
Nitrogen according the elemental analysis of fuel on working moisture (%)
- \({\text{H}}_{\text{dry}}^{t}\) :
-
Nitrogen according the elemental analysis of fuel on laboratory moisture (%)
- k :
-
Conversion factor to working moisture
- LHV:
-
Lower heating value of fuel (kJ/kg)
- \(\dot{m}_{\text{esc}}\) :
-
Slag mass flow (kg/s)
- \(\dot{m}_{\text{cen}}\) :
-
Mass flow of dragged ashes (kg/s)
- \(\dot{m}_{\text{fuel}}\) :
-
Fuel mass flow (kg/s)
- \(\dot{m}_{\text{vs}}\) :
-
Mass flow of superheated steam (kg/s)
- \(\dot{m}_{{{\text{vs}} . {\text{nom}}}}\) :
-
Nominal mass flow of superheated steam (kg/s)
- Nt :
-
Nitrogen according the elemental analysis of fuel on working moisture (%)
- \({\text{N}}_{\text{dry}}^{t}\) :
-
Nitrogen according the elemental analysis of fuel on laboratory moisture (%)
- Ot :
-
Oxygen according the elemental analysis of fuel on working moisture (%)
- \({\text{O}}_{\text{dry}}^{t}\) :
-
Oxygen according the elemental analysis of fuel on laboratory moisture (%)
- O2 :
-
Volumetric fraction of oxygen in the combustion products
- p vs :
-
Superheated steam pressure (Pa)
- Q c :
-
Sensible heat of fuel (kJ/kg)
- \(Q_{\text{d}}^{t}\) :
-
Total heat available in the fuel (kJ/kg)
- Q u :
-
Useful heat (kW)
- Q 1, q 1 :
-
Useful heat (kJ/kg, %)
- Q 2, q 2 :
-
Loss of heat by the enthalpy of the exhaust gas (kJ/kg, %)
- Q 3, q 3 :
-
Loss of heat by incomplete chemical combustion (kJ/kg, %)
- Q 4, q 4 :
-
Loss of heat with the carbon present in the ashes (kJ/kg, %)
- Q 5, q 5 :
-
Loss of heat to the environment through the walls (kJ/kg, %)
- q 5nom :
-
Nominal heat lost to the environment through the walls (%)
- Q 6, q 6 :
-
Sensible heat loss in slag and ash removed by the scrubber [kJ/kg, %]
- St :
-
Sulfur according the elemental analysis of fuel on working moisture (%)
- \({\text{S}}_{\text{dry}}^{\text{t}}\) :
-
Sulfur according the elemental analysis of fuel on laboratory moisture (%)
- T 0 :
-
Ambient temperature (K)
- T c :
-
Fuel temperature (K)
- T esc :
-
Slag temperature (K)
- T g :
-
Temperature of the exhaust (K)
- T vs :
-
Superheated steam temperature (K)
- \(V_{\text{air}}^{o}\) :
-
Theoretical volume of air (m3/kg)
- V gs :
-
Volume of dry gas (m3/kg)
- \(V_{{{\text{H}}_{2} {\text{O}}}}^{o}\) :
-
Water vapor volume (m3/kg)
- \(V_{{{\text{N}}_{2} }}^{o}\) :
-
Volume of nitrogen (m3/kg)
- \(V_{{{\text{RO}}_{2} }}\) :
-
Triatomic gas volume (m3/kg)
- \({\text{W}}_{1}^{t}\) :
-
Laboratory moisture of fuel (%)
- \({\text{W}}_{2}^{t}\) :
-
Working fuel moisture (%)
References
Barin, Ihsan, and Gregor Platzki. 1995. Thermochemical data of pure substances. Weinheim, New York: VCH.
Basu, Prabir, Cen Kefa, and Louis Jestin. 2012. Boilers and burners: Design and theory. New York: Springer.
Binnewies, M., and E. Milke. 1999. Thermochemical data of elements and compounds. Weinheim, New York: Wiley-VCH.
Centeno-González, Felipe Orlando, Electo Eduardo Silva Lora, Helcio Francisco Villa Nova, Lourival Jorge Mendes Neto, Arnaldo Martín Martínez Reyes, Albert Ratner, and Mohsen Ghamari. 2017. CFD modeling of combustion of sugarcane bagasse in an industrial boiler. Fuel 193: 31–38. https://doi.org/10.1016/j.fuel.2016.11.105.
JANAF, and Thermal Research Laboratory Dow Chemical Company. 1971. JANAF thermochemical tables. Washington: U.S. National Bureau of Standards; for sale by the Supt. of Docs., U.S. Govt. Print. Off.
Kakac, Sadik. 1991. Boilers, evaporators, and condensers. Weinheim: Wiley.
Mitor, V., and I. Dubovki. 1973. CALCULATION OF HEAT BOILER UNITS. Normative Methods (in Russian).
Ravit, M. 1967. Effectiveness of fuel utilization (in Russian).
Soler, Pedro Beatón, and Electo Eduardo Silva Lora. 1991. Pruebas de balance térmico en calderas para bagazo. Ediciones ISPJAM.
Acknowledgements
The authors would like to thank the financial support in the form of scholarship from CAPES, CNPq, PRH-16 ANP and FAPEMIG of Brazil and the financial support of the CNPq process 407050/2013-2. It is also important to acknowledge the support of boiler manufacturer Caldema Equipamentos Industriais Ltda; also thanks to Ph.D Oscar Almazán del Olmo (ICIDCA—Cuban Institute for Research on Sugar Cane Derivatives) for his contributions to the revision of the manuscript.
Funding
This study was funded by the Brazilian National Counsel of Technological and Scientific Development (CNPq) process number 407050/2013-2. Also this study received financial support in the form of scholarship from government agencies CAPES, CNPq, PRH-16 ANP and FAPEMIG.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Centeno-González, F.O., Lora, E.E.S., Nova, H.F.V. et al. Programming the Inverse Thermal Balance for a Bagasse-Fired Boiler, Including the Application of a Optimization Method in MATLAB. Sugar Tech 20, 585–590 (2018). https://doi.org/10.1007/s12355-017-0573-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-017-0573-z