Abstract
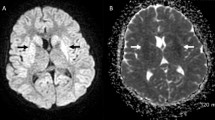
A 56-year-old man underwent cord blood transplantation for angioimmunoblastic T-cell lymphoma. He developed severe diarrhea and abdominal pain that persisted for more than 4 months. We suspected that he might have cord colitis syndrome (CCS), so metronidazole (MNZ) was administered. The patient’s abdominal pain and diarrhea showed some improvement after the initiation of MNZ therapy, but they worsened on the cessation of MNZ, which prompted us to resume MNZ treatment. After the patient had taken MNZ (1500–2000 mg/day) for 78 days, he developed somnolence and dysarthria. We diagnosed him with metronidazole-induced encephalopathy (MIE) based on the characteristic magnetic resonance imaging findings and the clinical course. The patient’s dysarthria and somnolence improved within a few days after the discontinuation of MNZ. CCS is a recently proposed clinical entity defined as a persistent diarrheal illness that is culture-negative, antibiotic-responsive, and not attributable to any known cause. Patients with CCS often have recurrent diarrhea after the discontinuation of MNZ and may require prolonged treatment for a median of 120 days. When treating CCS with MNZ, physicians should be alert for the development of MIE.



Similar content being viewed by others
References
Kuriyama A, Jackson JL, Doi A, et al. Metronidazole-induced central nervous system toxicity: a systematic review. Clin Neuropharmacol. 2011;34:241–7.
Herrera AF, Soriano G, Bellizzi AM, et al. Cord colitis syndrome in cord-blood stem-cell transplantation. N Engl J Med. 2011;365:815–24.
Sakemura R, Hayakawa H, Iida H, Ito M, Kajiguchi T. Successful treatment of a case of late-onset colitis after umbilical cord transplantation with metronidazole: a case report and literature review. Intern Med. 2017;56:3219–23.
Bhatt AS, Freeman SS, Herrera AF, et al. Sequence-based discovery of Bradyrhizobium enterica in cord colitis syndrome. N Engl J Med. 2013;369:517–28.
Gorkiewicz G, Trajanoski S, Högenauer C. Bradyrhizobium enterica in cord colitis syndrome. N Engl J Med. 2013;369:1866–7.
Shimoji S, Kato K, Eriguchi Y, et al. Evaluating the association between histological manifestations of cord colitis syndrome with GVHD. Bone Marrow Transpl. 2013;48:1249–52.
Alvarez RS, Richardson DA, Bent AE, et al. Central nervous system toxicity related to prolonged metronidazole therapy. Am J Obstet Gynecol. 1983;145:640–1.
Halloran TJ. Convulsions associated with high cumulative doses of metronidazole. Drug Intell Clin Pharm. 1982;16:409.
Patel K, Green-Hopkins I, Lu S, et al. Cerebellar ataxia following prolonged use of metronidazole: case report and literature review. Int J Infect Dis. 2008;12:e111–4.
Kim E, Na DG, Kim EY, et al. MR imaging of metronidazole-induced encephalopathy: lesion distribution and diffusion-weighted imaging findings. AJNR Am J Neuroradiol. 2007;28:1652–8.
Cação G, Fontes S, Salgado M, et al. Metronidazole-induced central and peripheral nervous system toxicity. Neurol Sci. 2015;36:1737–9.
Sarna JR, Brownell AK, Furtado S. Reversible cerebellar syndrome causedby metronidazole. Can Med Assoc J. 2009;181:611–3.
Acknowledgements
We are grateful to Dr. Ota for assisting with the examination of Bradyrhizobium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest in association with the present study.
Human rights
All procedures followed have been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iemura, T., Kitano, T., Ishii, A. et al. Metronidazole-induced encephalopathy during treatment for refractory diarrhea after cord blood transplantation. Clin J Gastroenterol 12, 414–419 (2019). https://doi.org/10.1007/s12328-019-00959-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12328-019-00959-x