Abstract
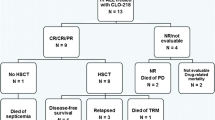
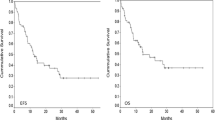
Treatment of acute myeloblastic leukemia in children, adolescents and young adults (AYA) is a challenge in low-income countries. To evaluate treatment outcomes of children (≤ 15 years) and AYA (15–30 years) diagnosed with novo AML and treated in a single center according to the AML-MA 2011 protocol. From January 2011 to December 2015, eligible patients (age ≤ 30 years) with novo AML had been enrolled on a uniform treatment protocol. The diagnosis was confirmed according to the FAB classification using the WHO 2008 criteria. Patients with WBC ≥ 50 G/L had pretreated 4 days of hydroxyurea followed by two inductions and two consolidations. Supportive care consisted of transfusion of labile blood products, antibiotics and antifungals, and patient and family education by the hygiene team. 155 patients were recruited, 41 were < 15 years old (22 boys, median age 7.8 years). Of the 114 AYA enrolled, (48 women, median age 23 years). Complete remission after two inductions was 28/41 (68.3%) of the children, including 100% of the children in the favorable group and 71/114 (62.3%) of the AYA, 22 of whom (68.7%) were in the favorable group. The number of deaths among children was 6 (14.6%). The evaluation of the AML-MA-2011 National Protocol in the age groups of children and AYA reveals that the objective of treatment is almost achieved in terms of complete remission in the two age groups.




Similar content being viewed by others
References
Guldane CS, Muhit O (2015) Treatment of acute myeloid leukemia in adolescent and young adult patients. J Clin Med 4(3):441–459
Cooper TM, Franklin J, Gerbing RB et al (2012) AAML03P1, a pilot study of the safety of gemtuzumab ozogamicin in combination with chemotherapy for newly diagnosed childhood acute myeloid leukemia: a report from the Children’s Oncology Group. Cancer 118(3):761–769
Creutzig U, Zimmermann M, Lehrnbecher T et al (2006) Less toxicity by optimizing chemotherapy, but not by addition of granulocyte colony-stimulating factor in children and adolescents with acute myeloid leukemia: results of AML-BFM 98. J Clin Oncol 24(27):4499–4506
Lange BJ, Smith FO, Feusner J et al (2008) Outcomes in CCG-2961, a children’s oncology group phase 3 trial for untreated pediatric acute myeloid leukemia: a report from the Children’s Oncology Group. Blood 111(3):1044–1053
Webb DK, Harrison G, Stevens RF et al (2001) Relationships between age at diagnosis, clinical features, and outcome of therapy in children treated in the Medical Research Council AML 10 and 12 trials for acute myeloid leukemia. Blood 98(6):1714–1720
Woods WG, Neudorf S, Gold S et al (2001) A comparison of allogeneic bone marrow transplantation, autologous bone marrow transplantation, and aggressive chemotherapy in children with acute myeloid leukemia in remission. Blood 97(1):56–62
Marielle I, Lamchaheb M, Khoubila N, Cherkaoui S, Oukache B, Hda N et al (2015) Favorable Risk Group acute myeloid leukemia in children and young adult treated in uniform protocol in Casablanca, Morocco: a preliminary analysis. Blood 126:4891
Jastaniah W, Al Ghemlas I, Al Daama S, Ballourah W, Bayoumy M, Al-Anzi F et al (2016) Clinical characteristics and outcome of childhood de novo acute myeloid leukemia in Saudi Arabia: a multicenter SAPHOS leukemia group study. Leuk Res 49:66–72
Canner J, Alonzo TA, Franklin J, Freyer DR, Gamis A, Gerbing RB et al (2013) Differences in outcomes of newly diagnosed acute myeloid leukemia for adolescent/young adult and younger patients: a report from the Children’s Oncology Group. Cancer 119(23):4162–4169
Creutzig U, Harbott J, Sperling C, Ritter J, Zimmermann M, Löffler H et al (1995) Clinical significance of surface antigen expression in children with acute myeloid leukemia: results of study AML-BFM-87. Blood 86(8):3097–3108
Gibson BE, Webb DK, Howman AJ, De Graaf SS, Harrison CJ, Wheatley K, United Kingdom Childhood Leukaemia Working Group and the Dutch Childhood Oncology Group (2011) Results of a randomized trial in children with acute myeloid leukaemia: medical research council AML12 trial. Br J Haematol 155(3):366–376
Tsukimoto I, Tawa A, Horibe K, Tabuchi K, Kigasawa H, Tsuchida M et al (2009) Risk-stratified therapy and the intensive use of cytarabine improves the outcome in childhood acute myeloid leukemia: the AML99 trial from the Japanese Childhood AML Cooperative Study Group. J Clin Oncol 27(34):4007–4013
Creutzig U, van den Heuvel-Eibrink MM, Gibson B, Dworzak MN, Adachi S, de Bont E et al (2012) Diagnosis and management of acute Myeloid Leukemia in children and adolescents: recommendations from an international expert panel. Blood 120(16):3187–3205
Rubnitz JE (2012) How I treat pediatric acute Myeloid Leukemia. Blood 119:5980–5988
Gamis AS, Alonzo TA, Meshinchi S, Sung L, Gerbing RB, Raimondi SC et al (2014) Gemtuzumab ozogamicin in children and adolescents with de novo acute myeloid leukemia improves event-free survival by reducing relapse risk: results from the randomized phase III Children’s Oncology Group trial AAML0531. J Clin Oncol 2014(32):3021–3032
Rubnitz JE, Inaba H (2012) Childhood acute myeloid leukaemia. Br J Haematol 159(3):259–276
Creutzig U, Büchner T, Sauerland MC, Zimmermann M, Reinhardt D, Döhner H, Schlenk RF (2008) Significance of age in acute myeloid leukemia patients younger than 30 years: a common analysis of the pediatric trials AML-BFM 93/98 and the adult trials AMLCG 92/99 and AMLSG HD93/98A. Cancer 112(3):562–571
Pession A, Masetti R, Rizzari C, Putti MC, Casale F, Fagioli F, AIEOP AML Study Group et al (2013) Results of the AIEOP AML 2002/01 multicenter prospective trial for the treatment of children with acute myeloid leukemia. Blood 122(2):170–178
Gibson BE, Wheatley K, Hann IM et al (2005) Treatment strategy and long-term results in paediatric patients treated in consecutive UK AML trials. Leukemia 19(12):2130–2138
Rubnitz JE, Inaba H, Dahl G et al (2010) Minimal residual disease-directed therapy for childhood acute myeloid leukaemia: results of the AML02 multicentre trial. Lancet Oncol 11(6):543–552
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not declare any conflict of interest
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Housou, B., Cherkaoui, S., Lamchahab, M. et al. Outcome of Acute Myeloid Leukemia in Children Adolescents and Young Adults Treated with an Uniform Protocol in Casablanca, Morocco. Indian J Hematol Blood Transfus 35, 255–259 (2019). https://doi.org/10.1007/s12288-018-1013-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12288-018-1013-2