Abstract
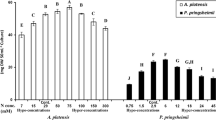
Two strains of the halophilic alga Dunaliella parva, a wild type (WT) and a transgenic strain (D-PSY) containing an exogenous phytoene synthase gene (PSY), were used to investigate the growth, carotenoid accumulation, and carotenoid antioxidant properties under nitrogen starvation, cobalt and biochar treatments. D-PSY had higher carotenoid content (1.8 times) compared to the WT. The applied stressors stimulated the carotenoid content of both WT and D-PSY especially. The carotenoids were assayed for the potential antioxidant activities by five different assays. Generally, the antioxidant activities of D-PSY carotenoids were superior to that of WT. The biochar and nitrogen treatments generally enhanced the antioxidant activities of the carotenoids, whereas cobalt came third in this respect. The D-PSY transgenic algal strain has both high carotenoids content and antioxidant properties which enhanced under the relatively lower concentrations of the applied stressors. The results have shown to lead to an accurate application of the transgenic alga as a source of potent antioxidant compounds.



Similar content being viewed by others
References
Benemann JR (1992) Microalgae Aquaculture Feeds J Appl Phycol 4:233–245
Borowitzka MA (1988) Algal growth media and sources of algal cultures. In: Borowitzka MA, Borowitzka LJ (eds) Micro-Algal Biotechnology. Cambridge University Press, New York, pp 456–465
Borowitzka MA (1992) Comparing carotenogenesis in Dunaliella and Haematococcus: implications for commercial strategies. In: Villa TG, Abalde J (eds) Profiles on Biotechnology. Servicio de Publicaciones, Universidad de Santiago, Santiago de Compostela, Spain, pp 301–310
Borowitzka LJ, Borowitzka MA (1990) Commercial production of β-carotene by Dunaliella salina in open ponds. Bull Mar Sci 47:244–252
Chen CY, Kao AL, Tsai ZC, Shen YM, Kao PH, Ng IS, Chang JS (2017) Expression of synthetic phytoene synthase gene to enhance β-carotene production in Scenedesmus sp. CPC2. Biotechnol J 12:1700204
Chokshi K, Pancha I, Ghosh A, Mishra S (2017) Salinity induced oxidative stress alters the physiological responses and improves the biofuel potential of green microalgae Acutodesmus dimorphus. Bioresour Technol 244:1376–1383
Cordero BF, Couso I, León R, Rodríguez H, Vargas MA (2011) Enhancement of carotenoids biosynthesis in Chlamydomonas reinhardtii by nuclear transformation using a phytoene synthase gene isolated from Chlorella zofingiensis. Appl Microbiol Biotechnol 91:341–351
Cordero BF, Couso I, Leon R, Rodriguez H, Vargas MA (2012) Isolation and characterization of a lycopene ε-cyclase gene of Chlorella (Chromochloris) zofingiensis. Regulation of the carotenogenic pathway by nitrogen and light. Mar Drugs 10:2069–2088
Du ST, Zhang P, Zhang RR, Lu Q, Liu L, Bao XW, Liu HJ (2016) Reduced graphene oxide induces cytotoxicity and inhibits photosynthetic performance of the green alga Scenedesmus obliquus. Chemosphere 164:499e507
Dubois M, Gillis KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
El-Sheekh MM, El-Naggar AH, Osman MEH, El-Mazaly E (2003) Effect of cobalt on growth, pigments and the photosynthetic electron transport in Monoraphidium minutum and Nitzchia perminuta. Braz J Plant Physiol 15:159–166
Fogg GE (1975) Algal Cultures and Phytoplankton Ecology. The University of Wisconsin press, USA, Text book, pp 12–36
Golldack D, Dietz KJ, Gimmleri H (1995) The effects of sudden salt stress on protein synthesis in the green alga Dunaliella parva. J Plant Physiol 146:508–514
Gong M, Bassi A (2016) Carotenoids from microalgae: A review of recent developments. Biotechnol Adv 34:1396–1412
Havaux M (2014) Carotenoid oxidation products as stress signals in plants. Plant J 79:597–606
Ismaiel MM, El-Ayouty YM, Said AA, Fathey HA (2018) Transformation of Dunaliella parva with PSY gene: Carotenoids show enhanced antioxidant activity under polyethylene glycol and calcium treatments. Biocatal Agric Biotechnol 16:378–384
Johnson EA, Schroeder WA (1996) Microbial carotenoids. Adv Biochem Eng Biotechnol 53:119–178
Lebrun M, Miard F, Scippa GS, Hano C, Morabito D, Bourgerie S (2020) Effect of biochar and redmud amendment combinations on Salix triandra growth, metal(loid) accumulation and oxidative stress response. Ecotoxicol Environ Saf 195:110466
Lustigman B, Lee LH, Weiss-Magasic C (1995) Effect of cobalt and pH on the growth of Chlamydomonas reinhardtii. Bull Environ Contam Toxicol 55:65–72
El Maghrabi DM (1997) The biotechnology of culturing Dunaliella salina for the production of some valuable metabolites, M.Sc. Thesis; Faculty of Science, Alexandria University, Egypt, pp 16–17.
Maoka T (2020) Carotenoids as natural functional pigments. J Nat Med 74:1–16
Metzner H, Rau H, Senger H (1965) Untersuchungen zur synchronisierbarkeit einzelner pigmentmangel-mutanten von Chlorella. Planta 65:186–194
Miller NJ, Sampson J, Candeias LP, Bramley PM, Rice-Evans CA (1996) Antioxidant activities of carotenes and xanthophylls. FEBS Lett 384:240–242
Mueller L, Boehm V (2011) Antioxidant activity of β-carotene compounds in different in vitro assays. Molecules 16:1055–1069
Murthy KC, Vanitha A, Rajesha J, Swamy MM, Sowmya PR, Ravishankar GA (2005) In vivo antioxidant activity of carotenoids from Dunaliella salina - a green microalga. Life Sci 76:1381–1390
Nethravathy MU, Mehar JG, Mudliar SN, Shekh AY (2019) Recent advances in microalgal bioactives for food, feed, and healthcare products: commercial potential, market space, and sustainability. Comp Rev Food Sci Food Saf 18:1882–1897
Novoveská L, Ross ME, Stanley MS, Pradelles R, Wasiolek V, Sassi JF (2019) Microalgal carotenoids: A review of production, current markets, regulations, and future direction. Mar Drugs 17:640
Oliveros E, Murasecco-Suardi P, Braun AM, Hansen H (1992) Efficiency of singlet oxygen quenching by carotenoids measured by near-infrared steady-state luminescence. Meth Enzymol 213:420–429
Orosa M, Franqueira D, Cid A, Abalde J (2005) Analysis and enhancement of astaxanthin accumulation in Haematococcus pluvialis. Bioresour Technol 96:373–378
Palace VP, Khaper N, Qin Q, Singal PK (1999) Antioxidant potentials of vitamin A and carotenoids and their relevance to heart disease. Free Radic Biol Med 26:746–761
Payne JK, Stewart JR (1988) The chemical composition of the thallus wall of Characiosiphon rivularis (Characiosiphonaceae, Chlorophyta). Phycologia 27:43–49
Pedraza-Chaverri J, Barrera D, Maldonado PD, Chirino YI, Macías-Ruvalcaba NA, Medina-Campos ON, Castro L, Salcedo MI, Hernández-Pando R (2004) S-allylmercaptocysteine scavenges hydroxyl radical and singletoxygen in vitro and attenuates gentamicin induced oxidative and nitrosative stress and renal damage in vivo. BMC Clin Pharmacol 4:5
Pereira S, Otero A (2019) Effect of light quality on carotenogenic and non-carotenogenic species of the genus Dunaliella under nitrogen deficiency. Algal Res 44:101725
Qiao-Hong Z, Xin-Hua P, Tai-Qing H, Zu-Bin X, Holden NM (2014) Effect of biochar addition on maize growth and nitrogen use efficiency in acidic red soils. Pedosphere 24:699–708
Robert RLG (1979) Growth measurements, Division rate. In: Stein RJ (ed) Physiological methods. Cambridge University Press, Cambridge, Culture and growth measurements, p 275
Safafar H, Van Wagenen J, Møller P, Jacobsen C (2015) Carotenoids, phenolic compounds and tocopherols contribute to the antioxidative properties of some microalgae species grown on industrial wastewater. Mar Drugs 13:7339–7356
Sánchez-Estudillo L, Freile-Pelegrin Y, Rivera-Madrid R, Robledo D, Narváez-Zapata JA (2006) Regulation of two photosynthetic pigment-related genes during stress-induced pigment formation in the green alga, Dunaliella salina. Biotechnol Lett 28:787–791
Shang C, Xu X, Yuan Z, Wang Z, Hu L, Alam MA, Xie J (2016) Cloning and differential expression analysis of geranylgeranyl diphosphate synthase gene from Dunaliella parva. J Appl Phycol 28:2397–2405
Shang C, Wang W, Zhu S, Wang Z, Qin L, Alam MA, Xie J, Yuan Z (2018) The responses of two genes encoding phytoene synthase (Psy) and phytoene desaturase (Pds) to nitrogen limitation and salinity up-shock with special emphasis on carotenogenesis in Dunaliella parva. Algal Res 32:1–10
Sheikhi J, Hosseini HM, Etesami H, Majidi A (2020) Biochar counteracts nitrification inhibitor DMPP-mediated negative effect on spinach (Spinacia oleracea L,) growth. Ecotoxicol Environ Saf 191:110243
Shi TQ, Wang LR, Zhang ZX, Sun XM, Huang H (2020) Stresses as first-line tools for enhancing lipid and carotenoid production in microalgae. Front Bioeng Biotechnol 8:610
Smirnoff N, Cumbes QJ (1989) Hydroxyl radical scavenging activity of compatible solutes. Phytochem 28:1057–1060
Wang HT, Yao CH, Ai JN, Cao XP, Xue S, Wang WL (2014) Identification of carbohydrates as the major carbon sink of the marine microalga Isochrysis zhangjiangensis (Haptophyta) and optimization of its productivity by nitrogen manipulation. Bioresour Technol 171:298–304
Wongsnansilp T, Yokthongwattana K, Roytrakul S, Juntawong N (2019) β-carotene production of UV-C induced Dunaliella salina under salt stress. J Pure Appl Microbiol 13:193–200
Woodall AA, Lee SWM, Weesie RJ, Jackson MJ, Britton G (1997) Oxidation of carotenoids by free radicals: relationship between structure and reactivity. Biochim Biophys Acta 1336:33–42
Ye ZW, Jiang JG, Wu GH (2008) Biosynthesis and regulation of carotenoids in Dunaliella: Progresses and prospects. Biotechnol Adv 26:352–360
Zhang Y, Yang R, Si X, Duan X, Quan X (2019a) The adverse effect of biochar to aquatic algae-the role of free radicals. Environ Pollut 248:429–437
Zhang J, Li QR, Zhang MH, You Y, Wang Y, Wang YH (2019b) Enhancement of carotenoid biosynthesis in Phaffia rhodozyma PR106 under stress conditions. Biosci Biotechnol Biochem 83:2375–2385
Zhao GR, Xiang ZJ, Ye TX, Yuan YJ, Guo ZX (2006) Antioxidant activities of Salvia miltiorrhiza and Panax notoginseng. Food Chem 99:676–774
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents on mulberry and their scavenging effects on superoxide radical. Food Chem 64:555–559
Zhu QY, Hackman RM, Ensunsa JL, Holt RR, Keen CL (2002) Antioxidative activities of Oolong tea. J Agric Food Chem 50:6929–6934
Acknowledgements
The authors would like to thank Prof. Dr. Alaa A. Said (Department of Botany and Microbiology, Faculty of Science, Zagazig University, Zagazig, Egypt) for his continuous advice, encouragement, and support. Also, we thank Dr. Chris Deduke (University of Manitoba, MB, Canada) for assistance revising this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ismaiel, M.M.S., El-Ayouty, Y.M. & Fathey, H.A. Disparity of the carotenoids antioxidant properties of wild-type and D-PSY-transgenic Dunaliella parva strains under three environmental stresses. Physiol Mol Biol Plants 27, 2151–2163 (2021). https://doi.org/10.1007/s12298-021-01077-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-021-01077-0