Abstract
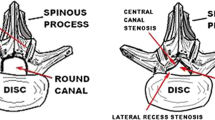
Spinal stenosis is the compression of nerve roots by bone or soft tissue secondary to the narrowing of the spinal canal, lateral recesses, or intervertebral foramina. Spinal stenosis may have acquired or congenital origins. Most cases are acquired and caused by hypertrophy of the ligamentum flavum, enlarged osteophytes, degenerative arthritis, disk herniations, and various systemic illnesses. The ligamentum flavum (LF) is a highly specialized elastic ligament that connects the laminae of the spine and fuses them to the facet joint capsules. There are a number of treatment options available for spinal stenosis. Implants and surgical interventions have grown in popularity recently, and a number of these have been shown to have varying efficacy, including the minimally invasive lumbar decompression (MILD®), Vertiflex®, Coflex® Interlaminar Stabilization, and MinuteMan G3® procedures. Minimally invasive lumbar decompression (MILD®) is a minimally invasive outpatient procedure to treat spinal stenosis related to hypertrophied ligamentum flavum. The Superion® Interspinous Spacer, also known as Vertiflex®, is a titanium implant that is delivered percutaneously to relieve back pain caused by lumbar spinal stenosis. The MinuteMan® is a minimally invasive, interspinous-interlaminar fusion device planned for the temporary fixation of the thoracic, lumbar, and sacral spine, which eventually results in bony fusion. Based on our review of the available current scientific literature, the novel interventions for symptomatic lumbar spinal stenosis, such as the MILD® procedure and the Superion® interspinous spacer, generally appear to be safe and effective. There is a possibility in the future that these interventions could disrupt current treatment algorithms for lumbar spinal stenosis.

Source: Vertos Medical Inc.

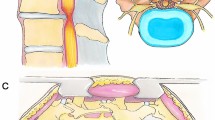
Source: Vertos Medical Inc.



Similar content being viewed by others
References
Andaloro A. Lumbar spinal stenosis. J Am Acad PAs. 2019;32(8):49–50.
Melancia JL, Francisco AF, Antunes JL. Spinal stenosis. In: Handbook of clinical neurology. Elsevier; 2014. p. 541–9. https://linkinghub.elsevier.com/retrieve/pii/B9780702040863000357. Accessed 6 Apr 2021.
Amudong A, Muheremu A, Abudourexiti T. Hypertrophy of the ligamentum flavum and expression of transforming growth factor beta. J Int Med Res. 2017;45(6):2036–41.
Ligamentum Flavum Hypertrophy | Definition, Cause And Treatment. Chiropractic and physiotherapy center combined. 2018. https://www.mychiro.com.my/articles/ligamentum-flavum-hypertrophy/. Accessed 6 Apr 2021.
Sun C, Zhang H, Wang X, Liu X. Ligamentum flavum fibrosis and hypertrophy: molecular pathways, cellular mechanisms, and future directions. FASEB J. 2020;34(8):9854–68.
Hartman J, Granville M, Jacobson RE. The Use of Vertiflex® Interspinous Spacer Device in Patients With Lumbar Spinal Stenosis and Concurrent Medical Comorbidities. Cureus. 11(8). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6786837/. Accessed 6 Apr 2021.
Minuteman for Spinal Stenosis Treatment. https://www.sprmedicine.com/blog/minuteman-for-spinal-stenosis-treatment.html. Accessed 6 Apr 2021.
Mekhail N, Vallejo R, Coleman MH, Benyamin RM. Long-term results of percutaneous lumbar decompression mild(®) for spinal stenosis. Pain Pract Off J World Inst Pain. 2012;12(3):184–93.
Kreiner DS, MacVicar J, Duszynski B, Nampiaparampil DE. The mild® procedure: a systematic review of the current literature. Pain Med Malden Mass. 2014;15(2):196–205.
Diwan S, Deer T. Advanced procedures for pain management a step-by-step atlas. Cham: Springer International Publishing; 2018.
Weinstein JN, Tosteson TD, Lurie JD, Tosteson ANA, Blood E, Hanscom B, et al. Surgical versus nonsurgical therapy for lumbar spinal stenosis. N Engl J Med. 2008;358(8):794–810.
Loguidice V, Bini W, Shabat S, Miller LE, Block JE. Rationale, design and clinical performance of the Superion® Interspinous Spacer: a minimally invasive implant for treatment of lumbar spinal stenosis. Expert Rev Med Devices. 2011;8(4):419–26.
Tekmyster G, Sayed D, Cairns KD, Raso LJ, Kim C, Block JE. Interspinous process decompression with the superion® spacer for lumbar spinal stenosis: real-world experience from a device registry. Med Devices Auckl NZ. 2019;3(12):423–7.
Nunley PD, Deer TR, Benyamin RM, Staats PS, Block JE. Interspinous process decompression is associated with a reduction in opioid analgesia in patients with lumbar spinal stenosis. J Pain Res. 2018;20(11):2943–8.
Bini W, Miller LE, Block JE. Minimally invasive treatment of moderate lumbar spinal stenosis with the superion® interspinous spacer. Open Orthop J. 2011;14(5):361–7.
Minuteman®—SPINEMarketGroup. http://thespinemarketgroup.com/minuteman/. Accessed 6 Apr 2021.
About Spinal Simplicity | Simple Solutions for Complex Spinal Problems. Spinal Simplicity. https://spinalsimplicity.com/about-us/. Accessed 6 Apr 2021.
Minuteman | Spineplus. http://www.spineplus.it/en/chirurgia-vertebrale/minuteman/. Accessed 6 Apr 2021.
Staats PS, Chafin TB, Golovac S, Kim CK, Li S, Richardson WB, et al. Long-term safety and efficacy of minimally invasive lumbar decompression procedure for the treatment of lumbar spinal stenosis with neurogenic claudication: 2-year results of MiDAS ENCORE. Reg Anesth Pain Med. 2018;43(7):789–94.
Benyamin RM, Staats PS, MiDAS EI. MILD® is an effective treatment for lumbar spinal stenosis with neurogenic claudication: MiDAS ENCORE Randomized Controlled Trial. Pain Physician. 2016;19(4):229–42.
Lingreen R, Grider JS. Retrospective review of patient self-reported improvement and post-procedure findings for mild (minimally invasive lumbar decompression). Pain Physician. 2010;13(6):555–60.
Wong WHM. mild Interlaminar decompression for the treatment of lumbar spinal stenosis: procedure description and case series with 1-year follow-up. Clin J Pain. 2012;28(6):534–8.
Brown LL. A double-blind, randomized, prospective study of epidural steroid injection vs the mild® procedure in patients with symptomatic lumbar spinal stenosis. Pain Pract Off J World Inst Pain. 2012;12(5):333–41.
Shabat S, Miller LE, Block JE, Gepstein R. Minimally invasive treatment of lumbar spinal stenosis with a novel interspinous spacer. Clin Interv Aging. 2011;6:227–33.
Nunley PD, Patel VV, Orndorff DG, Lavelle WF, Block JE, Geisler FH. Five-year durability of stand-alone interspinous process decompression for lumbar spinal stenosis. Clin Interv Aging. 2017;12:1409–17.
Diwan S, Sayed D, Deer TR, Salomons A, Liang K. An algorithmic approach to treating lumbar spinal stenosis: an evidenced-based approach. Pain Med Malden Mass. 2019;20(Suppl 2):S23-31.
Musacchio MJ, Lauryssen C, Davis RJ, et al. Evaluation of decompression and interlaminar stabilization compared with decompression and fusion for the treatment of lumbar spinal stenosis: 5-year follow-up of a prospective, randomized, controlled trial. Int J Spine Surg. 2016;10:6. https://doi.org/10.14444/3006.
Schmidt S, Franke J, Rauschmann M, Adelt D, Bonsanto MM, Sola S. Prospective, randomized, multicenter study with 2-year follow-up to compare the performance of decompression with and without interlaminar stabilization. J Neurosurg Spine. 2018;28(4):406–15. https://doi.org/10.3171/2017.11.SPINE17643.
Condez B, Parrish R, Camisa W, Leasure J, Randall J. Stability and decompression mechanics of several mis lumbar fixation technologies: a biomechanical study (2015).
Coflex. Interlaminar Stabilization. 2021. https://coflexsolution.com/clinical-research/.
Acknowledgements
Funding
No funding, medical writing or editorial assistance was used in the construction of this manuscript.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
ADK was involved in editing and the manuscript idea creation. ANE, SNT, AAC, and RJS were involved in the writing of the manuscript. AJK, BMD, MAA, EMC, OV, IU, AKC and AKD were involved in the editing of the manuscript.
Disclosures
Alan D. Kaye, Amber N. Edinoff, Shavona N. Temple, Aaron J. Kaye, Azem A. Chami, Rutvij J. Shah, Bruce M. Dixon, Michael A. Alvarado, Elyse M. Cornett, Omar Viswanath, Ivan Urits and Aaron K. Calodney have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
All data generated or analyzed during this study are included in this published article/as supplementary information files.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaye, A.D., Edinoff, A.N., Temple, S.N. et al. A Comprehensive Review of Novel Interventional Techniques for Chronic Pain: Spinal Stenosis and Degenerative Disc Disease—MILD Percutaneous Image Guided Lumbar Decompression, Vertiflex Interspinous Spacer, MinuteMan G3 Interspinous-Interlaminar Fusion. Adv Ther 38, 4628–4645 (2021). https://doi.org/10.1007/s12325-021-01875-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-021-01875-8