Abstract
Glycemic control is fundamental to the management of diabetes. However, studies suggest that a significant proportion of people with diabetes, particularly those using insulin, are not achieving glycemic targets. The reasons for this are likely to be multifactorial. The real and perceived risk of hypoglycemia and the need for multiple daily injections are widely recognized as key barriers to effective insulin therapy. Therefore, there is a clear unmet need for a treatment option which can help mitigate these barriers. Alternative methods of insulin administration have been under investigation for several years, and pulmonary delivery has shown the most promise to date. Inhaled Technosphere® Insulin (TI; Afrezza®; MannKind Corporation) was approved in 2014 for use as prandial insulin in people with diabetes. TI shows a more rapid onset of action and a significantly faster decline in activity than current subcutaneous rapid-acting insulin analogs (RAAs), and TI is more synchronized to the physiologic timing of the postprandial glucose excursion. This results in lower postprandial hypoglycemia with similar glycemic control compared with RAAs, and less weight gain. Together with the ease of use of the TI inhaler and the reduction in the number of daily injections, these findings imply that TI may be useful in helping to overcome patient resistance to insulin, improve adherence and mitigate clinical inertia in health-care providers, with potential beneficial effects on glycemic control.
Funding: Writing and editorial support in the preparation of this publication was funded by Sanofi US, Inc., Bridgewater, New Jersey, USA. Funding for the article processing charges for this publication was provided by MannKind Corporation.

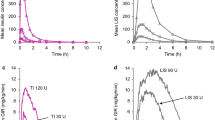
Reproduced with permission from Boss (2012) [46] © Diabetes Technology Society.

Reproduced with permission from Bode (2015) [59] © 2015 by the American Diabetes Association
Similar content being viewed by others
References
CDC National Diabetes Report 2014. http://www.cdc.gov/diabetes/pubs/statsreport14/national-diabetes-report-web.pdf. Accessed 27 Aug 2015.
Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach: update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2015;38:140–9.
Chiang JL, Kirkman MS, Laffel LM, Peters AL; Type 1 Diabetes Sourcebook Authors. Type 1 diabetes through the life span: a position statement of the American Diabetes Association. Diabetes Care. 2014;37:2034–54.
UK Porspective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837–53.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577–89.
Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977–86.
Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. N Engl J Med. 2000;342:381–9.
Martin CL, Albers J, Herman WH, Cleary P, Waberski B, Greene DA, Stevens MJ, Feldman EL; DCCT/EDIC Research Group. Neuropathy among the diabetes control and complications trial cohort 8 years after trial completion. Diabetes Care. 2006;29:340–4.
Stark Casagrande S, Fradkin JE, Saydah SH, Rust KF, Cowie CC. The prevalence of meeting A1C, blood pressure, and LDL goals among people with diabetes, 1988-2010. Diabetes Care. 2013;36:2271–9.
Zhuo X, Zhang P, Kahn HS, Bardenheier BH, Li R, Gregg EW. Change in medical spending attributable to diabetes: national data from 1987 to 2011. Diabetes Care. 2015;38:581–7.
Foos V, Varol N, Curtis BH, Boye KS, Grant D, Palmer JL, McEwan P. Economic impact of severe and non-severe hypoglycemia in patients with Type 1 and Type 2 diabetes in the United States. J Med Econ. 2015;18:420–32.
Rubin RR. Adherence to pharmacologic therapy in patients with type 2 diabetes mellitus. Am J Med. 2005;118(Suppl 5A):27S–34S.
Polonsky WH, Hajos TR, Dain MP, Snoek FJ. Are patients with type 2 diabetes reluctant to start insulin therapy? An examination of the scope and underpinnings of psychological insulin resistance in a large, international population. Curr Med Res Opin. 2011;27:1169–74.
Ross SA, Tildesley HD, Ashkenas J. Barriers to effective insulin treatment: the persistence of poor glycemic control in type 2 diabetes. Curr Med Res Opin. 2011;27(Suppl 3):13–20.
Peyrot M, Barnett AH, Meneghini LF, Schumm-Draeger PM. Factors associated with injection omission/non-adherence in the Global Attitudes of Patients and Physicians in Insulin Therapy study. Diabetes Obes Metab. 2012;14:1081–7.
Alvarez-Guisasola F, Yin DD, Nocea G, Qiu Y, Mavros P. Association of hypoglycemic symptoms with patients’ rating of their health-related quality of life state: a cross sectional study. Health Qual Life Outcomes. 2010;8:86.
Harris S, Mamdani M, Galbo-Jørgensen CB, Bøgelund M, Gundgaard J, Groleau D. The effect of hypoglycemia on health-related quality of life: Canadian results from a multinational time trade-off survey. Can J Diabetes. 2014;38:45–52.
Zoungas S, Patel A, Chalmers J, et al.; ADVANCE Collaborative Group. Severe hypoglycemia and risks of vascular events and death. N Engl J Med. 2010;363:1410–8.
Hsu PF, Sung SH, Cheng HM, et al. Association of clinical symptomatic hypoglycemia with cardiovascular events and total mortality in type 2 diabetes: a nationwide population-based study. Diabetes Care. 2013;36(4):894–900.
Cryer PE. Hypoglycemia in type 1 diabetes mellitus. Endocrinol Metab Clin North Am. 2010;39:641–54.
UK Hypoglycaemia Study Group. Risk of hypoglycaemia in types 1 and 2 diabetes: effects of treatment modalities and their duration. Diabetologia. 2007;50:1140–7.
Russell-Jones D, Khan R. Insulin-associated weight gain in diabetes—causes, effects and coping strategies. Diabetes Obes Metab. 2007;9:799–812.
Holman RR, Thorne KI, Farmer AJ, et al.; 4-T Study Group. Addition of biphasic, prandial, or basal insulin to oral therapy in type 2 diabetes. N Engl J Med. 2007;357:1716–30.
Ridderstråle M, Gudbjörnsdottir S, Eliasson B, Nilsson PM, Cederholm J; Steering Committee of the Swedish National Diabetes Register (NDR). Obesity and cardiovascular risk factors in type 2 diabetes: results from the Swedish National Diabetes Register. J Intern Med. 2006;259:314–22.
Snoek F, Skovlund SE, Pouwer F. Development and validation of the insulin treatment appraisal scale (ITAS) in patients with type 2 diabetes. BMC Health Qual Life Outcomes. 2007;5:69.
Purnell JQ, Zinman B, Brunzell JD; DCCT/EDIC Research Group. The effect of excess weight gain with intensive diabetes mellitus treatment on cardiovascular disease risk factors and atherosclerosis in type 1 diabetes mellitus: results from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study (DCCT/EDIC) study. Circulation. 2013;127:180–7.
Larkin ME, Capasso VA, Chen CL, et al. Measuring psychological insulin resistance: barriers to insulin use. Diabetes Educ. 2008;34:511–7.
Rubin RR, Peyrot M, Kruger DF, Travis LB. Barriers to insulin injection therapy: patient and health care provider perspectives. Diabetes Educ. 2009;35:1014–22.
Peyrot M, Rubin RR, Kruger DF, Travis LB. Correlates of insulin injection omission. Diabetes Care. 2010;33:240–5.
Davies MJ, Gagliardino JJ, Gray LJ, Khunti K, Mohan V, Hughes R. Real-world factors affecting adherence to insulin therapy in patients with Type 1 or Type 2 diabetes mellitus: a systematic review. Diabet Med. 2013;30:512–24.
Abu Hassan H, Tohid H, Mohd Amin R, Long Bidin MB, Muthupalaniappen L, Omar K. Factors influencing insulin acceptance among type 2 diabetes mellitus patients in a primary care clinic: a qualitative exploration. BMC Fam Pract. 2013;14:164.
Ignaut DA, Schwartz SL, Sarwat S, Murphy HL. Comparative device assessments: Humalog KwikPen compared with vial and syringe and FlexPen. Diabetes Educ. 2009;35:789–98.
Anderson BJ, Redondo MJ. What can we learn from patient-reported outcomes of insulin pen devices? J Diabetes Sci Technol. 2011;5:1563–71.
Ayyagari R, Wei W, Cheng D, Pan C, Wu E. Effects of insulin adherence and delivery device on real world outcomes among patients with type 2 diabetes mellitus (T2DM). Diabetes. 2013;62(Suppl 1):A327.
Berard L, Cameron B, Woo V. Pen needle preference in a population of Canadians with diabetes: results from a recent patient survey. Can J Diabetes. 2015;39:206–9.
Kruger DF, LaRue S, Estepa P. Recognition of and steps to mitigate anxiety and fear of pain in injectable diabetes treatment. Diabetes Metab Syndr Obes. 2015;8:49–56.
Hauber AB, Johnson FR, Sauriol L, Lescrauwaet B. Risking health to avoid injections: preferences of Canadians with type 2 diabetes. Diabetes Care. 2005;28:2243–5.
Boss AH, Yu W, Ellerman K. Prandial insulin: is inhaled enough? Drug Dev Res. 2008;69:138–42.
Heise T, Meneghini LF. Insulin stacking versus therapeutic accumulation: understanding the differences. Endocr Pract. 2014;20:75–83.
Silverman BL, Barnes CJ, Campaigne BN, Muchmore DB. Inhaled insulin for controlling blood glucose in patients with diabetes. Vasc Health Risk Manag. 2007;3:947–58.
Cefalu WT. Concept, strategies, and feasibility of noninvasive insulin delivery. Diabetes Care. 2004;27:239–46.
Patton JS, Byron PR. Inhaling medicines: delivering drugs to the body through the lungs. Nat Rev Drug Discov. 2007;6:67–74.
Mastrandrea LD. Inhaled insulin: overview of a novel route of insulin administration. Vasc Health Risk Manag. 2010;6:47–58.
Patton JS, Bukar JG, Eldon MA. Clinical pharmacokinetics and pharmacodynamics of inhaled insulin. Clin Pharmacokinet. 2004;43:781–801.
Heinemann L. New ways of insulin delivery. Int J Clin Pract Suppl. 2011;(170):31–46.
Boss AH, Petrucci R, Lorber D. Coverage of prandial insulin requirements by means of an ultra-rapid-acting inhaled insulin. J Diabetes Sci Technol. 2012;6:773–9.
Ceglia L, Lau J, Pittas AG. Meta-analysis: efficacy and safety of inhaled insulin therapy in adults with diabetes mellitus. Ann Intern Med. 2006;145:665–75.
Skyler JS, Jovanovic L, Klioze S, Reis J, Duggan W; Inhaled Human Insulin Type 1 Diabetes Study Group. Two-year safety and efficacy of inhaled human insulin (Exubera) in adult patients with type 1 diabetes. Diabetes Care. 2007;30:579–85.
Raskin P, Heller S, Honka M, et al. Pulmonary function over 2 years in diabetic patients treated with prandial inhaled Technosphere Insulin or usual antidiabetes treatment: a randomized trial. Diabetes Obes Metab. 2012;14:163–73.
FDA Med Watch: Exubera (insulin human [rDNA origin]) Inhalation Powder. September 2008: http://www.fda.gov/Safety/MedWatch/SafetyInformation/Safety-RelatedDrugLabelingChanges/ucm122978.htm. Accessed 16 May 2016.
Setji TL, Hong BD, Feinglos MN. Technosphere insulin: inhaled prandial insulin. Expert Opin Biol Ther. 2016;16:111–7.
Black C, Cummins E, Royle P, Philip S, Waugh N. The clinical effectiveness and cost-effectiveness of inhaled insulin in diabetes mellitus: a systematic review and economic evaluation. Health Technol Assess. 2007;11:1–126.
Sarala N, Bengalorkar G, Bhwana K. Technosphere: new drug delivery system for inhaled insulin. Future Prescriber. 2012;13:14–6.
Santos Cavaiola T, Edelman S. Inhaled insulin: a breath of fresh air? A review of inhaled insulin. Clin Ther. 2014;36:1275–89.
Potocka E, Hovorka R, Baughman RA, et al. AFRESA™ suppresses endogenous glucose production earlier than a rapid-acting analog (lispro) and inhaled Exubera®. Diabetes. 2009;58(Suppl 1):abstract 232-OR.
Rave K, Heise T, Pfützner A, Boss AH. Coverage of postprandial blood glucose excursions with inhaled technosphere insulin in comparison to subcutaneously injected regular human insulin in subjects with type 2 diabetes. Diabetes Care. 2007;30:2307–8.
Kapsner P, Bergenstal R, Rendell M, et al. Comparative efficacy and safety of technosphere insulin and a rapid-acting analogue both given with glargine in subjects with type 1 diabetes mellitus in a 52-week study. Diabetologia. 2009;52(Suppl 1):S386.
Rosenstock J, Lorber DL, Gnudi L, et al. Prandial inhaled insulin plus basal insulin glargine versus twice daily biaspart insulin for type 2 diabetes: a multicentre randomised trial. Lancet. 2010;375:2244–53.
Bode BW, McGill JB, Lorber DL, Gross JL, Chang PC, Bregman DB; Affinity 1 Study Group. Inhaled Technosphere insulin compared with injected prandial insulin in type 1 diabetes: a randomized 24-week trial. Diabetes Care. 2015;38:2266–73.
Zisser H, Dassau E, Lee JJ, Harvey RA, Bevier W, Doyle FJ 3rd. Clinical results of an automated artificial pancreas using technosphere inhaled insulin to mimic first-phase insulin secretion. J Diabetes Sci Technol. 2015;9:564–72.
Woerle HJ, Neumann C, Zschau S, et al. Impact of fasting and postprandial glycemia on overall glycemic control in type 2 diabetes Importance of postprandial glycemia to achieve target HbA1c levels. Diabetes Res Clin Pract. 2007;77:280–5.
Rosenstock J, Bergenstal R, Defronzo RA, et al. Efficacy and safety of Technosphere inhaled insulin compared with Technosphere powder placebo in insulin-naive type 2 diabetes suboptimally controlled with oral agents. Diabetes Care. 2008;31:2177–82.
Rosenstock J, Franco D, Korpachev V, et al. Inhaled Technosphere insulin versus inhaled technosphere placebo in insulin-naïve subjects with type 2 diabetes inadequately controlled on oral antidiabetes agents. Diabetes Care. 2015;38:2274–81.
Peyrot M, Rubin RR. Effect of technosphere inhaled insulin on quality of life and treatment satisfaction. Diabetes Technol Ther. 2010;12:49–55.
Peyrot M, Rubin RR. Patient reported outcomes in adults with type 2 diabetes using mealtime AFRESA (inhaled Technosphere insulin) or metformin + secretagogue or both. Presented at: 69th American Diabetes Association Scientific Sessions, June 5, 2009, New Orleans, LA. Poster 552-P.
Peyrot M, Rubin RR. Patient-reported outcomes in adults with type 2 diabetes using mealtime inhaled technosphere insulin and basal insulin versus premixed insulin. Diabetes Technol Ther. 2011;13:1201–6.
Afrezza Prescribing Information. Revised 07/2015. http://products.sanofi.us/afrezza/afrezza.html. Accessed 27 Aug 2015.
Acknowledgments
Writing and editorial support in the preparation of this publication was provided by Katherine Roberts, PhD, of Excerpta Medica, and funded by Sanofi US, Inc., Bridgewater, New Jersey, USA. The authors, individually and collectively, were responsible for all content and editorial decisions and received no payment from Sanofi directly or indirectly (through a third party) related to the development of this publication. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole and have given final approval for the version to be published. Technosphere and Afrezza are registered trademarks of MannKind Corporation. Funding for the article processing charges for this publication was provided by MannKind Corporation.
Disclosures
Janet B. McGill has received Grants to Washington University from Dexcom, Intarcia, Novartis, Novo Nordisk, Pfizer, and Lexicon; provides advising/consulting services to Boehringer Ingelheim/Lilly, Merck, Novo Nordisk, Janssen; and is on the speakers bureau for Janssen. Steven V. Edelman is a consultant for Sanofi, Lilly and Novo Nordisk. Tricia Santos Cavaiola provides advising/consulting services for Dexcom and is on the speakers bureau for Valeritas. David Ahn and C. Rachel Kilpatrick have nothing to disclose.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/5ED4F0604F3D80A5.
Rights and permissions
About this article
Cite this article
McGill, J.B., Ahn, D., Edelman, S.V. et al. Making Insulin Accessible: Does Inhaled Insulin Fill an Unmet Need?. Adv Ther 33, 1267–1278 (2016). https://doi.org/10.1007/s12325-016-0370-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-016-0370-1