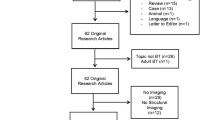
Abstract
Medulloblastoma is a malign posterior fossa brain tumor, mostly occurring in childhood. The CNS-directed chemoradiotherapy treatment can be very harmful to the developing brain and functional outcomes of these patients. However, what the underlying neurotoxic mechanisms are remain inconclusive. Hence, this review summarizes the existing literature on the association between advanced neuroimaging and neurocognitive changes in patients that were treated for pediatric medulloblastoma. The PubMed/Medline database was extensively screened for studies investigating the link between cognitive outcomes and multimodal magnetic resonance (MR) imaging in childhood medulloblastoma survivors. A behavioral meta-analysis was performed on the available IQ scores. A total of 649 studies were screened, of which 22 studies were included. Based on this literature review, we conclude medulloblastoma patients to be at risk for white matter volume loss, more frequent white matter lesions, and changes in white matter microstructure. Such microstructural alterations were associated with lower IQ, which reached the clinical cut-off in survivors across studies. Using functional MR scans, changes in activity were observed in cerebellar areas, associated with working memory and processing speed. Finally, cerebral microbleeds were encountered more often, but these were not associated with cognitive outcomes. Regarding intervention studies, computerized cognitive training was associated with changes in prefrontal and cerebellar activation and physical training might result in microstructural and cortical alterations. Hence, to better define the neural targets for interventions in pediatric medulloblastoma patients, this review suggests working towards neuroimaging-based predictions of cognitive outcomes. To reach this goal, large multimodal prospective imaging studies are highly recommended.



Similar content being viewed by others
References
Millard NE, De Braganca KC. Medulloblastoma. J Child Neurol. 2016;31:1341–53.
Komori T. The 2016 WHO Classification of Tumours of the Central Nervous System: the major points of revision. Neurol Med Chir (Tokyo). 2017;57:301–11.
Lynch CF, Hart MN, Jones MP. Medulloblastoma: a population-based study of 532 cases. J Neuropathol Exp Neurol. 1991:134–44.
Ostrom QT, De Blank PM, Kruchko C, Petersen CM, Liao P, Finlay JL, et al. Alex’s lemonade stand foundation infant and childhood primary brain and central nervous system tumors diagnosed in the United States in 2007-2011. Neuro-Oncology. 2015;16:x1–35.
McKean-Cowdin R, Razavi P, Barrington-Trimis J, Baldwin RT, Asgharzadeh S, Cockburn M, et al. Trends in childhood brain tumor incidence, 1973-2009. J Neuro-Oncol. 2013.
Stiller CA, Bayne AM, Chakrabarty A, Kenny T, Chumas P. Incidence of childhood CNS tumours in Britain and variation in rates by definition of malignant behaviour: population-based study. BMC Cancer. 2019;19:139.
Weil AG, Wang AC, Westwick HJ, Ibrahim GM, Ariani RT, Crevier L, et al. Survival in pediatric medulloblastoma: a population-based observational study to improve prognostication. J Neuro-Oncol Springer US. 2017;132:99–107.
Dressler EV, Dolecek TA, Liu M, Villano JL. Demographics, patterns of care, and survival in pediatric medulloblastoma. J Neuro-Oncol. United States. 2017;132:497–506.
Huang PI, Lin SC, Lee YY, Ho DMT, Guo WY, Chang KP, et al. Large cell/anaplastic medulloblastoma is associated with poor prognosis—a retrospective analysis at a single institute. Childs Nerv Syst. 2017;33:1285–94.
Northcott PA, Korshunov A, Witt H, Hielscher T, Eberhart CG, Mack S, et al. Medulloblastoma comprises four distinct molecular variants. J Clin Oncol. 2011;29:1408–14.
Taylor MD, Northcott PA, Korshunov A, Remke M, Cho YJ, Clifford SC, et al. Molecular subgroups of medulloblastoma: the current consensus. Acta Neuropathol. 2012;123:465–72.
Packer RJ, Gajjar A, Vezina G, Rorke-Adams L, Burger PC, Robertson PL, et al. Phase III study of craniospinal radiation therapy followed by adjuvant chemotherapy for newly diagnosed average-risk medulloblastoma. J Clin Oncol. 2006;24:4202–8.
Von Hoff K, Rutkowski S. Medulloblastoma. Curr Treat Options Neurol. 2012;14:416–26.
Packer RJ, Goldwein J, Nicholson HS, Vezina LG, Allen JC, Ris MD, et al. Treatment of children with medulloblastomas with reduced-dose craniospinal radiation therapy and adjuvant chemotherapy: a Children’s Cancer Group Study. J Clin Oncol. United States. 1999;17:2127–36.
Packer RJ, Zhou T, Holmes E, Vezina G, Gajjar A. Survival and secondary tumors in children with medulloblastoma receiving radiotherapy and adjuvant chemotherapy: results of Children’s Oncology Group trial A9961. Neuro-Oncology. England. 2013;15:97–103.
Srinivasan VM, Ghali MGZ, North RY, Boghani Z, Hansen D, Lam S. Modern management of medulloblastoma: molecular classification, outcomes, and the role of surgery. Surg Neurol Int India. 2016;7:S1135–41.
Rutkowski S, Cohen B, Finlay J, Luksch R, Ridola V, Valteau-Couanet D, et al. Medulloblastoma in young children. Pediatr Blood Cancer. United States. 2010;54:635–7.
Khong P-L, Leung LHT, Chan GCF, Kwong DLW, Wong WHS, Cao G, et al. White matter anisotropy in childhood medulloblastoma survivors: association with neurotoxicity risk factors. Radiology United States. 2005;236:647–52.
Rutkowski S, Bode U, Deinlein F, Ottensmeier H, Warmuth-Metz M, Soerensen N, et al. Treatment of early childhood medulloblastoma by postoperative chemotherapy alone. N Engl J Med. 2005;352:978–86.
Rutkowski S, Gerber NU, von Hoff K, Gnekow A, Bode U, Graf N, et al. Treatment of early childhood medulloblastoma by postoperative chemotherapy and deferred radiotherapy. Neuro-Oncology. England. 2009;11:201–10.
von Bueren AO, von Hoff K, Pietsch T, Gerber NU, Warmuth-Metz M, Deinlein F, et al. Treatment of young children with localized medulloblastoma by chemotherapy alone: results of the prospective, multicenter trial HIT 2000 confirming the prognostic impact of histology. Neuro-Oncology. England. 2011;13:669–79.
Chi SN, Gardner SL, Levy AS, Knopp EA, Miller DC, Wisoff JH, et al. Feasibility and response to induction chemotherapy intensified with high-dose methotrexate for young children with newly diagnosed high-risk disseminated medulloblastoma. J Clin Oncol. 2004;22:4881–7.
Mason WP, Grovas A, Halpern S, Dunkel IJ, Garvin J, Heller G, et al. Intensive chemotherapy and bone marrow rescue for young children with newly diagnosed malignant brain tumors. J Clin Oncol. United States. 1998;16:210–21.
Lafay-Cousin L, Smith A, Chi SN, Wells E, Madden J, Margol A, et al. Clinical, pathological, and molecular characterization of infant medulloblastomas treated with sequential high-dose chemotherapy. Pediatr Blood Cancer. United States. 2016;63:1527–34.
Ashley DM, Merchant TE, Strother D, Zhou T, Duffner P, Burger PC, et al. Induction chemotherapy and conformal radiation therapy for very young children with nonmetastatic medulloblastoma: Children’s Oncology Group study P9934. J Clin Oncol. United States. 2012;30:3181–6.
King AA, Seidel K, Di C, Leisenring WM, Perkins SM, Krull KR, et al. Long-term neurologic health and psychosocial function of adult survivors of childhood medulloblastoma/PNET: a report from the Childhood Cancer Survivor Study. Neuro-Oncology. England. 2017;19:689–98.
Wells EM, Ullrich NJ, Seidel K, Leisenring W, Sklar CA, Armstrong GT, et al. Longitudinal assessment of late-onset neurologic conditions in survivors of childhood central nervous system tumors: a Childhood Cancer Survivor Study report. Neuro-Oncology England. 2018;20:132–42.
Mabbott DJD, Penkman L, Witol A, Strother D, Bouffet E. Core neurocognitive functions in children treated for posterior fossa tumors. Neuropsychology United States. 2008;22:159–68.
Mulhern RK, Palmer SL, Merchant TE, Wallace D, Kocak M, Brouwers P, et al. Neurocognitive consequences of risk-adapted therapy for childhood medulloblastoma. J Clin Oncol. United States. 2005;23:5511–9.
Palmer SL, Goloubeva O, Reddick WE, Glass JO, Gajjar A, Kun L, et al. Patterns of intellectual development among survivors of pediatric medulloblastoma: a longitudinal analysis. J Clin Oncol United States. 2001;19:2302–8.
Saury J-MG, Emanuelson I. Cognitive consequences of the treatment of medulloblastoma among children. Pediatr Neurol United States. 2011;44:21–30.
Palmer SL, Armstrong C, Onar-Thomas A, Wu S, Wallace D, Bonner MJ, et al. Processing speed, attention, and working memory after treatment for medulloblastoma: an international, prospective, and longitudinal study. J Clin Oncol. United States. 2013;31:3494–500.
Fry AF, Hale S. Relationships among processing speed, working memory, and fluid intelligence in children. Biol Psychol. 2000;54:1–34.
King TZ, Ailion AS, Fox ME, Hufstetler SM. Neurodevelopmental model of long-term outcomes of adult survivors of childhood brain tumors. Child Neuropsychol Routledge. 2017;00:1–21.
Palmer SL. Neurodevelopmental impact on children treated for medulloblastoma: a review and proposed conceptual model. Dev Disabil Res Rev United States. 2008;14:203–10.
Wolfe KR, Madan-Swain A, Kana RK. Executive dysfunction in pediatric posterior fossa tumor survivors: a systematic literature review of neurocognitive deficits and interventions. Dev Neuropsychol England. 2012;37:153–75.
Hoang DH, Pagnier A, Guichardet K, Dubois-Teklali F, Schiff I, Lyard G, et al. Cognitive disorders in pediatric medulloblastoma: what neuroimaging has to offer. J Neurosurg Pediatr. United States. 2014;14:136–44.
Moxon-Emre I, Bouffet E, Taylor MD, Laperriere N, Sharpe MB, Laughlin S, et al. Vulnerability of white matter to insult during childhood: evidence from patients treated for medulloblastoma. J Neurosurg Pediatr United States. 2016;18:29–40.
Thust SC, Blanco E, Michalski AJ, Chong WK, Gaze MN, Phipps K, et al. MRI abnormalities in children following sequential chemotherapy, hyperfractionated accelerated radiotherapy and high-dose thiotepa for high-risk primitive neuroectodermal tumours of the central nervous system. J Med Imaging Radiat Oncol Australia. 2014;58:683–90.
Uh J, Merchant TE, Li Y, Feng T, Gajjar A, Ogg RJ, et al. Differences in brainstem fiber tract response to radiation: a longitudinal diffusion tensor imaging study. Int J Radiat Oncol Biol Phys. United States. 2013;86:292–7.
Reddick WE, Russell JM, Glass JO, Xiong X, Mulhern RK, Langston JW, et al. Subtle white matter volume differences in children treated for medulloblastoma with conventional or reduced dose craniospinal irradiation. Magn Reson Imaging. 2000;18:787–93.
Duncan EC, Reddick WE, Glass JO, Hyun JW, Ji Q, Li Y, et al. Application of probabilistic fiber-tracking method of MR imaging to measure impact of cranial irradiation on structural brain connectivity in children treated for medulloblastoma. Med Imaging 2016 Biomed Appl Mol Struct Funct Imaging. 2016.
Nagtegaal SHJ, David S, van der Boog ATJ, Leemans A, Verhoeff JJC. Changes in cortical thickness and volume after cranial radiation treatment: a systematic review. Radiother Oncol. 2019;135:33–42.
Riggs L, Bouffet E, Laughlin S, Laperriere N, Liu F, Skocic J, et al. Changes to memory structures in children treated for posterior fossa tumors. J Int Neuropsychol Soc. 2014;20:168–80.
Palmer SL, Reddick WE, Glass JO, Ogg R, Patay Z, Wallace D, et al. Regional white matter anisotropy and reading ability in patients treated for pediatric embryonal tumors. Brain Imaging Behav United States. 2010;4:132–40.
Palmer SL, Glass JO, Li Y, Ogg R, Qaddoumi I, Armstrong GT, et al. White matter integrity is associated with cognitive processing in patients treated for a posterior fossa brain tumor. Neuro-Oncology. England. 2012;14:1185–93.
Mulhern RK, Reddick WE, Palmer SL, Glass JO, Elkin TD, Kun LE, et al. Neurocognitive deficits in medulloblastoma survivors and white matter loss. Ann Neurol United States. 1999;46:834–41.
Shan ZY, Liu JZ, Glass JO, Gajjar A, Li C-S, Reddick WE. Quantitative morphologic evaluation of white matter in survivors of childhood medulloblastoma. Magn Reson Imaging Netherlands. 2006;24:1015–22.
Riva D, Giorgi C, Nichelli F, Bulgheroni S, Massimino M, Cefalo G, et al. Intrathecal methotrexate affects cognitive function in children with medulloblastoma. Neurology United States. 2002;59:48–53.
Fouladi M, Chintagumpala M, Laningham FH, Ashley D, Kellie SJ, Langston JW, et al. White matter lesions detected by magnetic resonance imaging after radiotherapy and high-dose chemotherapy in children with medulloblastoma or primitive neuroectodermal tumor. J Clin Oncol. United States. 2004;22:4551–60.
Khajuria RK, Blankenburg F, Wuithschick I, Rueckriegel S, Thomale U-W, Mansour M, et al. Morphological brain lesions of pediatric cerebellar tumor survivors correlate with inferior neurocognitive function but do not affect health-related quality of life. Childs Nerv Syst Germany. 2015;31:569–80.
Glass JO, Ogg RJ, Hyun JW, Harreld JH, Schreiber JE, Palmer SL, et al. Disrupted development and integrity of frontal white matter in patients treated for pediatric medulloblastoma. Neuro-Oncology. 2017;19:1408–18.
Decker AL, Szulc KU, Bouffet E, Laughlin S, Chakravarty MM, Skocic J, et al. Smaller hippocampal subfield volumes predict verbal associative memory in pediatric brain tumor survivors. Hippocampus United States. 2017;27:1140–54.
Szychot E, Seunarine K, Mankad K, Thust S, Clark C, Gaze MN, et al. Impact of induction chemotherapy, hyperfractionated accelerated radiotherapy and high-dose thiotepa on brain volume loss and functional status of children with primitive neuroectodermal tumour. Pediatr Blood Cancer United States. 2017;64.
Hoang DH, Pagnier A, Cousin E, Guichardet K, Schiff I, Icher C, et al. Anatomo-functional study of the cerebellum in working memory in children treated for medulloblastoma. J Neuroradiol Elsevier Masson SAS. 2019;46:207–13.
Aukema EJ, Caan MWA, Oudhuis N, Majoie CBLM, Vos FM, Reneman L, et al. White matter fractional anisotropy correlates with speed of processing and motor speed in young childhood cancer survivors. Int J Radiat Oncol Biol Phys. 2009;74:837–43.
Khong P-L, Leung LHT, Fung ASM, Fong DYT, Qiu D, Kwong DLW, et al. White matter anisotropy in post-treatment childhood cancer survivors: preliminary evidence of association with neurocognitive function. J Clin Oncol. United States. 2006;24:884–90.
Mabbott DJ, Noseworthy MD, Bouffet E, Rockel C, Laughlin S. Diffusion tensor imaging of white matter after cranial radiation in children for medulloblastoma: correlation with IQ. Neuro-Oncology. 2006;8:244–52.
Khong P-L, Kwong DLW, Chan GCF, Sham JST, Chan F-L, Ooi G-C. Diffusion-tensor imaging for the detection and quantification of treatment-induced white matter injury in children with medulloblastoma: a pilot study. AJNR Am J Neuroradiol United States. 2003;24:734–40.
Rueckriegel SM, Bruhn H, Thomale UW, Hernaiz Driever P. Cerebral white matter fractional anisotropy and tract volume as measured by MR imaging are associated with impaired cognitive and motor function in pediatric posterior fossa tumor survivors. Pediatr Blood Cancer. United States. 2015;62:1252–8.
Law N, Smith ML, Greenberg M, Bouffet E, Taylor MD, Laughlin S, et al. Executive function in paediatric medulloblastoma: the role of cerebrocerebellar connections. J Neuropsychol England. 2017;11:174–200.
Li MD, Forkert ND, Kundu P, Ambler C, Lober RM, Burns TC, et al. Brain perfusion and diffusion abnormalities in children treated for posterior fossa brain tumors. J Pediatr. 2017;185:173–180.e3.
Brinkman TM, Reddick WE, Luxton J, Glass JO, Sabin ND, Srivastava DK, et al. Cerebral white matter integrity and executive function in adult survivors of childhood medulloblastoma. Neuro-Oncology. England. 2012;14(Suppl 4):iv25–36.
Yeom KW, Lober RM, Partap S, Telischak N, Tsolinas R, Barnes PD, et al. Increased focal hemosiderin deposition in pediatric medulloblastoma patients receiving radiotherapy at a later age: clinical article. J Neurosurg Pediatr. 2013;12:444–51.
Zou P, Conklin HM, Scoggins MA, Li Y, Li X, Jones MM, et al. Functional MRI in medulloblastoma survivors supports prophylactic reading intervention during tumor treatment. Brain Imaging Behav. 2016;10:258–71.
Kates R, Atkinson D, Brant-Zawadzki M. Fluid-attenuated inversion recovery (FLAIR): clinical prospectus of current and future applications. Top Magn Reson Imaging United States. 1996;8:389–96.
Mulhern RK, Palmer SL, Reddick WE, Glass JO, Kun LE, Taylor J, et al. Risks of young age for selected neurocognitive deficits in medulloblastoma are associated with white matter loss. J Clin Oncol. United States. 2001;19:472–9.
Debnam JM, Schellingerhout D. Diffusion MR imaging of the brain in patients with cancer. Int J Mol Imaging. 2011;2011:714021.
Sener RN. Diffusion MRI: apparent diffusion coefficient (ADC) values in the normal brain and a classification of brain disorders based on ADC values. Comput Med Imaging Graph United States. 2001;25:299–326.
Ravn S, Holmberg M, Sorensen P, Frokjaer JB, Carl J. Differences in supratentorial white matter diffusion after radiotherapy--new biomarker of normal brain tissue damage? Acta Oncol. England. 2013;52:1314–9.
Hua C, Merchant TE, Gajjar A, Broniscer A, Zhang Y, Li Y, et al. Brain tumor therapy-induced changes in normal-appearing brainstem measured with longitudinal diffusion tensor imaging. Int J Radiat Oncol Biol Phys. United States. 2012;82:2047–54.
Leung LHT, Ooi GC, Kwong DLW, Chan GCF, Cao G, Khong PL. White-matter diffusion anisotropy after chemo-irradiation: a statistical parametric mapping study and histogram analysis. Neuroimage. 2004;21:261–8.
Qiu D, Kwong DLW, Chan GCF, Leung LHT, Khong PL. Diffusion tensor magnetic resonance imaging finding of discrepant fractional anisotropy between the frontal and parietal lobes after whole-brain irradiation in childhood medulloblastoma survivors: reflection of regional white matter radiosensitivity? Int J Radiat Oncol Biol Phys. 2007;69:846–51.
Rueckriegel SM, Driever PH, Blankenburg F, Lüdemann L, Henze G, Bruhn H. Differences in Supratentorial damage of white matter in pediatric survivors of posterior fossa tumors with and without adjuvant treatment as detected by magnetic resonance diffusion tensor imaging. Int J Radiat Oncol Biol Phys. 2010;76:859–66.
Lee MH, Smyser CD, Shimony JS. Resting-state fMRI: a review of methods and clinical applications. AJNR Am J Neuroradiol. United States. 2013;34:1866–72.
Ogawa S, Lee TM, Kay AR, Tank DW. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. Proc Natl Acad Sci U S A. United States. 1990;87:9868–72.
Hernandez-Garcia L, Lahiri A, Schollenberger J. Recent progress in ASL. Neuroimage. United States. 2018.
Passos J, Nzwalo H, Valente M, Marques J, Azevedo A, Netto E, et al. Microbleeds and cavernomas after radiotherapy for paediatric primary brain tumours. J Neurol Sci. 2017;372:413–6.
Miura M, Nakajima M, Fujimoto A, Kaku Y, Kawano T, Watanabe M, et al. High prevalence of small vessel disease long after cranial irradiation. J Clin Neurosci. Scotland. 2017;46:129–35.
Humphries TJ, Mathew P. Cerebral microbleeds: hearing through the silence – a narrative review. Curr Med Res Opin. Taylor & Francis. 2018;0:1–15.
Passos J, Nzwalo H, Marques J, Azevedo A, Netto E, Nunes S, et al. Late cerebrovascular complications after radiotherapy for childhood primary central nervous system tumors. Pediatr Neurol. 2015;53:211–5.
Greenberg SM, Vernooij MW, Cordonnier C, Viswanathan A, Al-Shahi Salman R, Warach S, et al. Cerebral microbleeds: a guide to detection and interpretation. Lancet Neurol Elsevier Ltd. 2009;8:165–74.
Tsui Y-K, Tsai FY, Hasso AN, Greensite F, Nguyen BV. Susceptibility-weighted imaging for differential diagnosis of cerebral vascular pathology: a pictorial review. J Neurol Sci. Netherlands. 2009;287:7–16.
Palmer SL, Reddick WE, Glass JO, Gajjar A, Goloubeva O, Mulhern RK. Decline in corpus callosum volume among pediatric patients with medulloblastoma: longitudinal MR imaging study. AJNR Am J Neuroradiol. 2002;23:1088–94.
Othman RT, Abdullah KG. Serial MRI scan of posterior fossa tumours predict patients at risk of developing neurocognitive impairment. Asian Pac J Cancer Prev. 2017.
Papathanasiou A, Messinis L, Zampakis P, Papathanasopoulos P. Corpus callosum atrophy as a marker of clinically meaningful cognitive decline in secondary progressive multiple sclerosis. Impact on employment status. J Clin Neurosci Off J Neurosurg Soc Australas. Scotland. 2017;43:170–5.
Manca R, Sharrack B, Paling D, Wilkinson ID, Venneri A. Brain connectivity and cognitive processing speed in multiple sclerosis: a systematic review. J Neurol Sci Netherlands. 2018;388:115–27.
Owens JA, Spitz G, Ponsford JL, Dymowski AR, Willmott C. An investigation of white matter integrity and attention deficits following traumatic brain injury. Brain Inj England. 2018;32:776–83.
Nagel BJ, Palmer SL, Reddick WE, Glass JO, Helton KJ, Wu S, et al. Abnormal hippocampal development in children with medulloblastoma treated with risk-adapted irradiation. AJNR Am J Neuroradiol. 2004;25:1575–82.
Liu AK, Marcus KJ, Fischl B, Grant PE, Young Poussaint T, Rivkin MJ, et al. Changes in cerebral cortex of children treated for medulloblastoma. Int J Radiat Oncol Biol Phys. 2007.
Palmer SL, Leigh L, Ellison SC, Onar-Thomas A, Wu S, Qaddoumi I, et al. Feasibility and efficacy of a computer-based intervention aimed at preventing reading decoding deficits among children undergoing active treatment for medulloblastoma: results of a randomized trial. J Pediatr Psychol United States. 2014;39:450–8.
Szulc-Lerch KU, Timmons BW, Bouffet E, Laughlin S, de Medeiros CB, Skocic J, et al. Repairing the brain with physical exercise: cortical thickness and brain volume increases in long-term pediatric brain tumor survivors in response to a structured exercise intervention. NeuroImage Clin. 2018;18:972–85.
Riggs L, Piscione J, Laughlin S, Cunningham T, Timmons BW, Courneya KS, et al. Exercise training for neural recovery in a restricted sample of pediatric brain tumor survivors: a controlled clinical trial with crossover of training versus no training. Neuro-Oncology. England. 2017;19:440–50.
Conklin HM, Ogg RJ, Ashford JM, Scoggins MA, Zou P, Clark KN, et al. Computerized cognitive training for amelioration of cognitive late effects among childhood Cancer survivors: a randomized controlled trial. J Clin Oncol. United States. 2015;33:3894–902.
Neu MA, Tanyildizi Y, Wingerter A, Henninger N, El Malki K, Alt F, et al. Susceptibility-weighted magnetic resonance imaging of cerebrovascular sequelae after radiotherapy for pediatric brain tumors. Radiother Oncol Ireland. 2018;127:280–6.
Nieman BJ, Elizabeth De Guzman A, Gazdzinski LM, Lerch JP, Mallar Chakravarty M, Pipitone J, et al. White and gray matter abnormalities after cranial radiation in children and mice. Int J Radiat Oncol Biol Phys. 2015.
Funding
Kinderkankerfonds Leuven
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wauters, M., Uyttebroeck, A., De Waele, L. et al. Neuroimaging Biomarkers and Neurocognitive Outcomes in Pediatric Medulloblastoma Patients: a Systematic Review. Cerebellum 20, 462–480 (2021). https://doi.org/10.1007/s12311-020-01225-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-020-01225-4