Abstract
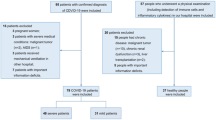
The severe acute respiratory distress syndrome-associated coronavirus-2 infection can activate innate and adaptive immune responses which may lead to harmful tissue damage, both locally and systemically. C3, a member of complement system of serum proteins, is a major component of innate immune and inflammatory responses. This study is aimed to assess serum C3 as a marker of COVID-19 severity and a predictor of disease progression. A total of 150 COVID-19 patients, confirmed by RT-PCR, and 50 healthy controls were recruited. Serum C3 levels were determined by using direct colorimetric method. Median levels of serum C3 in total cases and controls were 157.8 and 165.7 mg/dL respectively. Serum C3 although not significantly decreased, they were lower in cases when compared to controls. Similarly, significant differences were found between the groups, with severe group (140.6 mg/dL) having low levels of serum C3 protein when compared to mild (161.0 mg/dL) and moderate group (167.1 mg/dL). Interestingly, during hospitalization, significant difference between baseline (admission) and follow-up (discharge) was observed only in patients with moderate disease. Based on our results, lower levels of C3, with an increase in IL-6 and d-dimer levels, are associated with higher odds of mortality. Therefore, we would like to emphasize that measuring serum C3 levels along with other inflammatory markers might give an added advantage in early identification of patients who are prone to having a severe disease course and can help in a more effective follow-up of disease progression.



Similar content being viewed by others
References
Coronavirus [Internet]. [cited 2022 Feb 10]. Available from: https://www.who.int/westernpacific/health-topics/coronavirus
Chang D, Lin M, Wei L, Xie L, Zhu G, Dela Cruz CS, et al. Epidemiologic and clinical characteristics of novel coronavirus infections involving 13 patients outside Wuhan, China. JAMA. 2020;323:1092–3.
Li J, Gong X, Wang Z, Chen R, Li T, Zeng D, et al. Clinical features of familial clustering in patients infected with 2019 novel coronavirus in Wuhan. China Virus Res. 2020;286:198043.
Diao K, Han P, Pang T, Li Y, Yang Z. HRCT imaging features in representative imported cases of 2019 novel coronavirus pneumonia. Precis Clin Med. 2020;3:9–13.
Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–62.
O’Brien KB, Morrison TE, Dundore DY, Heise MT, Schultz-Cherry S. A protective role for complement C3 protein during pandemic 2009 H1N1 and H5N1 influenza A virus infection. PLoS ONE. 2011;6:e17377.
Tomo S, Kumar KP, Roy D, Sankanagoudar S, Purohit P, Yadav D, et al. Complement activation and coagulopathy––an ominous duo in COVID19. Expert Rev Hematol. 2021;14:155–73.
Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, Harp J, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020;220:1–13.
Li X, Zhong X, Wang Y, Zeng X, Luo T, Liu Q. Clinical determinants of the severity of COVID-19: a systematic review and meta-analysis. PLoS ONE. 2021;16:e0250602.
Lassandro G, Palladino V, Palmieri VV, Amoruso A, Del Vecchio GC, Giordano P. Covid-19 and children with immune thrombocytopenia: emerging issues. Mediterr J Hematol Infect Dis. 2020;12:e2020028.
Yadav D, Pvsn KK, Tomo S, Sankanagoudar S, Charan J, Purohit A, et al. Association of iron-related biomarkers with severity and mortality in COVID-19 patients. J Trace Elem Med Biol. 2022;74:127075.
Pvsn KK, Tomo S, Purohit P, Sankanagoudar S, Charan J, Purohit A, et al. Comparative analysis of serum zinc, copper and magnesium level and their relations in association with severity and mortality in SARS-CoV-2 patients. Biol Trace Elem Res. 2022;201(1):23–30.
Kumar PK, Banerjee M, Bajpayee A, Mandal S, Mitra P, Sharma P, et al. SARS-CoV-2 IgG antibody and its clinical correlates in convalescent plasma donors: an Indian experience. Indian J Clin Biochem. 2022;37:423–31.
Dharmalingam K, Birdi A, Tomo S, Sreenivasulu K, Charan J, Yadav D, et al. Trace elements as immunoregulators in SARS-CoV-2 and other viral infections. Indian J Clin Biochem. 2021;36(4):1–11.
Delanghe JR, De Buyzere ML, Speeckaert MM. C3 and ACE1 polymorphisms are more important confounders in the spread and outcome of COVID-19 in comparison with ABO polymorphism. Eur J Prev Cardiol. 2020;27:1331–2.
Zhang Z, Li X, Zhang W, Shi Z-L, Zheng Z, Wang T. Clinical features and treatment of 2019-nCov pneumonia patients in Wuhan: report of a couple cases. Virol Sin. 2020;35:330–6.
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan China. Clin Infect Dis. 2020. https://doi.org/10.2139/ssrn.3541136.
Tomo S, Karli S, Dharmalingam K, Yadav D, Sharma P. The clinical laboratory: a key player in diagnosis and management of COVID-19. EJIFCC. 2020;31:326–46.
Vlaar APJ, de Bruin S, Busch M, Timmermans SAMEG, van Zeggeren IE, Koning R, et al. Anti-C5a antibody IFX-1 (vilobelimab) treatment versus best supportive care for patients with severe COVID-19 (PANAMO): an exploratory, open-label, phase 2 randomised controlled trial. Lancet Rheumatol. 2020;2:e764–73.
Lim EHT, van Amstel RBE, de Boer VV, van Vught LA, de Bruin S, Brouwer MC, et al. Complement activation in COVID-19 and targeted therapeutic options: a scoping review. Blood Rev. 2023;57:100995.
Java A, Apicelli AJ, Liszewski MK, Coler-Reilly A, Atkinson JP, Kim AH, et al. The complement system in COVID-19: friend and foe? JCI Insight. 2020;5:140711.
Nandy K, Salunke A, Pathak SK, Pandey A, Doctor C, Puj K, et al. Coronavirus disease (COVID-19): a systematic review and meta-analysis to evaluate the impact of various comorbidities on serious events. Diabetes Metab Syndr. 2020;14:1017–25.
Zhang J, Wang Z, Wang X, Hu Z, Yang C, Lei P. Risk factors for mortality of COVID-19 patient based on clinical course: a single center retrospective case-control study. Front Immunol. 2021;12:581469.
de Nooijer AH, Grondman I, Janssen NAF, Netea MG, Willems L, van de Veerdonk FL, et al. Complement activation in the disease course of coronavirus disease 2019 and Its effects on clinical outcomes. J Infect Dis. 2021;223:214–24.
Fang S, Wang H, Lu L, Jia Y, Xia Z. Decreased complement C3 levels are associated with poor prognosis in patients with COVID-19: a retrospective cohort study. Int Immunopharmacol. 2020;89:107070.
Sinkovits G, Réti M, Müller V, Iványi Z, Gál J, Gopcsa L, et al. Associations between the von Willebrand factor-ADAMTS13 axis, complement activation, and COVID-19 severity and mortality. Thromb Haemost. 2022;122:240–56.
Sinkovits G, Mező B, Réti M, Müller V, Iványi Z, Gál J, et al. Complement overactivation and consumption predicts in-hospital mortality in SARS-CoV-2 infection. Front Immunol. 2021;12:663187.
Defendi F, Leroy C, Epaulard O, Clavarino G, Vilotitch A, Le Marechal M, et al. Complement alternative and mannose-binding lectin pathway activation is associated with COVID-19 mortality. Front Immunol. 2021;12:742446.
Zinellu A, Mangoni AA. Serum complement C3 and C4 and COVID-19 severity and mortality: a systematic review and meta-analysis with meta-regression. Front Immunol. 2021;12:696085.
Gralinski LE, Sheahan TP, Morrison TE, Menachery VD, Jensen K, Leist SR, et al. Complement activation contributes to severe acute respiratory syndrome coronavirus pathogenesis. mBio. 2018;9:e01753-18.
Jiang H, Chen Q, Zheng S, Guo C, Luo J, Wang H, et al. Association of complement C3 with clinical deterioration among hospitalized patients with COVID-19. IJGM. 2022;15:849–57.
Cheng W, Hornung R, Xu K, Hong YC, Li J. Complement C3 identified as a unique risk factor for disease severity among young COVID-19 patients in Wuhan, China. Sci Rep. 2021;11:7857.
Zhan H, Chen H, Liu C, Cheng L, Yan S, Li H, et al. Diagnostic value of D-Dimer in COVID-19: a meta-analysis and meta-regression. Clin Appl Thromb Hemost. 2021;27:10760296211010976.
Zhang L, Yan X, Fan Q, Liu H, Liu X, Liu Z, et al. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J Thromb Haemost. 2020;18:1324–9.
Yao Y, Cao J, Wang Q, Shi Q, Liu K, Luo Z, et al. D-dimer as a biomarker for disease severity and mortality in COVID-19 patients: a case control study. J Intensive Care. 2020;8:49.
Coomes EA, Haghbayan H. Interleukin-6 in Covid-19: a systematic review and meta-analysis. Rev Med Virol. 2020;30:1–9.
Aziz M, Fatima R, Assaly R. Elevated interleukin-6 and severe COVID-19: a meta-analysis. J Med Virol. 2020;92:2283–5.
Varela JC, Tomlinson S. Complement: an overview for the clinician. Hematol Oncol Clin North Am. 2015;29:409–27.
Shekhawat J, Gauba K, Gupta S, Purohit P, Mitra P, Garg M, et al. Interleukin-6 perpetrator of the COVID-19 cytokine storm. Indian J Clin Biochem. 2021;36:1–11.
Campbell CM, Kahwash R. Will complement inhibition be the new target in treating COVID-19-related systemic thrombosis? Circulation. 2020;141:1739–41.
Ciceri F, Beretta L, Scandroglio AM, Colombo S, Landoni G, Ruggeri A, et al. Microvascular COVID-19 lung vessels obstructive thromboinflammatory syndrome (MicroCLOTS): an atypical acute respiratory distress syndrome working hypothesis. Crit Care Resusc. 2020;22:95–7.
Macor P, Durigutto P, Mangogna A, Bussani R, De Maso L, D’Errico S, et al. Multiple-organ complement deposition on vascular endothelium in COVID-19 patients. Biomedicines. 2021;9:1003.
Ajona D, Ortiz-Espinosa S, Pio R. Complement anaphylatoxins C3a and C5a: emerging roles in cancer progression and treatment. Semin Cell Dev Biol. 2019;85:153–63.
Allegra A, Di Gioacchino M, Tonacci A, Musolino C, Gangemi S. Immunopathology of SARS-CoV-2 infection: immune cells and mediators, prognostic factors, and immune-therapeutic implications. Int J Mol Sci. 2020;21:E4782.
Fletcher-Sandersjöö A, Bellander B-M. Is COVID-19 associated thrombosis caused by overactivation of the complement cascade? A Lit Rev Thromb Res. 2020;194:36–41.
Shivshankar P, Li Y-D, Mueller-Ortiz SL, Wetsel RA. In response to complement anaphylatoxin peptides C3a and C5a, human vascular endothelial cells migrate and mediate the activation of B-cells and polarization of T-cells. FASEB J. 2020;34:7540–60.
Risitano AM, Mastellos DC, Huber-Lang M, Yancopoulou D, Garlanda C, Ciceri F, et al. Complement as a target in COVID-19? Nat Rev Immunol. 2020;20:343–4.
Didangelos A. COVID-19 hyperinflammation: what about neutrophils? mSphere. 2020;5:e00367-20.
Carvelli J, Demaria O, Vély F, Batista L, Chouaki Benmansour N, Fares J, et al. Association of COVID-19 inflammation with activation of the C5a–C5aR1 axis. Nature. 2020;588:146–50.
Paredes RM, Reyna S, Vernon P, Tadaki DK, Dallelucca JJ, Sheppard F. Generation of complement molecular complex C5b–9 (C5b–9) in response to poly-traumatic hemorrhagic shock and evaluation of C5 cleavage inhibitors in non-human primates. Int Immunopharmacol. 2018;54:221–5.
Ortega-Paz L, Capodanno D, Montalescot G, Angiolillo DJ. Coronavirus disease 2019-associated thrombosis and coagulopathy: review of the pathophysiological characteristics and implications for antithrombotic management. J Am Heart Assoc. 2021;10:e019650.
Barkoff CM, Mousa SA. Pharmacotherapy in COVID 19: potential impact of targeting the complement system. Biomedicines. 2020;9:11.
Gozzo L, Viale P, Longo L, Vitale DC, Drago F. The potential role of heparin in patients with COVID-19: beyond the anticoagulant effect. A Rev Front Pharmacol. 2020;11:1307.
Lappegård KT, Fung M, Bergseth G, Riesenfeld J, Lambris JD, Videm V, et al. Effect of complement inhibition and heparin coating on artificial surface-induced leukocyte and platelet activation. Ann Thorac Surg. 2004;77:932–41.
Hwang Y-C, Lu R-M, Su S-C, Chiang P-Y, Ko S-H, Ke F-Y, et al. Monoclonal antibodies for COVID-19 therapy and SARS-CoV-2 detection. J Biomed Sci. 2022;29:1.
Posch W, Vosper J, Noureen A, Zaderer V, Witting C, Bertacchi G, et al. C5aR inhibition of nonimmune cells suppresses inflammation and maintains epithelial integrity in SARS-CoV-2-infected primary human airway epithelia. J Allergy Clin Immunol. 2021;147:2083-2097.e6.
Mastellos DC, Pires da Silva BGP, Fonseca BAL, Fonseca NP, Auxiliadora-Martins M, Mastaglio S, et al. Complement C3 vs C5 inhibition in severe COVID-19: early clinical findings reveal differential biological efficacy. Clin Immunol. 2020;220:108598.
Mastaglio S, Ruggeri A, Risitano AM, Angelillo P, Yancopoulou D, Mastellos DC, et al. The first case of COVID-19 treated with the complement C3 inhibitor AMY-101. Clin Immunol. 2020;215:108450.
Amyndas Pharmaceuticals S.A. A Phase 2 Clinical Trial to Assess the Safety and Efficacy of Complement 3 Inhibitor, AMY-101, in Patients With Acute Respiratory Distress Syndrome Due to COVID-19 (SAVE) [Internet]. clinicaltrials.gov; 2021 Feb. Report No.: NCT04395456. Available from: https://clinicaltrials.gov/ct2/show/NCT04395456
Funding
All-India Institute of Medical Sciences Jodhpur
Author information
Authors and Affiliations
Contributions
Conceptualisation: PP, DY, SS; Material preparation, data collection and analysis: PKK, PP, SS, DY. Manuscript first draft preparation: PKK, PP, SS, DY; Writing, review and editing: PKK, PP, ST, DY, SS, JC; Sample testing: PKK, ST, AP, VLN; Patient recruiting: PKKND, PKB; Supervision: KS, MKG, SM and PS.
Corresponding author
Ethics declarations
Ethical Approval
The study was conducted as per the Declaration of Helsinki and approved by the institutional ethics committee of the All India Institute of Medical Sciences, Jodhpur, India. (Ethical approval number: AIIMS/IEC/2020–21/1090).
Informed Consent
All the participants were recruited after obtaining written informed consent. For participants that could not provide consent, consent on their behalf accordingly from the near family relative (Spouse/Parents) available at that time. All authors read and approved the final manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tomo, S., Kiran Kumar, P., Yadav, D. et al. Association of Serum Complement C3 Levels with Severity and Mortality in COVID 19. Ind J Clin Biochem 38, 447–456 (2023). https://doi.org/10.1007/s12291-023-01148-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12291-023-01148-x