Abstract
Introduction
An easy-to-use, multiuse, single-patient, electromechanical autoinjector, the SMARTCLIC®/CLICWISE® injection device, was recently developed to improve the self-administration options available to patients with chronic inflammatory disease treated with biologic agents. An extensive series of studies were conducted to guide the design and development of this device and to ensure its safety and effectiveness.
Methods
Participants in two user preference studies and three formative human factor (HF) studies evaluated evolving iterations of the autoinjector device, dose dispenser cartridge, graphical user interface, and informational materials; participants in a summative HF test subsequently assessed the final proposed commercially representative product. In the user preference studies, rheumatologists and patients with chronic inflammatory disease, interviewed online and in-person, provided feedback on the design and functionality of four prototypes. In the HF studies, the safety, effectiveness, and usability of adapted prototypes were assessed under simulated-use conditions by patients with chronic inflammatory disease, caregivers, and healthcare professionals (HCPs). The safety and effectiveness of the final refined device and system were confirmed in a summative HF test by patients and HCPs in simulated-use scenarios.
Results
Rheumatologists (n = 204) and patients (n = 39) interviewed in the two user preference studies provided feedback on the device size, feature ergonomics, and usability that guided prototype development in the subsequent formative HF studies. Observations from patients, caregivers, and HCPs (n = 55) participating in the latter studies yielded additional critical design revisions that culminated in development of the final device and system. Of 106 injection simulations conducted in the summative HF test, all resulted in successful medication delivery, and no potential harms were associated with injection-related use events.
Conclusion
Findings from this research facilitated development of the SmartClic/ClicWise autoinjector device and demonstrated that it could be used safely and effectively by participants representative of the intended-use population of patients, lay caregivers, and HCPs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Various self-injection devices are available for patients with chronic inflammatory diseases such as rheumatoid arthritis, but some may pose substantial obstacles for users, particularly for those with hand function impairment and/or anxiety related to self-injection. |
An easy-to-use, multiuse, single-patient electromechanical autoinjector called the SmartClic/ClicWise injection device was recently developed to address the needs of patients who regularly self-inject biologic therapies. |
A comprehensive series of user preference and human factor studies were conducted to guide the development of this device and assess its usability, safety, and effectiveness. |
What was learned from the studies? |
Critical feedback from study participants on device size, feature ergonomics, and usability culminated in the development of the final SmartClic/ClicWise injection device and system. |
In the summative human factor test, medication was successfully delivered in all injection simulations, and no potential harms resulted from injection-related use events. |
The SmartClic/ClicWise injection device can be used safely and effectively by patients, caregivers, and healthcare professionals. |
Introduction
Chronic inflammatory diseases affect an estimated 5–7% of adults in Western society [1] and are a leading cause of mortality, associated with more than 50% of all deaths worldwide [2]. Over the past few decades, the introduction of anti-inflammatory biologic therapies has marked a critical advance in the treatment of chronic inflammatory diseases such as rheumatoid arthritis (RA) [3]. Biologics are frequently administered via subcutaneous injection, often by patients at home [4]. Self-injectable biologics provide a range of benefits, including reduced costs, greater treatment adherence, enhanced “self-efficacy,” and reduced caregiver burden [5, 6]. Patients’ ability to confidently self-inject may help them experience a greater sense of autonomy, reduce the psychological impact of living with a chronic condition, and improve their health-related quality of life [6,7,8].
Patients with chronic inflammatory diseases such as RA currently have access to a variety of devices for self-injection, including vials and empty syringes, prefilled syringes, and autoinjectors, but certain obstacles can impede the achievement of consistent and successful self-injections. Pain in the hands, and/or impaired hand function, can increase the difficulty of using a self-injection device, potentially increasing the risk of drug administration errors and injection site reactions [9]. In addition, feelings of anxiety associated with self-injection, as well as aversion to or fear of the process, are reasons for treatment discontinuation among patients with RA [10, 11].
The type of device used for biologic delivery may also influence the self-injection experience of patients with chronic inflammatory diseases. Easy-to-use self-injection devices can help patients overcome barriers and improve long-term disease management and outcomes [12]. Recently, electromechanical autoinjector devices or electronic autoinjector devices have been developed to address the needs of patients who regularly self-inject. In patients with multiple sclerosis, electronic autoinjector devices have been shown to improve injection tolerability and patient satisfaction compared with manual injection and help increase adherence [13]. By allowing adjustment of injection speed and concealment of the needle during injection, new electronic autoinjector devices may assist patients in overcoming psychological barriers to self-injection and improve comfort [14]. Autoinjector devices can also record injection history, supporting better communication between patients and healthcare professionals (HCPs) about self-injection patterns [11].
The development of self-injection devices heavily relies on the performance of end-user preference and human factor (HF) studies to ensure optimum design and usability and to confirm safety and effectiveness [15, 16]. By placing the intended user at the center of the design process, this type of research improves designers’ understanding of human preferences and limitations that affect device usability. Iterative refinement of the device’s design through end-user and HF research improves the likelihood that the device will be user-friendly and will support treatment adherence. Currently, several regulatory agencies, including the European Medicines Agency, the Japanese Ministry of Health, Labour, and Welfare, and US Food and Drug Administration, emphasize the importance of HF studies in the device development process, specifying that formative usability evaluations and summative (validation) usability tests be conducted during the product design and development period [15,16,17,18,19,20].
An electromechanical autoinjector, the SMARTCLIC®/CLICWISE® injection device (manufactured by PHC Corporation, Tokyo, Japan), was recently developed for subcutaneously administered biologic therapies. The development program for this device was conceived and implemented to create an easy-to-use, multiuse, single-patient autoinjector that would provide patients and caregivers an administration option beyond the previously developed self-injection options, including vial and empty syringe, prefilled pen or syringe, and other autoinjector presentations. This report provides a summary of the main methods and findings of the fundamental studies in this program that contributed to final model development of the SmartClic/ClicWise injection device.
Methods
A comprehensive series of seven studies [two user preference studies, three formative HF studies, one summative HF test, and one sharps injury prevention (SIP) study] was conducted to guide device development and to ensure the safety and effectiveness of the SmartClic/ClicWise injection device (Fig. 1). A combination of HCPs, lay caregivers, and patients with chronic inflammatory diseases were enrolled to assess the autoinjector device itself, the main components of the device [e.g., the dose dispenser cartridge (DDC) and the graphical user interface (GUI)], and the device system’s supporting informational materials [e.g., instructions for use (IFU), user manual, labeling, and packaging].
User Preference Studies
Two user preference studies were conducted by Cello Health Insight (New York, NY, USA) between July and September 2018, at four locations in Argentina, France, Germany, and Japan. The original working prototype of the electronic autoinjector device (Prototype X) was created by the former Panasonic Healthcare Co., Ltd. (currently, PHC Corporation), and three additional prototypes (Prototypes A, B, and C) were created from a platform designed by PHC Corporation (Tokyo, Japan) in collaboration with Pfizer (Cambridge, UK). Prototype X was evaluated in the first user preference study involving rheumatologists, and all four prototypes were evaluated in the second user preference study involving patients with chronic inflammatory diseases. The main objective of these studies was to obtain feedback from rheumatologists and patients about the design and function of the autoinjector models and the overall device system. This feedback would help guide the device designers’ selection of the prototype to be advanced for subsequent evaluation in the HF studies.
Participants
The first user preference study involved rheumatologists who satisfied the following screening criteria: qualified in their specialty for 3–35 years; spent ≥ 75% of their professional time involved in direct clinical care; personally responsible for the initiation and management of biological therapy in ≥ 10% of patients and a total of ≥ 5 patients; and provided care for ≥ 10 patients with moderate to severe RA, axial spondyloarthritis (axSpA), or psoriatic arthritis (PsA) in a typical month (only RA in Japan).
The second user preference study included men or women, 18–75 years of age, who had previously received a diagnosis of RA, axSpA, or PsA (only RA in Japan). Eligible patient participants were also currently taking, or had previously taken, a biologic agent or were biologic-naïve but had used a self-injectable device and were currently using an autoinjector to self-inject medication.
Interviews
The participating rheumatologists completed a 40-min online interview, answering questions about their perceptions of the potential impact of a new autoinjector device (Prototype X) versus an existing autoinjector (MYCLIC) for the treatment of chronic inflammatory disease, the probability of their prescribing and patients using a new autoinjector device, and potential motivations and obstacles to the device’s use.
Participating patients took part in a 60-min, in-person interview after viewing an introductory video and visually inspecting and/or handling the four autoinjector prototypes (Prototypes X, A, B, and C). They were asked to provide feedback on their prior experiences with self-injection devices, impressions of the autoinjector device models, and recommendations for potential improvements in the autoinjector design and functionality.
Feedback on the design and function of the autoinjector models and the overall device system was used to design an initial prototype device and DDC cap that were assessed by participants in the subsequent HF studies.
Formative Human Factor Studies
Three consecutive formative HF studies were conducted in February, June, and November 2019. The first HF study was conducted in London, UK, by Design Science (Philadelphia, PA, USA) and the second and third HF studies in Manchester and London, UK, by Emergo by UL (Cambridge, UK). The aim of these studies was to assess the autoinjector prototypes, DDC, and other components of the overall system and determine if the device could be used safely and effectively under conditions of simulated use. After each study, the prototype design was evaluated and refined in response to user feedback to ensure the final design met the overall development goals of being easy to use and intuitive.
Participants
A distinct group of participants took part in each formative study. Individuals eligible to participate were representative of the intended-use population, including patients with a chronic inflammatory disease (i.e., RA, PsA, axSpA, or plaque psoriasis), caregivers, and HCPs. Patients and caregivers were at least 18 years of age, injection-experienced or injection-naïve, and had possible hearing, visual, dexterity, or other impairments. HCPs (e.g., licensed practical nurses, registered nurses, nurse practitioners, and physician assistants) were 18–70 years old, with experience providing care for patients with chronic inflammatory disease and in injecting others. All participants were naïve to the SmartClic/ClicWise injection device.
Study Devices and Materials
The working prototypes of the autoinjector device and DDC used in the testing sessions included device screen instructions and sounds, but no labels, and did not contain any medication. The prototypes were updated between each formative HF study on the basis of feedback guiding device engineering and usability. (Major updates are described in the subsequent Results section; a detailed description of minor adaptations is beyond the scope of this article.)
Instructional materials included the IFU for the DDC and a user manual for the device, provided in printed paper format. The instructional materials for these sessions were produced using the same high-quality paper, and in the same size and shape, as the package leaflets to be included in the autoinjector device package when commercially available to patients.
Participants representing patients performed simulated injections using commercially available injection pads attached to their abdomen or thigh, which had nonwoven conductive foil to ensure correct function of the autoinjector device’s skin sensor. Participants representing caregivers and HCPs simulated injections into injection pads attached to mannequins.
Use Scenarios
In the formative HF studies, participants took part in single, one-on-one testing sessions. Study moderators provided participants with a series of injection tasks to simulate intuitive-use scenarios, in which participants only received the autoinjector prototype and DDC, and commercially representative, simulated-use scenarios, in which they also received instructional materials. In the third formative HF study, users were divided into trained and untrained subgroups, with trained participants receiving instruction from an HCP about how to use the device an hour before the test session.
In each session, moderators observed and objectively recorded participants’ behavior and feedback as the participants performed the injection tasks. Moderators subsequently asked follow-up questions regarding prototype use. After the injection tasks, participants also completed a knowledge comprehension assessment designed to evaluate how easily they understood the device’s GUI, IFU, and user manual. Recording of use events (i.e., use errors, close calls, and use difficulties) and participant feedback throughout the three formative studies facilitated iterative refinement of the device components with the aim of mitigating the likelihood of such events.
Summative Human Factor Test
The summative HF test was conducted by Design Science in Munich, Germany, in June/July 2020. Participants received the autoinjector device, DDC, IFU, user manual, and packaging that had been iteratively refined during the three formative HF studies as an accurate representation of the final, to-be-marketed, overall system. The objective of the summative HF test was to validate that the refined device and system could be used safely and effectively by the intended population in the intended environment of use. It was also conducted to validate that users could learn from errors committed during initial injections by correcting the errors on subsequent injections, and/or demonstrating a clear understanding of the instructional materials and steps involved in performing safe and effective injections so they would improve their performance on subsequent injections.
Participants
A discrete set of participants representative of the intended user population was selected for the summative HF test. Eligible patient participants had RA, juvenile idiopathic arthritis, PsA, axSpA, or plaque psoriasis; were 18–70 years of age; were injection-experienced or injection-naïve; and had possible hearing, visual, dexterity, or other impairments. Eligible HCPs (e.g., licensed practical nurses, registered nurses, nurse practitioners, and physician assistants) were at least 18 years old, had experience providing care for patients with arthritis, and had experience in injecting others. None of the participants had been trained to use the device.
Use scenarios
Participants in the summative HF test were given the opportunity to perform ≤ 3 simulated injections into an injection pad worn on their bodies (patients) or attached to a mannequin (HCPs). All participants were required to complete two injection simulations without training (use of instructional material was optional); the first injection simulated a user’s first time injecting with the device, and the second injection simulated a routine injection. Those who did not refer to instructional materials during the first or second injection simulations were asked to perform a third injection after reviewing the instructional materials. A knowledge assessment was conducted to test risk mitigations aimed at avoiding safety-related harms, and other important tasks not evaluated in the injection simulations.
Sharps Injury Prevention Study
As an adjunct to the user preference and HF studies, the SIP study was conducted by Design Science in Munich, Germany, in September/October 2020. The aim of this study was to assess the autoinjector device and DDC’s SIP feature under simulated-use conditions. After taking part in the injury prevention segment of the study, participants also provided their opinions on the ease of use, feature design, effectiveness, and injection speed of the SmartClic/ClicWise device, and the functionality of the device’s proposed mobile app. The methodology and results of the injury prevention segment are summarized in this report, but those of the opinion segment of the study are published elsewhere [21].
Participants
Licensed HCPs (including registered, licensed practical, and licensed vocational nurses; certified nursing assistants; physician assistants; medical doctors; and doctors of osteopathic medicine) were eligible to participate in the study if they were at least 18 years of age, cared for patients with rheumatologic/rheumatic conditions, and had experience in injection administration.
Use Scenarios
After receiving initial training by the moderator on how to correctly use the device, the HCPs completed ≤ 2 practice injections. The participants subsequently viewed a demonstration on the proper use of the device and DDC, during which they had access to the IFU. In a 30-min session conducted in a simulated clinical environment, they completed 20 injections into an injection pad using a single device and 20 DDCs to demonstrate the successful activation of the SIP feature (as confirmed by the moderator). After completing the injection simulations, the participants were interviewed about the device’s ease of use and the SIP feature.
Ethical Considerations
In the above studies, research instrument design, participant recruitment, compensation, consent, data handling, data privacy, and volunteers’ statements were handled following the standard operating procedures of the companies conducting the research (i.e., Cello Health Insight, Design Science, and Emergo by UL). Participants consented to participate in testing the device as well as having their data analyzed and published in medical journals. All participants received compensation for their participation within “fair market value” rules.
Results
User Preference Studies
First User Preference Study (Rheumatologists)
A total of 204 rheumatologists participated in the first user preference study of the original working autoinjector device (Prototype X). The majority (84%) were 35–64 years of age, and spent most of their time (89%) providing direct clinical care and provided care for an average of 87, 29, and 34 patients with RA, AxSpA, and PsA, respectively, each month (Supplementary material, Table S1A).
In their review of the prototype, the rheumatologists described several benefits and obstacles that would influence device use (Figs. 2A, 3). An essential benefit was the potential for the device to improve patients’ ability to maintain the prescribed dosing schedule because the device allows them to track their injection history, receive injection reminders, and have a better experience with self-injection. Major concerns were that older or disabled patients may find the device challenging to use, and patients and caregivers may require training. The device’s battery was also considered a potential weakness, as depleted batteries could leave patients unable to self-inject their medications. Recommendations for improvements were primarily focused on device size and usability, e.g., making the device smaller and lighter; improving the grip with finger indentations; increasing the size and clarity of lettering on the device; and allowing the injection button to be triggered only when the user has placed the device on the skin. Participants also suggested use of a rechargeable battery or inclusion of an indicator of the battery level remaining and/or the number of injections possible with the battery to help minimize concerns about battery life.
The rheumatologists considered the device’s potential connectivity with an accompanying app via Bluetooth® communication, to record and plan injections, would be beneficial because it would allow patients to receive injection reminders and have a greater sense of autonomy, with potentially fewer physician visitsFootnote 1. However, this feature also raised some concerns about the ability of older or disabled patients to successfully use an app. Moreover, the rheumatologists emphasized the importance of data security to protect patients’ sensitive information as well as patient–physician trust.
Second User Preference Study (Patients)
Thirty-nine adult rheumatology patients were interviewed in the second user preference study. Their ages ranged from 20 to 75 years, and most (62%) had RA (Supplementary material, Table S1B). In initial interviews, the patients indicated their comfort and confidence in the self-injection process were of paramount importance, and any device features that specifically enhanced these feelings were desirable. Such features included mechanisms allowing the device to provide feedback on its correct placement on the skin, confirm the successful administration of a dose, and record and plan injections. They also cited the desirability of an ergonomic form that felt secure in their hands while requiring minimal effort to use. The possibility of pairing the device with an app via Bluetooth communication was broadly accepted. Some participants who owned smartphones indicated that they were already using apps to record and plan their injections. A minority suggested that such “smart” devices may overcomplicate the process of self-injection.
After viewing the introductory video and/or visually inspecting and handling the four prototypes (original working Prototype X and Prototypes A, B, and C), the patients provided their feedback on each (Fig. 2B). Features that had an impact on patients’ preferences were (1) a smaller overall size and textured surface that made handling feel more secure for the majority and (2) ergonomic positioning of a large, angled injection button below a display screen. These features allowed a natural positioning of the thumb and facilitated single-handed use, while always maintaining visibility of the GUI.
Formative Human Factor Studies
A total of 55 participants (patients, n = 23; lay caregivers, n = 20; and HCPs, n = 12) provided feedback on iterative versions of the autoinjector device, IFU, user manual, labeling, and packaging in three consecutive formative HF studies. Demographic and other characteristics of these individuals are summarized in the Supplementary material, Table S2.
Across these studies, all but one participant successfully completed an injection in both the intuitive-use and simulated-use scenarios (Table 1). One participant in the second formative study did not deliver the injection in the correct site because the participant held the device upside down on the skin pad. The participant assumed that the direction of the needle would be “through” the device in the same direction as the cartridge was inserted. The participant understood that the needle was located at the other end of the cartridge during a second attempt. As shown in Table 1, the number of observed use events decreased in each successive study, demonstrating that the iterative adaptations of the prototype improved its usability.
The greatest number of design revisions occurred between the first and second formative HF studies (Fig. 1). Based on researchers’ observations of use events and participants’ feedback during the first formative HF study, researchers found that the device design did not provide visual cues about how the device should be oriented or the DDC should be inserted, and the GUI and its prompts were not sufficiently prominent. The observed use events related to the button configuration were attributed to the inability of the 2-button interface to allow multidirectional navigation; the buttons’ unclear functionality during injection delivery; and the buttons’ inconspicuous location, shape, texture, color, and labeling. Some terminology and phrasing used in the GUI and IFU (e.g., “update”, “eject”, “press and hold,” and “pull”) and some graphical elements (e.g., arrow styles) were reported to be unclear and potentially contributed to use events.
Prior to the second formative HF study, the prototype was adapted in several ways to address the above-mentioned findings. Two new buttons labeled with intuitive symbols indicating their functions were incorporated into the device immediately below the injection button to simplify navigation through the GUI. The “cancel/eject” button was relocated from above the display screen to directly beneath the “menu down” button, forming a row of three new buttons below the injection button. User interaction cues were cleared from the top of the device so that device orientation during injection would be more obvious. The color of the injection button was changed from grey to teal, and a dot-within-a-circle symbol was added to improve users’ recognition of the feature as a button. The same teal color was also used on the lower part of the device that housed the cartridge door and skin sensor, and a downward arrow symbol was added to indicate where the DDC would be inserted and where the injection point would be located during use.
In addition, all wording displayed on the GUI menu was reviewed and revised to avoid any potentially ambiguous words or phrases and to improve clarity. The animated action illustrations, arrows, and visual confirmation feedback were also refined and simplified. The GUI was updated to be consistent with the illustrations shown in the IFU; this alignment allowed the same illustrated action to be just as effective and understandable on the small, low-resolution GUI as on the larger printed IFU.
In addition to observations related to the simulated injections performed in the third formative HF study (Table 1), this study provided important feedback on device packaging, including the box used to package the device, the inner tray used to secure the items packaged within the box, the graphic appearing underneath the lid, labeling, and the cartridge box and sleeve. In many cases, a clear “favorite” was identified. For example, about 90% of participants preferred the box with a magnetic closure lid over the box with a non-magnetic closure lid. The proportions of participants who preferred the inner tray made from hard plastic, foam, or paper foam were more evenly distributed (39%, 33%, and 28%, respectively), but all participants were able to easily remove the inner tray during simulated use.
General subjective participant feedback on each element of the device and system was also captured in the third formative HF study. Most of the opinions shared by the participants were positive. For example, 11% of participants thought the device was a good size, easy to load, and the skin sensor was impressive; 16% thought the device was easy to hold and not too heavy; and 32% thought it was easy to use.
Throughout the three formative HF studies, numerous changes were also executed in the layout, text, and illustrations included in the IFU, user manual, labeling, and packaging to ensure maximum clarity and improve the usability of the device and DDC.
Summative Human Factor Test
Forty-five untrained individuals participated in the summative HF test (injection-experienced patients, n = 15; injection-naïve patients, n = 15; HCPs, n = 15). All 106 injection simulations observed during the summative HF test would have resulted in the successful delivery of medication (Table 1). Based on objective and subjective data collected in this test, the use events were primarily attributable to first-time use of the device by the participants (e.g., assumptions regarding the device’s use without reference to the instructional materials or lapse of attention), study artifacts (e.g., use of an injection pad instead of real skin), or negative transfer (i.e., participants’ applying knowledge from prior use of other injection devices). No potential harms were associated with any use events related to the injection process.
In the post-injection knowledge assessment, all participants successfully explained how they knew when the device was ready to inject, when the injection started and finished, and when a full dose had been delivered. Participants who committed errors during their initial injections were able to learn from that experience and made the necessary corrections during subsequent injections. After completion of the summative HF test, several updates were executed in the IFU, user manual, labeling, and packaging to address residual use errors.
Sharps Injury Prevention Study
Twenty-five nurses participated in the SIP study and completed a total of 500 injections. No lock-out or SIP feature failures were reported during the study. True failure rates of the device and related materials were estimated to be ≤ 0.7% and ≤ 1.1%, with 97.5% and 99.5% confidence, respectively. As reported previously, in the follow-up interviews, the participants indicated that the device was easy to use and had the potential to reduce medication errors [21]. Due to the positive responses and outcomes obtained in this study, further alterations in the device design or functionality were not considered warranted.
Discussion
The SmartClic/ClicWise injection device is a reusable autoinjector that provides an additional option beyond the vial and empty syringe, prefilled syringe, or autoinjector presentations currently available for patients who self-inject medications. After two user preference studies were conducted with rheumatologists and patients to establish the basic shape and design of the device, three formative HF studies were completed that enrolled 55 participants representative of the intended-use population. The findings from the latter studies were crucial to the development of the final device and its associated components (Fig. 4). Finally, a summative HF test was conducted in 45 individuals who completed more than 100 injection simulations using the final device, resulting in the successful delivery of medication in all attempts without potential harm associated with any use event.
Evidence suggests that difficulties with self-injecting, such as those due to manual dexterity disabilities or a dislike or fear of the procedure, can negatively impact a patient’s adherence to treatment [9,10,11]. Development of a device that is easy to use and intuitive for patients and caregivers should help to mitigate some of these barriers. Simplification of treatment and provision of clear instructions are recognized strategies for improving adherence [22].
Feedback from the user preference and HF studies was crucial to optimizing the design and features of the SmartClic/ClicWise injection device for use by patients with chronic inflammatory diseases such as RA. For example, feedback from the rheumatologist user preference study encouraged design adaptations ensuring the device does not run out of battery and that the battery level is displayed. Each device is designed to administer 160 injections of medication and 20 training injections, allowing 3 years of use in patients injecting once weekly. As a result, potential user concern regarding battery expiration is avoided. Moreover, based on participant input, single-handed use of the device was facilitated by altering the number and configuration of the buttons and minimizing the device’s weight by limiting the amount of material used in its manufacture. Although a clear consensus regarding the device size was not obtained, as preferences for small and large grips appeared equally distributed, a smaller overall size was selected for the device. The back of the device was curved to allow the index finger to support the device. In addition, certain aspects of the device’s design (e.g., lack of texture, knobs, or ruffles in the plastic, which would have created dirt traps) were selected because they enable easier cleaning. This consideration is particularly important because the device is intended to last up to 3 years, and many users are treated with immunomodulators, for whom hygiene is critical.
Studies have found that the majority of patients report a preference for prefilled pens/autoinjectors over prefilled syringes because they are convenient, quick and easy to use, and more comfortable/less painful to use, and have built-in safety features [23,24,25]. Patients with needle phobia are likely to prefer the use of an autoinjector over a prefilled syringe, as an autoinjector’s needle is concealed throughout the injection process. Patients who suffer from injection-related pain, which can cause pain catastrophizing [26, 27] and dread of injecting [28, 29] over time, may prefer a prefilled syringe over an autoinjector because the former allows greater control of the speed/duration of injection [14]. With this in mind, from the outset, the SmartClic/ClicWise injection device was developed with three factory-set options for injection speed, providing patients with greater control than the single speed offered by current autoinjectors. Instead of the spring found in current autoinjectors, geared motors are used to operate the device’s plunger rod to allow for variable injection speeds. The ability to alter the injection speed should help patients adjust the speed based on their individual comfort level. It is important to note that slower injection speeds have previously been reported to better prevent bruising and pain than faster speeds [30].
In addition to the ergonomic design and variable injection speed, the SmartClic/ClicWise injection device includes other safety features, such as a simple capacitive skin sensor and needlestick prevention features. The skin sensor confirms the device is placed in full contact with the skin, reducing the likelihood of accidental injection misfire and drug wastage [6]. To avoid needlestick injuries, the needle cap can only be removed once the DDC is correctly inserted into the device. The shape and texture of the cap were refined specifically on the basis of feedback obtained from the user preference and HF studies. Following an injection, the DDC releases a guard that extends over the needle and locks into place. A sensor within the device ensures the needle guard is in the correct position before allowing the DDC to be released.
Similar to other autoinjectors, the needle of the SmartClic/ClicWise injection device remains hidden throughout the injection procedure. The risk of an incomplete injection is also minimized by the device delivering a fixed dose and ensuring the required “hold time” prior to needle withdrawal. This hold time, in combination with the 90° angle of injection, help to minimize the chance of drug leakage from the injection site, as it provides sufficient time for the skin’s elasticity to absorb some of the force of the fluid being injected [31].
As mentioned above, each SmartClic/ClicWise device is equipped with a training cartridge to administer up to 20 training injections, with the aim of improving both safety and adherence. Using training cartridges at home allows the user to become familiar with the device and injection process and to practice injection technique. While a new user often undergoes in-office training sessions with his/her healthcare provider, at-home practice can help reinforce learnings from those sessions at the user’s own pace.
The SmartClic/ClicWise injection device can be paired with a dedicated optional app via Bluetooth communication to provide treatment tracking and reporting features (e.g., the date/time of injections). Patients can program either the device or the app with their treatment schedule and can set reminders about their next scheduled injection. The device also warns about possible excessive injections. The potential connection between the injection device and the app via Bluetooth communication was generally well received by participants. Although a minority considered this option to be an added complication, this perspective may diminish over time as technology is increasingly used in people’s everyday lives. Importantly, however, the device is fully operational without the app, allowing users to choose based on their personal comfort, experience level, and assessment of the app’s usefulness.
Another important design feature is the radiofrequency identification (RFID) tag embedded in the DDC that logs the dose strength and serialization (i.e., lot number) for pharmacovigilance and clinical reporting purposes. As a result, if an injection is only partially administered, the autoinjector can record the information (including the DDC lot number) as an injection error, prompting an investigation. RFID technology also provides expiration date information and helps prevent counterfeiting and reuse of the same cartridges.
In addition to its clinical and lifestyle benefits to patients and caregivers, the SmartClic/ClicWise reusable injection device and associated DDCs consume less plastic compared with disposable autoinjectors. When comparing the amount of plastics used over the lifetime of a single SmartClic/ClicWise injection device (i.e., for the device itself, 160 doses provided by 160 DDCs, and the associated plastic packaging) with that used for 160 disposable autoinjectors, an estimated 1.8 kg of plastics would be saved over the lifetime of each SmartClic/ClicWise injection device, representing a 41% reduction. Moreover, this reduction in plastics would result in 4.2 kg less CO2 being generated via incineration of waste per device.
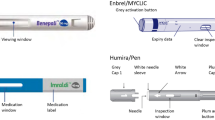
Devices similar to the SmartClic/ClicWise injection device are currently available or in development, including the AutoTouch® (Amgen, Thousand Oaks, CA, USA) [32], the ava Connect® (UCB, Brussels, Belgium) [33], RebiSmart® (Merck Serono, Geneva, Switzerland) [34], BETACONNECT™ (Bayer Healthcare, Whippany, NJ, USA) [35], Easypod® (Merck Serono, Geneva, Switzerland) [36], and GROWJECTOR® L (PHC Corporation, Tokyo, Japan, and JCR Pharmaceuticals Co., Ltd., Hyogo, Japan) [37]. The SmartClic/ClicWise injection device shares several important features with other electromechanical autoinjectors designed to improve patients’ experience with self-injection, including audible and visual signals, recording of the prior dose, confirmation of injection completion, on-screen instructions, and a skin sensor. In a phase 3b study of the RebiSmart autoinjector, patients rated these features as useful or very useful [38]. In addition, evidence from subsequent research suggested that these features contributed to the high rate of treatment adherence observed with that autoinjector’s use (96.5%), which correlated with improvements in clinical outcomes [39]. In HF and open-label studies of the ava Connect autoinjector, an electromechanical injection device for biologic administration similar to the SmartClic/ClicWise autoinjector, patients reported the device was safe and effective to use, and they reported high levels of confidence, satisfaction, and a positive self-injection experience [40,41,42].
A complete description of the strengths and limitations of HF and usability testing of devices is beyond the scope of this report. However, it is important to note that the participant HF evaluations were conducted under simulated-use conditions (which are typical and acceptable for this type of injection device as part of the development process) and not under actual-use conditions; as a result, the real experience of performing an injection into human skin could not be evaluated. Real-world studies with prescribed biotherapeutics would be valuable to further our understanding of device benefits and provide insights into potential further device development.
Conclusions
In this series of studies, the SmartClic/ClicWise injection device and its associated components were used in a safe and effective manner by participants representative of the intended-use population of patients, lay caregivers, and HCPs. Feedback from the study participants contributed to important adaptations of the device design and operation prior to selection of the current, final model. This research facilitated optimization of the autoinjector, enabling patients to reliably and confidently self-administer their medication at home, and potentially enhancing treatment adherence, patient outcomes, and quality of life.
Notes
The Bluetooth® word mark and logos are registered trademarks owned by Bluetooth SIG, Inc.
References
El-Gabalawy H, Guenther LC, Bernstein CN. Epidemiology of immune-mediated inflammatory diseases: incidence, prevalence, natural history, and comorbidities. J Rheumatol Suppl. 2010;85:2–10.
Furman D, Campisi J, Verdin E, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019;25:1822–32.
McInnes IB, Gravallese EM. Immune-mediated inflammatory disease therapeutics: past, present and future. Nat Rev Immunol. 2021;21:680–6.
Bittner B, Richter W, Schmidt J. Subcutaneous administration of biotherapeutics: an overview of current challenges and opportunities. BioDrugs. 2018;32:425–40.
Náfrádi L, Nakamoto K, Schulz PJ. Is patient empowerment the key to promote adherence? A systematic review of the relationship between self-efficacy, health locus of control and medication adherence. PLoS ONE. 2017;12: e0186458.
Boeri M, Szegvari B, Hauber B, et al. From drug-delivery device to disease management tool: a study of preferences for enhanced features in next-generation self-injection devices. Patient Prefer Adherence. 2019;13:1093–110.
Larsson I, Bergman S, Fridlund B, Arvidsson B. Patients' independence of a nurse for the administration of subcutaneous anti-TNF therapy: a phenomenographic study. Int J Qual Stud Health Well-being. 2010; 5.
Salmon P, Hall GM. Patient empowerment and control: a psychological discourse in the service of medicine. Soc Sci Med. 2003;57:1969–80.
Keininger D, Coteur G. Assessment of self-injection experience in patients with rheumatoid arthritis: psychometric validation of the Self-Injection Assessment Questionnaire (SIAQ). Health Qual Life Outcomes. 2011;9:2.
Bolge SC, Goren A, Tandon N. Reasons for discontinuation of subcutaneous biologic therapy in the treatment of rheumatoid arthritis: a patient perspective. Patient Prefer Adherence. 2015;9:121–31.
van den Bemt BJF, Gettings L, Domańska B, Bruggraber R, Mountian I, Kristensen LE. A portfolio of biologic self-injection devices in rheumatology: how patient involvement in device design can improve treatment experience. Drug Deliv. 2019;26:384–92.
Maniadakis N, Toth E, Schiff M, et al. A targeted literature review examining biologic therapy compliance and persistence in chronic inflammatory diseases to identify the associated unmet needs, driving factors, and consequences. Adv Ther. 2018;35:1333–55.
Bayas A. Improving adherence to injectable disease-modifying drugs in multiple sclerosis. Expert Opin Drug Deliv. 2013;10:285–7.
Verdun di Cantogno E, Russell S, Snow T. Understanding and meeting injection device needs in multiple sclerosis: a survey of patient attitudes and practices. Patient Prefer Adherence. 2011; 5:173–80.
United States Food and Drug Administration. Human factors studies and related clinical study considerations in combination product design and development. Draft guidance for industry and FDA staff 2016. https://www.fda.gov/files/about%20fda/published/Human-Factors-Studies-and-Related-Clinical-Study-Considerations-in-Combination-Product-Design-and-Development.pdf. Accessed October 21, 2022.
United States Food and Drug Administration. Applying human factors and usability engineering to medical devices. Guidance for industry and Food and Drug Administration staff. 2016. https://www.fda.gov/media/80481/download. Accessed October 21, 2022.
MHRA. Guidance on applying human factors and usability engineering to medical devices including drug-device combination products in Great Britain. 2021. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/970563/Human-Factors_Medical-Devices_v2.0.pdf. Accessed October 21, 2022.
Pelayo S, Marcilly R, Bellandi T. Human factors engineering for medical devices: European regulation and current issues. Int J Qual Health Care. 2021;33:31–6.
International Electrotechnical Commission. Medical devices—Part 1: application of usability engineering to medical devices (IEC 62366–1:2015). 2015. https://webstore.iec.ch/publication/21863. Accessed October 21, 2022.
Medical Device Evaluation Division and Compliance and Narcotics Division, Pharmaceutical Safety and Environmental Health Bureau. Concerning requirements for usability engineering of medical devices: Handling of revisions to Japanese Industrial Standards 2022. https://www.mhlw.go.jp/hourei/doc/tsuchi/T221004I0050.pdf.
Alten R, Moss S, Hahne S, Muriset A, Gruben D, Latymer M. High usability and applicability ratings for the new Smartclic®/ClicWise® injection device: evidence from a health care professional opinion study (in press). Rheumatol Ther. 2022.
Osterberg L, Blaschke T. Adherence to medication. N Engl J Med. 2005;353:487–97.
Dashiell-Aje E, Harding G, Pascoe K, DeVries J, Berry P, Ramachandran S. Patient evaluation of satisfaction and outcomes with an autoinjector for self-administration of subcutaneous belimumab in patients with systemic lupus erythematosus. Patient. 2018;11:119–29.
Roszkiewicz J, Swacha Z, Smolewska E. Prefilled pen versus prefilled syringe: a pilot study evaluating two different methods of methotrexate subcutaneous injection in patients with JIA. Pediatr Rheumatol Online J. 2020;18:64.
Vermeire S, D’Heygere F, Nakad A, et al. Preference for a prefilled syringe or an auto-injection device for delivering golimumab in patients with moderate-to-severe ulcerative colitis: a randomized crossover study. Patient Prefer Adherence. 2018;12:1193–202.
Hammer HB, Uhlig T, Kvien TK, Lampa J. Pain catastrophizing, subjective outcomes, and inflammatory assessments including ultrasound: results from a longitudinal study of rheumatoid arthritis patients. Arthritis Care Res (Hoboken). 2018;70:703–12.
St Clair-Jones A, Prignano F, Goncalves J, Paul M, Sewerin P. Understanding and minimising injection-site pain following subcutaneous administration of biologics: a narrative review. Rheumatol Ther. 2020;7:741–57.
Peyrot M, Rubin RR, Kruger DF, Travis LB. Correlates of insulin injection omission. Diabetes Care. 2010;33:240–5.
Aronson R. The role of comfort and discomfort in insulin therapy. Diabetes Technol Ther. 2012;14:741–7.
Kim H, Park H, Lee SJ. Effective method for drug injection into subcutaneous tissue. Sci Rep. 2017;7:9613.
Præstmark KA, Stallknecht B, Jensen ML, Sparre T, Madsen NB, Kildegaard J. Injection technique and pen needle design affect leakage from skin after subcutaneous injections. J Diabetes Sci Technol. 2016;10:914–22.
Amgen Inc. Enbrel Mini® cartridge with AutoTouch® autoinjector. 2020. https://www.enbrel.com/resources/enbrel-mini-cartridge-with-autotouch-autoinjector. Accessed October 21, 2022.
UCB. UCB gains CE mark for ava Connect[®], a first-in-class electromechanical device for use with biologic treatment in rheumatology and dermatology. 2021. https://www.ucb.com/stories-media/Press-Releases/article/UCB-Gains-CE-Mark-for-ava-Connect-a-first-in-class-electromechanical-device-for-use-with-biologic-treatment-in-rheumatology-and-dermatology. Accessed October 21, 2022.
Merck Serono. Merck Serono launches RebiSmart(TM), first electronic injection device for delivery of nultiple sclerosis treatment Rebif(R). 2009. https://www.science20.com/newswire/merck_serono_launches_rebismarttm_first_electronic_injection_device_delivery_multiple_sclerosis_treatment_rebifr. Accessed October 21, 2022.
Bayer. BETACONNECT™ autoinjector. 2022. https://www.betaseron.com/betaconnect-system/betaconnecttm-electronic-autoinjector. Accessed October 21, 2022.
Merck Serono. Merck Serono launches easypod(R), first electronic growth hormone injection device. 2007. https://www.biospace.com/article/releases/merck-serono-launches-easypod-r-first-electronic-growth-hormone-injection-device. Accessed Oct 21, 2022.
PHC Corporation. PHC and JCR offer growth hormone adherence application for use in clinical research. 2020. https://www.phchd.com/global/phc/news/2020/0915. Accessed Oct 21, 2022.
Devonshire V, Arbizu T, Borre B, et al. Patient-rated suitability of a novel electronic device for self-injection of subcutaneous interferon beta-1a in relapsing multiple sclerosis: an international, single-arm, multicentre. Phase IIIb study BMC Neurol. 2010;10:28.
Edo Solsona MD, Monte Boquet E, Casanova Estruch B, Poveda Andrés JL. Impact of adherence on subcutaneous interferon beta-1a effectiveness administered by Rebismart(®) in patients with multiple sclerosis. Patient Prefer Adherence. 2017;11:415–21.
Domańska B, Stumpp O, Poon S, Oray S, Mountian I, Pichon C. Using patient feedback to optimize the design of a certolizumab pegol electromechanical self-injection device: insights from human factors studies. Adv Ther. 2018;35:100–15.
Pouls BPH, Kristensen LE, Petersson M, et al. A pilot study examining patient preference and satisfaction for ava®, a reusable electronic injection device to administer certolizumab pegol. Expert Opin Drug Deliv. 2020;17:705–11.
Tatla D, Mountian I, Szegvari B, VanLunen B, Schiff M. A multicenter, open-label study to evaluate the safe and effective use of a new electromechanical auto-injection device for self-injection of certolizumab pegol. Expert Opin Drug Deliv. 2020;17:855–62.
Acknowledgements
Funding
The studies described in this manuscript were sponsored by Pfizer; funding for the publication of the manuscript, including the journal’s Rapid Service and Open Access Fees were provided by Pfizer.
Medical Writing Assistance
Medical writing support was provided by David Wateridge, PhD, and Donna McGuire of Engage Scientific Solutions and was funded by Pfizer.
Author Contributions
All authors (Kyle Berman, Simon Moss, Barry Holden-Theunissen, Nobuhiko Satou, Kenji Okada, Mark Latymer, Attila Antalfy) contributed to the conception, design, and development of the product; the conception or design of the studies; and/or the analysis or interpretation of the data. All authors were involved in the writing and revising of the manuscript, and all read and approved the submitted manuscript. All authors had access to the data and take responsibility for the integrity of the data and accuracy of the data analyses.
Disclosures
Kyle Berman, Simon Moss, Barry Holden-Theunissen, Mark Latymer, and Attila Antalfy are employees of Pfizer and hold stock and/or stock options with Pfizer. Nobuhiko Satou and Kenji Okada are employees of PHC Corporation, the manufacturer of the SmartClic/ClicWise device, and hold stock with PHC Corporation.
Compliance with Ethics Guidelines
Research instrument design, participant recruitment, compensation, consent, data handling, data privacy and volunteers’ statements were handled following the standard operating procedures of the companies conducting the research (i.e., Cello Health Insight, Design Science, and Emergo by UL). Participants consented to participate in testing the device and having their data analyzed and published in medical journals. All participants received compensation for their participation within “fair market value” rules.
Data Availability
Upon request, and subject to review, Pfizer will provide the data that support the findings of this study. Subject to certain criteria, conditions, and exceptions, Pfizer may also provide access to the related individual de-identified participant data. See https://www.pfizer.com/science/clinical-trials/trial-data-and-results for more information.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article was published with a multi-page graphical plain language summary. This can be found in the article's supplementary material.
Digital Features
This article is published with digital features, including a multi-page graphical plain language summary, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.22361542. This can also be found in the article's supplementary materials section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Berman, K., Moss, S., Holden-Theunissen, B. et al. Design Development of the SMARTCLIC®/CLICWISE® Injection Device for Self-Administered Subcutaneous Therapies: Findings from Usability and Human Factor Studies. Adv Ther 40, 3070–3086 (2023). https://doi.org/10.1007/s12325-023-02512-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-023-02512-2