Abstract
Qualitative platelet function defects may be congenital, e.g., Glanzmann thrombasthenia, or acquired. Acquired platelet function disorders are more common than reported. Glanzmann thrombasthenia, an inherited platelet function disorder, is caused by mutations in the ITGA2B and ITGB3 genes encoding the αIIbβ3 integrin. An acquired form of GT is also seen, which is caused by antibodies to platelet integrin αIIbβ3. This affects fibrinogen binding and blocks platelet aggregation. These patients have low platelet count with a moderate-to-severe bleeding tendency, while some patients may have normal platelet counts. It is commonly found in association with autoimmune disorders, hematological malignancies, and infections. Glanzmann thrombasthenia-like state can also be seen in immune thrombocytopenic purpura (ITP) due to presence of antibodies to αIIbβ3. These patients present with severe bleeding even with mild thrombocytopenia. We describe a patient of ITP with borderline low platelet count and severe bleeding, who posed a diagnostic challenge. However, an accurate diagnosis and suitable management helped to avoid catastrophic bleeding.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Inherited Glanzmann thrombasthenia (GT) is an autosomal recessive bleeding disorder with absent or dysfunctional platelet integrin alphaIIbbeta3 (αIIbβ3), previously known as GPIIb/IIIa, causing failure of platelets to bind to fibrinogen resulting in blocking of platelet aggregation. It is caused by mutations encoding the integrin αIIbβ3 [1]. Clinical manifestations range from easy bruising to severe life-threatening hemorrhages [2]. In contrast to this, acquired GT (aGT) is a rare hemorrhagic disorder caused by autoantibodies, alloantibodies, or paraproteins directed against platelet αIIbβ3 [3]. It is usually associated with autoimmune conditions, pregnancy, various hematological malignancies, previous platelet transfusions, and some drugs; e.g., quinidine and quinine are known to form antibodies to αIIbβ3 [3, 4]. Abciximab, eptifibatide, and tirofiban act by blocking αIIbβ3 function [5]. Acquired GT is usually accompanied by thrombocytopenia [6]. Patients may present with spontaneous moderate-to-severe bleeding tendency in the form of mucosal bleeding, menorrhagia or gastrointestinal bleeding [3, 7].
In primary immune thrombocytopenia (ITP), antibodies are formed to various platelet receptors including anti-αIIbβ3 producing a GT-like state leading to severe bleeding. Though splenectomy normalizes platelet count, the bleeding diathesis persists in these patients [5]. We report one such case of a patient who presented with life-threatening bleeding and posed a diagnostic and therapeutic challenge.
Case history
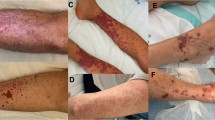
A 40-year-old female presented with severe menorrhagia since 2 years. She had 2 uneventful pregnancies with no past/family history of bleeding or of blood/platelet transfusion. A complete hemogram (CBC) showed a reduced platelet count of 93 × 109/L. Prothrombin time, activated partial thromboplastin time, thrombin time, serum fibrinogen, and von Willebrand factor (vWF) antigen were normal. Ultrasonography of abdomen and pelvis did not reveal a local cause for menorrhagia. Tests to rule out autoimmune conditions such as ANA, anti-ds DNA, and antiphospholipid antibody syndrome screen were negative. In view of bleeding disproportionate to a platelet count of 93 × 109/L, a bone marrow examination was performed which showed a normocellular marrow with adequate megakaryocytes. Hence, she was diagnosed as a case of ITP, started on steroids, and discharged.
Few months later, the patient came back with no improvement in menorrhagia and was advised therapeutic hysterectomy. She was referred to a hematologist for preoperative work-up. The CBC showed a platelet count of 95 × 109/L, but her hemoglobin (Hb) had dropped from 110 to 65 g/L due to severe menorrhagia. At this stage, she was advised to undergo a platelet function test (PFT) which was performed on Helena Platelet Aggregometer using platelet rich plasma (PRP) of the patient. A normal control PRP was also run along with patient sample. The result showed markedly low aggregation response to both high and low concentrations of ADP (10 mmol/L and 5 mmol/L, respectively), epinephrine (10 μM/L), collagen (4 μg/mL), and 500 μg/mL of arachidonic acid (AA) as compared to normal control. The aggregation pattern to high and low concentration of ristocetin (1500 and 300 μg/mL) was normal, thus ruling out vWD 2B (Fig. 1a, b, b, d). Platelet count for patient’s PRP was 120 × 109/L and for control was 200 × 109/L.
a Reduced patient aggregation response to high concentration of ADP (violet, channel 1), normal control aggregation response to high concentration of ADP (red, channel 2), normal control aggregation response to low concentration of ADP (green, channel 3), and reduced patient aggregation response to low concentration of ADP (blue, channel 4). b Normal control aggregation response to high dose of ristocetin (violet, channel 1), normal control aggregation response to low dose of ristocetin (red, channel 2), normal patient aggregation response to high dose of ristocetin (green, channel 3), normal patient aggregation response to low dose of ristocetin (blue, channel 4). c Normal control aggregation response to epinephrine (violet, channel 1), low patient aggregation response to epinephrine (red, channel 2), normal control aggregation response to collagen (green, channel 3), low patient aggregation response to collagen (blue, channel 4). d Low patient aggregation response to arachidonic acid (violet, channel 1), normal control aggregation response to arachidonic acid (red, channel 2)
These findings were suggestive of GT. Platelet receptor/antigen study was performed on BD FACSCanto II flow cytometer (FC) using antibodies CD41/61 and CD42b. This revealed a normal expression of CD41/CD61 and CD42b, thus ruling out hereditary GT and posing a diagnostic dilemma (Fig. 2).
In view of above findings of significant platelet dysfunction by aggregometry along with normal antigen expression on flow cytometry, a mixing platelet aggregation study was performed to rule out presence of inhibitors. Normal control platelets were mixed with patient’s platelet poor plasma (PPP) in a ratio of 1:4, and PFT was performed along with a control using normal PRP. Result of mixing study showed poor aggregation response to ADP, epinephrine, collagen, and AA as compared to the control (Fig. 3a, b, c) indicating the presence of antibodies in the patient’s plasma causing inhibition of aggregation in normal platelets. The patient received one unit of single donor platelets, yet her platelet count did not rise, further suggesting platelet destruction by antibodies. However, studies to isolate antibodies could not be performed due to non-availability of these tests.
Result of mixing study. a Platelet aggregation to ADP high concentration control + patient (violet, channel 1), ADP high concentration control (red, channel 2), ADP low concentration control (green, channel 3), ADP low concentration control + patient (blue, channel 4). b Platelet aggregation to epinephrine control (violet, channel1), epinephrine control + patient (red, channel2), collagen control (green, channel 3), collagen control + patient (blue, channel 4). c Platelet aggregation to arachidonic acid control + patient (violet, channel 1), arachidonic acid control (red, channel 2)
Thus, she was best regarded as ITP with possible presence of antibodies producing GT-like state, i.e., ITP with aGT. She was further investigated for an underlying malignancy but no primary focus was detected. In view of severe bleeding, therapeutic hysterectomy was deferred. She was discharged from the hospital and advised to take progesterone tablets, tranexamic acid, and eltrombopag. The patient had significant withdrawal bleeding after stopping progesterone tablets and was re-admitted. In order to alleviate her distressing symptoms, it was decided to perform an alternative, less conventional procedure, a hysteroscopic transcervical resection of endometrium with ablation. She responded to this procedure and continues to remain asymptomatic.
Discussion
Acquired GT is a rare hemorrhagic disorder. Patients often present with sudden onset of severe bleeding history [3, 7]. This is due to the formation of auto-antibodies to platelet integrin αIIbβ3 causing failure of fibrinogen and other adhesive proteins binding, leading to reduced aggregation and thus absence of thrombus formation [1, 5]. Usually, these patients present with reduced platelet count and prolonged bleeding time [3, 6]. Our patient presented with thrombocytopenia and bleeding disproportionate to platelet count. This prompted us to investigate her further, including performing PFT which showed reduced aggregation with all agonists (ADP, epinephrine, arachidonic acid) except ristocetin [3], as was noted in our patient.
PFA-100 (R) closure times, with either collagen or epinephrine, and collagen/ADP cartridges are not measurable in these patients (> 300 s) [8].
Platelet membrane glycoproteins (GPIIb/IIIa) are expressed normally, which can be studied by expression of CD41-61, on platelets by flow cytometry as was noted in our patient.
Platelet count can be reduced due to antiplatelet antibody binding to platelets causing destruction of opsonized platelets with Fc fragment recognition by phagocytic cells in the spleen [5, 8, 9]. Gianni et al. studied that IgG1 and IgG3 antibodies have a higher capacity to interact with IgG-Fc receptors than IgG2 and IgG4 [8]. Additionally, these antiplatelet antibodies can also act on megakaryocytes in the bone marrow and interfere with platelet production, resulting in low platelet counts [10].
In primary ITP, the observed bleeding phenotype is variable due to antibodies to platelet αIIbβ3 [11, 12]. However, loss of platelet aggregation occurs only if there is extensive blockade of active sites on surface αIIbβ3 by autoantibodies [11]. Close proximity of epitope on αIIbβ3 with regard to fibrinogen binding domain [1] and class and density of antibody have a bearing on severity of bleeding [7, 8]. Frelinger et al. emphasized significance of performing platelet function in ITP to predict bleeding [13]. Ghosh et al. suggested antigenic mimicry of viral/bacterial peptides to platelet glycoproteins as a possible mechanism of autoantibody formation resulting in ITP and GT-like state [14]. However, the exact molecular mechanisms in this condition remain unknown.
Based on bleeding history of sudden onset and consolidated results of all investigations, a final diagnosis of ITP with aGT was made in our patient.
The result of platelet aggregation mixing study with the patient’s PPP mixed with normal PRP suggested the possible presence of antibodies in our patient’s plasma. This mixing study is not standardized laboratory procedure; however, it can be used in these patients to further consolidate diagnosis [3, 15]. Crompton et al. studied platelet aggregometry on whole blood and performed mixing study with normal whole blood, showing that the result of aggregometry on whole blood was comparable to PRP [15]. Additionally, platelet aggregation study can also be performed by flow cytometry. Binding of PAC-1 against activated GPIIb/IIIA was found to be absent in these patients.
Also, antibodies to αIIbβ3 on platelet eluates and in plasma or serum can be identified by ELISA. The antigen capture ELISA, modified antigen capture ELISA, and monoclonal antibody immobilization of platelet antigens assay are glycoprotein-specific antigen capture assay which are commonly in use [8].
Gangaraju et al. studied platelet associated antibodies using intact platelets by immunofluorescence detection on FC [16].
Porcelijn et al. also described 2 patients with IgG1 antibodies to αIIbβ3 [7]. Acquired GT may also be observed following transfusion, various hematological malignancies or pregnancy [3].
Our patient did not have any underlying disease. Tholouli et al. described 2 cases and reviewed the literature of 12 aGT cases. They found that 9 of 12 reviewed cases were associated with malignant lymphoid and autoimmune disorders. Five of these initially presented with ITP. Similar to these cases, our patient presented as ITP. The natural history of aGT is variable. Tholouli et al. observed that spontaneous remission occurred in 2 of their reviewed cases, neither of which were associated with an underlying disorder. Two patients died from hemorrhage [3].
Successful treatment of the underlying malignancy usually induces remission of the bleeding diathesis [3]. Resistance to platelet transfusions and variable response to conventional immunosuppressive treatments pose a challenge in management of aGT, especially those not associated with an underlying disorder, as our patient. Post splenectomy, for refractory bleeding patients with ITP, displayed a persistent or worsening hemorrhagic diathesis as destruction of antibody-bound platelets is inhibited [3]. Factor VIIa (NovoSeven) has been used as treatment for some patients as in inherited GT [6]. Rituximab has been used in several patients with success [6].
Only a few published case reports are available on this disorder, thereby suggesting that this might be an under diagnosed condition. If misdiagnosed, it is life threatening. These patients are at high risk of bleeding, and even a minor surgical challenge could cause serious bleeding as seen by Dinakaran et al. [17]. In our patient, a timely diagnosis was instrumental in deferring a major surgery. Instead, a minimally invasive surgical procedure was adopted which proved life saving for this patient.
This case highlights a rare complication of a common disorder which at present is underreported. Awareness can lead to correct diagnosis and adequate reporting, thus enabling building a data base for future reference.
References
Nurden AT, Fiore M, Nurden P, Pillois X (2011) Glanzmann thrombasthenia: a review of ITGA2B and ITGB3 defects with emphasis on variants, phenotypic variability, and mouse models. Blood 118:5996–6005
Swathi J, Gowrishankar A, Jayakumar SA, Jain K (2017) A rare case of bleeding disorder: Glanzmann's thrombasthenia. Ann Afr Med 16:196–198
Tholouli E, Hay CR, O'Gorman P, Makris M (2004) Acquired Glanzmann's thrombasthenia without thrombocytopenia: a severe acquired autoimmune bleeding disorder. Br J Haematol 127:209–213
Aster RH, Bougie DW (2007) Drug-induced immune thrombocytopenia. N Engl J Med 357(6):580–587
Schneider DJ (2011) Anti-platelet therapy: glycoprotein IIb-IIIa antagonists. Br J Clin Pharmacol 72:672
Nurden AT (2019) Acquired Glanzmann thrombasthenia: from antibodies to anti-platelet drugs. Blood Rev 36:10–22
Porcelijn L, Huiskes E, Maatman R, de Haas M et al (2008) Acquired Glanzmann's thrombasthenia caused by glycoprotein IIb/IIIa autoantibodies of the immunoglobulin G1(IgG1), IgG2 or IgG4 subclass: a study in six cases. Vox Sang 95:324–330
Giannini S, Mezzasoma AM, Guglielmini G, Rossi R, Falcinelli E, Gresele P (2008) 2007. A new case of acquired Glanzmann's thrombasthenia: diagnostic value of flow cytometry. Cytometry Part B 74B:194–199
Meyer M, Kirchmaier CM, Schirmer A, Spangenberg P, Ströhl C, Breddin K (1991) Acquired disorder of platelet function associated with autoantibodies against membrane glycoprotein IIb-IIIa complex-1. Glycoprotein analysis. Thromb Haemost 65(5):491–496
Iraqi M, Perdomo J, Yan F, Choi PY, Chong BH (2015) Immune thrombocytopenia: antiplatelet autoantibodies inhibit proplatelet formation by megakaryocytes and impair platelet production in vitro. Haematologica 100(5):623–632
Van Bladel ER, Laarhoven AG, van der Heijden LB, Heitink-Pollé KM, Porcelijn L et al (2014) Functional platelet defects in children with severe chronic ITP as tested with 2 novel assays applicable for low platelet counts. Blood 123:1556–1563
Rand ML, Dean JA (1998) Platelet function in autoimmune (idiopathic) thrombocytopenic purpura. Acta Paediatr Suppl 424:57–60
Frelinger AL, Grace RF, Gerrits AJ, Berny-Lang MA, Brown T, Carmichael SL, Neufeld EJ, Michelson AD (2015) Platelet function tests, independent of platelet count, are associated with bleeding severity in ITP. Blood 126(7):873–879
Ghosh K, Kulkarni B, Shetty S, Nair S (2009) Antiplatelet antibodies in cases of Glanzmann's thrombasthenia with and without a history of multiple platelet transfusion. Indian J Hum Genet 15(1):23
Compton F, Sarode R, Rutherford C, Curtis B, De Simone N (2020) Acquired Glanzmann's thrombasthenia: diagnosis aided by platelet aggregation mixing study. Haemophilia. 26:e41–e43. https://doi.org/10.1111/hae.13914
Gangaraju R, Rets A, Smock KJ, Heikal NM (2017) Rare complication of treated immune thrombocytopenia. Am J Hematol 92:1262–1266
Dinakaran S, Edwards MP, Hampton KK (2003) Acquired Glanzmann's thrombasthenia causing prolonged bleeding following phacoemulsification. Br J Ophthalmol 87:1189–1190
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The work has been carried out at the P.D. Hinduja Hospital and Medical Research Centre, Mumbai, India, by the Departments of Clinical Hematology and Laboratory Medicine, Hematology Section
Rights and permissions
About this article
Cite this article
Padate, B., Mansukhani, D., Jijina, F. et al. Acquired Glanzmann thrombasthenia: a rare disorder. J Hematopathol 14, 145–150 (2021). https://doi.org/10.1007/s12308-020-00421-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12308-020-00421-9